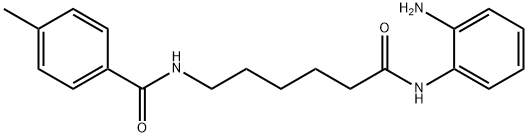
RGFP 109 synthesis
- Product Name:RGFP 109
- CAS Number:1215493-56-3
- Molecular formula:C20H25N3O2
- Molecular Weight:339.43
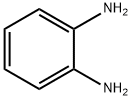
95-54-5
![Hexanoic acid, 6-[(4-Methylbenzoyl)aMino]-](/CAS/20150408/GIF/78521-43-4.gif)
78521-43-4

1215493-56-3
The general procedure for the synthesis of N-[6-(2-aminophenylamino)-6-oxohexyl]-4-methylbenzamide (R01) from o-phenylenediamine and 6-(4-methylbenzoylamino)hexanoic acid was as follows: under nitrogen protection, 6-(4-methylbenzoylamino)hexanoic acid (498 mg, 2 mmol), o-phenylenediamine (216 mg, 2 mmol), EDCI (383 mg, 2 mmol), HOBt (405 mg, 3 mmol) and triethylamine (404 mg, 4 mmol) were dissolved in dichloromethane (30 mL) and the reaction was stirred at room temperature. After completion of the reaction, the reaction mixture was washed with water and brine, dried over anhydrous sodium sulfate and concentrated under reduced pressure. The residue was purified by silica gel column chromatography to afford the target compound N-[6-(2-aminophenylamino)-6-oxohexyl]-4-methylbenzamide (227 mg, 33.9% yield) as a white solid. The product was confirmed to be >95% pure by 1H NMR (DMSO-d6) and LC-MS characterization.1H NMR (DMSO-d6) δ: 9.06 (s, 1H), 8.35 (s, 1H), 7.73 (d, J = 8.0 Hz, 2H), 7.24 (d, J = 8.0 Hz, 2H), 7.14 (t, J = 7.5 Hz, 1H), 6.86-6.89 (m, 1H), 6.70 (d, J = 7.5 Hz, 1H), 6.50 (t, J = 7.5 Hz, 1H), 4.80 (s, 2H), 3.22-3.26 (m, 2H), 2.30-2.35 (m, 5H), 1.53-1.65 (m, 4H), 1.36-1.38 (m, 2H). LC-MS (ESI): m/z 340 [M+H]+.

95-54-5
535 suppliers
$9.00/1g
![Hexanoic acid, 6-[(4-Methylbenzoyl)aMino]-](/CAS/20150408/GIF/78521-43-4.gif)
78521-43-4
14 suppliers
inquiry

1215493-56-3
105 suppliers
$49.00/5mg
Yield: 33.9%
Reaction Conditions:
with benzotriazol-1-ol;1-ethyl-(3-(3-dimethylamino)propyl)-carbodiimide hydrochloride;triethylamine in dichloromethane at 0;Inert atmosphere;
Steps:
7
N-[6-(2-aminophenylamino)-6-oxohexyl]-4-methylbenzamide (R01) A mixture of 6-(4-methylbenzamido)hexanoic acid (498 mg, 2 mmol), o-phenylenediamine (216 mg, 2 mmol), EDCI (383 mg, 2 mmol), HOBt (405 mg, 3 mmol), and triethylamine (404 mg, 4 mmol) in dichloromethane (30 mL) was stirred at room temperature under nitrogen overnight. The reaction mixture was washed with water and brine, dried over Na2SO4, and evaporated. The residue was purified by chromatography on silica gel to give the title compound (227 mg, 33.9%) as a white solid. 1H NMR (DMSO): δ 9.06 (s, 1H), 8.35 (s, 1H), 7.73 (d, J=3.0 Hz, 2H), 7.24 (d, J=3.0 Hz, 2H), 7.14 (1H, J=3.0, d), 6.86-6.89 (m, 1H), 6.70 (d, J=3.0 Hz, 1H), 6.50 (M, 1H), 4.80 (s, 2H), 3.22-3.26 (m, 2H), 2.30-2.35 (m, 5H), 1.53-1.65 (m, 4H), 1.36-1.38 (m, 2H). LC-MS: 340 (MH)+. purity>95%.
References:
REPLIGEN CORPORATION US2012/94971, 2012, A1 Location in patent:Page/Page column 40
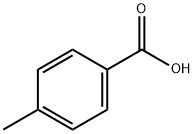
99-94-5
609 suppliers
$9.00/1g

1215493-56-3
105 suppliers
$49.00/5mg
874-60-2
418 suppliers
$10.00/5g

1215493-56-3
105 suppliers
$49.00/5mg
60-32-2
597 suppliers
$5.00/10g

1215493-56-3
105 suppliers
$49.00/5mg
![Ethanamine, N-[(3,5-dichlorophenyl)methylene]-2,2-diethoxy-](/CAS/20210305/GIF/1000210-73-0.gif)
1000210-73-0
0 suppliers
inquiry

1215493-56-3
105 suppliers
$49.00/5mg