| Identification | More | [Name]
Darifenacin hydrobromide | [CAS]
133099-07-7 | [Synonyms]
{1-[2-(2,3-dihydrobenzofuran-5-yl)ethyl]-3-pyrrolidnyl}-2,2-diphenylacetamide hydrobromide
darifenacin hydrobromide
DARIFENACIN HYDROBROMIDE 99.0%
Darifenacin hydrobromide (Under R&D)
(3S)-1-[2-(2,3-Dihydro-5-benzofuranyl)ethyl]-a-a-diphenyl-3-pyrrolidineacetamide Hydrobromide
Emsele
Enablex
DARIFENACIN HBR
Darifenacin Hydrobromid
(3S)-1-[2-(2,3-Dihydro-5-benzofuranyl)ethyl]-α-a-diphenyl-3-pyrrolidineacetamide Hydrobromide
Emselex | [EINECS(EC#)]
603-705-7 | [Molecular Formula]
C28H29BrN2O2 | [MDL Number]
MFCD08141803 | [Molecular Weight]
505.446 | [MOL File]
133099-07-7.mol |
| Chemical Properties | Back Directory | [Appearance]
White Solid | [Melting point ]
228-2300C | [alpha ]
25D -30.3° (c = 1.0 in methylene chloride) | [storage temp. ]
-20°C | [solubility ]
DMSO: soluble20mg/mL, clear | [form ]
powder | [color ]
white to beige | [Optical Rotation]
[α]/D +41 to +49°, c = 1 in methylene chloride | [Usage]
Used as a medication to treat urinary incontinence. It works by blocking the M3 muscarinic acetylcholine receptor | [InChIKey]
UQAVIASOPREUIT-YEZAQYPZNA-N | [SMILES]
C(C1C=CC=CC=1)(C1C=CC=CC=1)([C@@H]1CCN(CCC2=CC=C3OCCC3=C2)C1)C(=O)N.Br |&1:13,r| | [CAS DataBase Reference]
133099-07-7(CAS DataBase Reference) |
| Safety Data | Back Directory | [WGK Germany ]
3 | [HS Code ]
2934990002 | [Storage Class]
11 - Combustible Solids | [Hazard Classifications]
Eye Irrit. 2
Skin Irrit. 2
STOT SE 3 |
| Hazard Information | Back Directory | [Description]
Darifenacin, an orally active, once a day selective M3
receptor antagonist, was launched for the treatment of overactive
bladder in patients with symptoms of urge urinary
incontinence, urgency and frequency. The drug selectively
inhibits M3 receptor in the detrusor muscle while sparing the M1 and M2 receptors that are believed to be involved
in central nervous system and cardiovascular function respectively.
The compound was originally developed by
Pfizer and licensed to Novartis and Bayer. | [Chemical Properties]
White Solid | [Uses]
acute lymphoblastic leukemia therapeutic | [Uses]
calcium replenisher | [Uses]
Darifenacin hydrobromide is used as a medication to treat urinary incontinence. It works by blocking the M3 muscarinic acetylcholine receptor.
| [Definition]
ChEBI: The hydrobromide salt of darifenacin. A selective antagonist for the M3 muscarinic acetylcholine receptor, which is primarily responsible for bladder muscle contractions, it is used in the management of urinary incontinence. | [Brand name]
Enablex (Novartis). | [General Description]
Darfenacin (Enablex),(s)-2-{1-[2-(2,3-dihydrobenzofuran-5-yl)ethyl]-3-pyrrolidinyl}-2,2-diphen-ylacetamide, is an antimuscarinicagent that has selectivity for the M3 muscarinicsubtype receptor. By competitively blocking of the muscarinicreceptors results in a reduction of the smoothmuscle tone, allowing for greater volume of urine to bestored in the bladder. This results in less urinary incontinence,urgency, and frequency. It is a white to almostwhite, crystalline powder, with a molecular weight of507.5. Darifenacin is metabolized by the isozymesCYP2D6 and CYP3A4 with the primary metabolic routesbeing monohydroxylation of the dihydrobenzofuran ring,opening of the dihydrobenzofuran ring, and N-dealkylationof the pyrrolidine nitrogen. | [Biochem/physiol Actions]
