Moxidectin
- CAS No.
- 113507-06-5
- Chemical Name:
- Moxidectin
- Synonyms
- Moxidectin CRS;milbemycin B;Quest;Equest;proheart;Moxetine;Moxdectin;Moxicitin;Moxicodin;Moxicotin
- CBNumber:
- CB4874771
- Molecular Formula:
- C37 H53 N O8
- Molecular Weight:
- 639.82
- MDL Number:
- MFCD00866560
- MOL File:
- 113507-06-5.mol
- MSDS File:
- SDS
- TDS File:
- TDS
| Product description | Number | Pack Size | Price |
| Moxidectin European Pharmacopoeia (EP) Reference Standard | Y0000772 | 70 mg | $175 |
| Moxidectin for system suitability European Pharmacopoeia (EP) Reference Standard | Y0000747 | 10 mg | $254 |
| Moxidectin VETRANAL | 33746 | 25mg | $218 |
| Moxidectin United States Pharmacopeia (USP) Reference Standard | 1448559 | 200mg | $778 |
| Moxidectin | M3136 | 250MG | $127 |
| More product size | |||
| Melting point | 132 °C |
|---|---|
| Boiling point | 790.0±70.0 °C(Predicted) |
| Density | 1.23±0.1 g/cm3(Predicted) |
| storage temp. | Sealed in dry,Store in freezer, under -20°C |
| solubility | ≥128 mg/mL in EtOH; ≥129.4 mg/mL in DMSO; ≥3.27 mg/mL in H2O with gentle warming and ultrasonic |
| pka | 12.46±0.70(Predicted) |
| form | Solid |
| color | White to Almost white |
| optical activity | 139.1°(C=0.01 g/mI, CHCL3 , 20°C, 589nm) |
| Stability | Light Sensitive |
| InChIKey | YZBLFMPOMVTDJY-LSGXYNIPSA-N |
| SMILES | CO\N=C1/C[C@]2(C[C@@H]3CC(C\C=C(C)\C[C@@H](C)\C=C\C=C4/COC5[C@H](O)C(C)=C[C@@H](C(=O)O3)[C@]45O)O2)O[C@@H]([C@H]1C)\C(C)=C\C(C)C |
| FDA 21 CFR | 556.426 |
| FDA UNII | NGU5H31YO9 |
| NCI Drug Dictionary | moxidectin |
| ATC code | P02CX03 |
| UNSPSC Code | 41116107 |
| NACRES | NA.77 |
SAFETY
Risk and Safety Statements
| Symbol(GHS) |   GHS06,GHS09 |
|||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Signal word | Danger | |||||||||
| Hazard statements | H301-H400 | |||||||||
| Precautionary statements | P264-P270-P273-P301+P310-P391-P405 | |||||||||
| Hazard Codes | T,N | |||||||||
| Risk Statements | 25-50 | |||||||||
| Safety Statements | 45-61-24/25 | |||||||||
| RIDADR | 2588 | |||||||||
| WGK Germany | 3 | |||||||||
| RTECS | PY5438800 | |||||||||
| HazardClass | 6.1(b) | |||||||||
| PackingGroup | III | |||||||||
| HS Code | 29322090 | |||||||||
| Storage Class | 6.1C - Combustible acute toxic Cat.3 toxic compounds or compounds which causing chronic effects |
|||||||||
| Hazard Classifications | Acute Tox. 3 Oral Aquatic Acute 1 |
|||||||||
| NFPA 704 |
|
Moxidectin price More Price(42)
| Manufacturer | Product number | Product description | CAS number | Packaging | Price | Updated | Buy |
|---|---|---|---|---|---|---|---|
| Sigma-Aldrich | Y0000772 | Moxidectin European Pharmacopoeia (EP) Reference Standard | 113507-06-5 | 70 mg | $175 | 2026-04-30 | Buy |
| Sigma-Aldrich | Y0000747 | Moxidectin for system suitability European Pharmacopoeia (EP) Reference Standard | 113507-06-5 | 10 mg | $254 | 2026-04-30 | Buy |
| Sigma-Aldrich | 33746 | Moxidectin VETRANAL | 113507-06-5 | 25mg | $218 | 2026-04-30 | Buy |
| Sigma-Aldrich | 1448559 | Moxidectin United States Pharmacopeia (USP) Reference Standard | 113507-06-5 | 200mg | $778 | 2026-04-30 | Buy |
