What is N-Bromosuccinimide?
Jul 2,2020
General Description
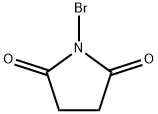
N-Bromosuccinimide is five-membered cyclic dicarboximide compound having a bromo substituent on the nitrogen atom. N-Bromosuccinimide (NBS) has been extensively used both in the bromination and oxidation of many classes of organic compounds. Introduced originally by Ziegler and his co-workers. N-Bromosuccinimide is a brominated succinimide used as a chemical reagent in radical substitution and electrophilic addition reactions in organic synthesis.
Uses
NBS is a chemical reagent used in organic chemistry in electrophilic addition reactions and radical substitution. In bromination of olefins; in oxidation of alcohols to aldehydes and ketones and of aldehydes to acid bromides.
N-Bromosuccinimide (NBS) in combination with LiBr can also be used in the dibromination of alkenes, alkynes, and allenes.21 Even though NBS also serves as a bromination reagent via a radical pathway, it can also be a source of electrophilic Br+. The dibromination reaction proceeds within minutes at room temperature in THF and leads to the products in good yields (up to 94%). In addition, the protocol can be used for the dibromination of a range of methylenecyclopropanes of type 15 to form the 2,3-dibrominated products 16 (equation 4).
Preparation
NBS is commercially available. It can also be synthesized in the laboratory. To do so, sodium hydroxide and bromine are added to an ice-water solution of succinimide. The NBS product precipitates and can be collected by filtration.
Crude NBS gives better yield in the Wohl-Ziegler reaction. In other cases, impure NBS (slightly yellow in color) may give unreliable results. It can be purified by recrystallization from 90–95 °C water (10 g of NBS for 100 mL of water).
Wohl–Ziegler bromination
The Wohl–Ziegler reaction is a chemical reaction that involves the allylic or benzylic bromination of hydrocarbons using an N-bromosuccinimide and a radical initiator.
The mechanism by which the Wohl-Ziegler reaction proceeds was proposed by Paul Goldfinger in 1953, and his reaction mechanism is one of two proposed pathways through which aliphatic, allylic, and benzylic bromination with N-bromosuccinimide (NBS) occurs.It has been shown that the Goldfinger mechanism is the proper mechanism as opposed to the previously accepted mechanism proposed by George Bloomfield, which, though consistent during selectivity studies, turned out to be overly simplistic.
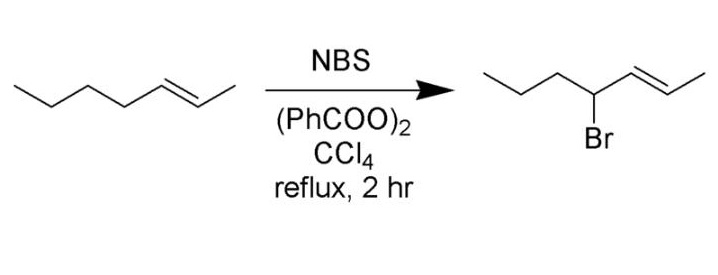
Best yields are achieved with N-bromosuccinimide in carbon tetrachloride solvent. Several reviews have been published.
In a typical setup, a stoichiometric amount of N-bromosuccinimide solution and a small quantity of initiator are added to a solution of the substrate in CCl4, and the reaction mixture is stirred and heated to the boiling point. Initiation of the reaction is indicated by more vigorous boiling; sometimes the heat source may need to be removed. Once all N-bromosuccinimide (which is denser than the solvent) has been converted to succinimide (which floats on top) the reaction has finished. Due to the high toxicity and ozone-depleting nature of carbon tetrachloride, trifluorotoluene has been proposed as an alternative solvent suitable for the Wohl-Ziegler bromination.
The corresponding chlorination reaction cannot generally be achieved with N-chlorosuccinimide, although more specialized reagents have been developed, and the reaction can be achieved industrially with chlorine gas.
Toxicity
Poison by intraperitoneal route. An irritating poison to skin, eyes, and mucous membranes. Reacts explosively with adme, dtallyl sulfide, and hydrazine hydrate. Explosive reaction with propiononitrile after heating to 105℃ for 24 hours. Violent reaction with dtbenzoyl peroxide + 4-tok acid. When heated to decomposition it emits toxic fumes of Brand NOx.
- Related articles
- Related Qustion
- How does NBS work in organic chemistry? Apr 17, 2024
N-bromosuccinimide is a five-membered cyclic dicarboximide compound having a bromo substituent on the nitrogen atom.
- N-Bromosuccinimide:Chemical synthesis,Reactions Mar 20, 2023
N-Bromosuccinimide (NBS) is a convenient source of bromine for both radical substitution and electrophilic addition reactions.
Isopropanol is also known as isopropyl alcohol. It is the simplest secondary alcohol and is one of the isomers of n-propanol. It is a kind of flammable liquid which is colorless with strong smell.....
Jul 1,2020Organic ChemistryPantothenic acid is a precursor of coenzyme A, which serves as a cofactor for a variety of enzyme-catalyzed reactions involving transfer of acetyl groups. The final step in the synthesis of acetylcholine consists of the choline acetylase....
Jul 6,2020Organic reagentsN-Bromosuccinimide
128-08-5You may like
N-Bromosuccinimide manufacturers
- N-Bromosuccinimide
-

- $0.00 / 25KG
- 2025-12-18
- CAS:128-08-5
- Min. Order: 1KG
- Purity: 99%
- Supply Ability: 2000mt
- N-Bromosuccinimide
-

- 2025-12-17
- CAS:128-08-5
- Min. Order:
- Purity: 0.99
- Supply Ability:
- N-Bromosuccinimide
-

- $1.00 / 1PCS
- 2025-12-11
- CAS:128-08-5
- Min. Order: 1PCS
- Purity: 99%
- Supply Ability: 10 mt