Darifenacin hydrobromide is an antispasmodic muscarinic antagonist, selective for blocking the M3 muscarinic acetylcholine receptor, which is primarily responsible for bladder muscle contractions. Darifenacin hydrobromide has 9 and 12-fold greater affinity for M3 compared to M1 and M5, respectively, and 59-fold greater affinity for M3 compared to both M2 and M4. Darifenacin is used clinically to treat urinary incontinence and overactive bladder syndrome. | [Synthesis]
The synthesis of
darifenacin is depicted in Scheme 5. Commercially
available (2S,4R)-(-)-4-hydroxy-2-pyrrolidinecarboxylic acid
(17), anhydrous cyclohexanol and 2-cyclohexen-1-one were
heated at 154oC to give de-carboxylated compound 18 in 69
% yield. The 3-(R)-hydroxypyrrolidine (18) was N-tosylated
with p-toluenesulfonyl chloride in pyridine yielding compound
19 in 26 % yield . The N-tosylated alcohol 19 was
subjected to Mitsunobu reaction in the presence of methyl ptoluenesulfonate,
triphenylphosphine and diethyl azodicarboxylate
(DEAD) in THF to afford N-tosyl-3(S)-(tosyloxy)
pyrrolidine (20) in 70% yield, which was then condensed
with 2,2-diphenylacetonitrile with NaH in refluxing toluene
to give 2,2-diphenyl-2-[1-(p-toluenesulfonyloxy)pyrrolidin-
2(S)-yl]acetonitrile (21). The tosyl group of 21 was removed
with 48% HBr and phenol in refluxing water to yield 2,2-
diphenyl-2-[2(S)-pyrrolidinyl] acetonitrile as its corresponding
hydrogen bromide salt (22), which was coupled to 2-(2,
3-dihydrobenzofuran-5-yl) acetic acid (23) by treatment with
carbonyldiimidazole (CDI) in ethyl acetate to the corresponding
amide 24 in a quantitative yield. The amide (24)
was dissolved in toluene and reduced with sodium borohydride
in THF with slow addition of boron trifluoride THF complex to keep the temperature below 10??C to give free
amine in 88% yield. The free amine was converted to corresponding
hydrogen bromide salt (25) with 48% HBr in
methanol. Compound 25 was hydrolyzed with potassium
hydroxide in refluxing 2-methyl-butan-2-ol for twenty hours
to give acetamide which was crystallized from toluene as a
toluene solvated form in 84% yield. Finally, the toluene solvated
compound was converted to darfenacin hydrobromide
(IV) with 48% HBr in 2-methyl-butan-2-ol.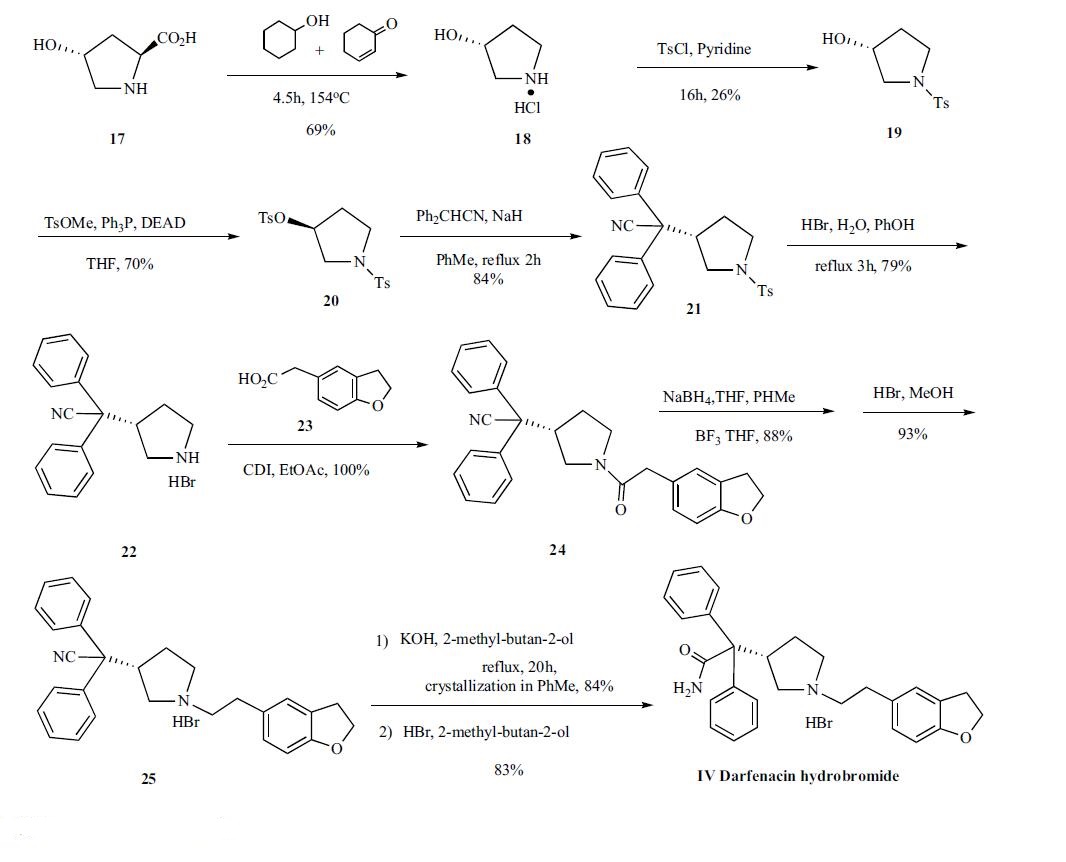
| [References]
[1]. hegde ss1,choppin a,bonhaus d,briaud s,loeb m,moy tm,loury d,eglen rm.functional role of m2 andm3muscarinic receptorsin the urinary bladder of rats in vitro and in vivo.br j pharmacol.1997 apr;120(8):1409-18.
[2]. brann mr1,ellis j,jrgensen h,hill-eubanks d,jones sv. muscarinicacetylcholinereceptorsubtypes: localization and structure/function.prog brain res.1993;98:121-7.
[3]. miller dw1,hinton m,chen f.evaluation of drug efflux transporter liabilities of darifenacin in cell culture models of the blood-brain and blood-ocular barriers. neurourol urodyn.2011 nov;30(8):1633-8. doi: 10.1002/nau.21110. epub 2011 aug 8.
[4]. haab f1,stewart l,dwyer p.darifenacin, anm3selectivereceptorantagonist, is an effective and well-tolerated once-daily treatment for overactive bladder. eur urol.2004 apr;45(4):420-9; discussion 429. |
|
|