| TCI Chemical | M3136 | Moxidectin | 113507-06-5 | 250MG | $127 | 2026-04-30 | Buy |
Moxidectin Chemical Properties,Uses,Production
Description
Moxidectin is a macrocyclic lactone endectocide and a derivative of nemadectin. It reduces fecal nematode egg counts by 98.4 and 99.8% in naturally infected calves when administered subcutaneously at doses of 0.2 and 0.3 mg/kg, respectively. Moxidectin (0.2 and 0.3 mg/kg) reduces the worm burden of O. ostertagi and T. axei in the abomasum, and Cooperia species and N. helvetianus in the small intestine, of naturally infected calves. It potentiates GABA-gated currents in Xenopus oocytes expressing rat α1β2γ2 subunit-containing GABAA receptors with an EC50 value of 11.8 nM. Formulations containing moxidectin have been used in the treatment of onchocerciasis, as well as in the prevention and treatment of parasitic infections in veterinary medicine.
Uses
anthelmintic, antiparasitic
Uses
Moxidectin is a semi-synthetic milbemycin derived from nemadectin by selective oxidation followed by methyloximation. Moxidectin was patented in 1991 as an anthelmintic for internal parasite control. The presence of the methyloxime affords moxidectin a greater hydrophobicity and longer biological half-life compared to nemadectin. Moxidectin binds selectively to parasite glutamate-gated chloride ion channels and disrupts neurotransmission leading to paralysis and death of the parasite.
Uses
Moxidectin is a macrocyclic lactone and semisynthetic derivative of nemadectin. Moxidectin is a parasiticide used for the prevention and control of heartworm and intestinal worms. Moxidectin is is fou nd in veterinary medicine used to treat animals such as dogs, cats, horses, cattle and sheep.
Definition
ChEBI: Moxidectin is a milbemycin.
Synthesis
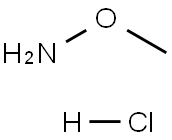
593-56-6
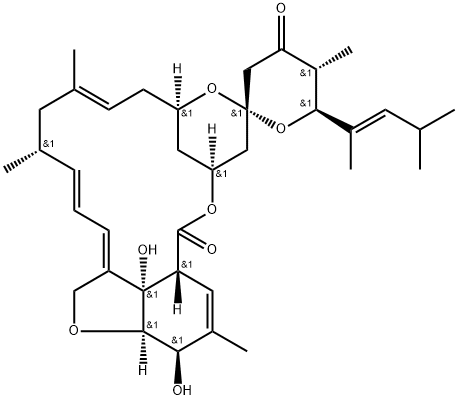
112124-81-9
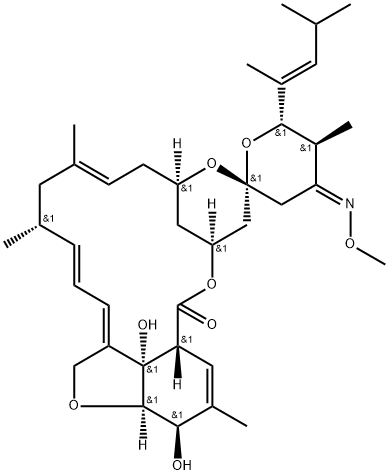
113507-06-5
1. The solid product (0.087 mol) was dissolved in 640 mL of 1,4-dioxane and anhydrous sodium acetate (0.13 mol), acetic acid (0.028 mol) and methoxyamine hydrochloride (0.14 mol) were added sequentially under nitrogen protection. The reaction mixture was stirred at room temperature for 10 hours. The progress of the reaction was monitored by TLC and after confirming the complete reaction of the ingredients, the reaction was terminated by adding 1000 mL of pure water. The reaction mixture was extracted with 1000 mL of ethyl acetate and the extraction was repeated once. The organic phases were combined, dried with anhydrous sodium sulfate and concentrated to give 78.4 g solid. 2. The above solid was dissolved in 4120 mL of ethanol and 1760 mL of pure water was added to adjust the ethanol concentration to 70% (v/v). After filtration to remove insoluble material, 7840 mL of pure water was added for dilution. Extraction was repeated with 13,720 mL of heptane. The heptane layers were combined and concentrated until crystals precipitated, stopping the concentration. Cooled to 20 °C with slow stirring, filtered to separate the crystals and dried under vacuum at 45 °C, -0.08 MPa to give a white solid (0.071 mol). 3. Dissolve the dried crystals (0.071 mol) in 1270 mL of anhydrous methanol and cool to 0 °C. A solution of p-toluenesulfonic acid (0.13 mol) in methanol (120 mL) was added slowly dropwise, keeping the reaction temperature at 0 °C. After dropwise addition, the reaction was continued for 4 h. The reaction progress was monitored by HPLC. The reaction solution was transferred to 2780 mL of saturated sodium bicarbonate solution and washed thoroughly. It was extracted twice with 1270 mL of ethyl acetate, the organic phases were combined, dried over anhydrous sodium sulfate and concentrated to give a solid product (0.054 mol). 4. The resulting solid was dissolved in 23030 mL of 70% (v/v) aqueous methanol and filtered to remove insoluble material. The filtrate was upsampled onto a HP20 macroporous resin column and eluted with 80% (v/v) methanol aqueous solution. The eluate was detected by HPLC and the fraction with 98% purity was collected. Concentrate the eluate to 35% (v/v) methanol and stop the concentration. Cooled to 20°C, stirred slowly for 5 h. The precipitate was withdrawn and dried to give moxidectin (0.015 mol, 96.2% content).
Veterinary Drugs and Treatments
In dogs and cats, moxidectin with lufenuron is indicated as a once
a month topical preventative for the prevention of heartworm, flea
adulticide, ear mites (cats) and treatment for hookworms, roundworms,
and whipworms (dogs). It has also been successfully used as
a treatment for generalized demodicosis.
In cattle, moxidectin is indicated for the treatment and control
of the following internal [adult and fourth stage larvae (L4)]
and external parasites: Gastrointestinal roundworms: Ostertagia
ostertagi (adult and L4, including inhibited larvae), Haemonchus
placei (adult), Trichostrongylus axei (adult and L4), Trichostrongylus
colubriformis (adult), Cooperia oncophora (adult), Cooperia punctata
(adult), Bunostomum phlebotomum (adult), Oesophagostomum
radiatum (adult), Nematodirus helvetianus (adult); Lungworm:
Dictyocaulus viviparus (adult and L4); Cattle Grubs: Hypoderma
bovis, Hypoderma lineatum Mites: Chorioptes bovis, Psoroptes
ovis (Psoroptes communis var. bovis); Lice: Linognathus vituli,
Haematopinus eurysternus, Solenopotes capillatus, Damalinia bovis;
Horn flies: Haematobia irritans. To control infections and to protect
from reinfection from Ostertagia ostertagi for 28 days after treatment
and from Dictyocaulus viviparus for 42 days after treatment.
In sheep, oral moxidectin is indicated for the control of
Haemonchus contortus (adult and L4), Teladosrsagia circumcincta
& trifurcata (adult and L4), Trichostrongylus colubriformis, axei, &
vitrinius (adult & L4), Cooperia curticei & oncophora (adult and L4),
Oesophagostomum columbianum & venolosum (adult & L4), and
Nematodirus battus, filicollis, & spathiger (adult & L4).
In horses and ponies, moxidectin is indicated for the treatment
and control of the following stages of gastrointestinal parasites:
Large strongyles: Strongylus vulgaris (adults and L4L5 arterial
stages); Strongylus edentatus (adults and tissue stages);
Triodontophorus brevicauda (adults); Triodontophorus serratus
(adults); Small strongyles (adults and larvae): Cyathostomum spp.
(adults); Cylicocyclus spp. (adults); Cylicostephanus spp. (adults);
Gyalocephalus capitatus (adults); undifferentiated lumenal larvae;
Encysted cyathostomes: late L3 and L4 mucosal cyathostome larvae;
Ascarids: Parascaris equorum (adults and L4 larval stages); Pin
worms: Oxyuris equi (adults and L4 larval stages); Hair worms:
Trichostrongylus axei (adults); Large-mouth stomach worms:
Habronema muscae (adults); Horse stomach bots: Gasterophilus intestinalis
(2nd and 3rd instars). When combined with praziquantel,
additional coverage against Anoplocephala spp. occurs.
References
[1] Tetrahedron Letters, 1988, vol. 29, # 21, p. 2595 - 2598
[2] Patent: CN104017001, 2016, B. Location in patent: Paragraph 0018; 0049; 0050
[3] Patent: CN104277050, 2016, B. Location in patent: Paragraph 0101-0104
Moxidectin Preparation Products And Raw materials
Raw materials
1of2
Preparation Products
| Supplier | Tel | Country | ProdList | Advantage | |
|---|---|---|---|---|---|
| AFINE CHEMICALS LIMITED | +86-0571-85134551 | sales@afinechem.com | China | 15082 | 58 |
| Shaanxi Dideu Medichem Co. Ltd | +86-29-81139210 +86-18192627656 | 1059@dideu.com | China | 3590 | 58 |
| Shaanxi Dideu Medichem Co. Ltd | +8617392709771 | 1097@dideu.com | China | 3996 | 58 |
| Wuhan Fortuna Chemical Co., Ltd | +86-27-59207850 | info@fortunachem.com | China | 5974 | 58 |
| Hebei Chuanghai Biotechnology Co,.LTD | +86-86-13131129325 +8613131129325 | sales1@chuanghaibio.com | China | 5235 | 58 |
| Hebei Chuanghai Biotechnology Co., Ltd | +8615531151365 | mina@chuanghaibio.com | China | 18126 | 58 |
| Hebei Jingbo New Material Technology Co., Ltd | +8619931165850 | hbjbtech@163.com | China | 1000 | 58 |
| airuikechemical co., ltd. | +86-18353166132 | sales02@airuikechemical.com | China | 983 | 58 |
| Hebei Longbang Technology Co., LTD | +86-18633929156 | admin@hblongbang.com | China | 972 | 58 |
| HebeiShuoshengImportandExportco.,Ltd | +86-18532138899 | L18532138899@163.com | China | 939 | 58 |
Related articles
- A macrocyclic lactone anthelmintic: Moxidectin
- Moxidectin is a 16-membered macrocyclic lactone of the milbemycin class. The starting material for the synthesis of the drug i....
- Dec 21,2023
View Lastest Price from Moxidectin manufacturers
| Image | Update time | Product | Price | Min. Order | Purity | Supply Ability | Manufacturer | |
|---|---|---|---|---|---|---|---|---|
 |
2026-05-02 | Moxidectin
113507-06-5
|
0.99 | RongNa Biotechnology Co.,Ltd | ||||
 |
2026-05-01 | Moxidectin
113507-06-5
|
US $0.00 / kg | 1kg | 98% | Customise | Hefei Lbao Physical & Chemical Science Co.,Ltd | |
 |
2026-04-30 | Moxidectin
113507-06-5
|
US $0.00 / g | 100g | 99% | 100 kg | Xi an Biohorlden Industry Trade Co Ltd |
-

- Moxidectin
113507-06-5
- 0.99
- RongNa Biotechnology Co.,Ltd
-

- Moxidectin
113507-06-5
- US $0.00 / kg
- 98%
- Hefei Lbao Physical & Chemical Science Co.,Ltd
-

- Moxidectin
113507-06-5
- US $0.00 / g
- 99%
- Xi an Biohorlden Industry Trade Co Ltd





