PHALLACIDIN
- CAS No.
- 26645-35-2
- Chemical Name:
- PHALLACIDIN
- Synonyms
- PHALLICIDIN;PHALLACIDIN;Phallacidin
DISCONTINUED;CYCLO(ALA-2-MERCAPTO-TRP-4;phallacidin from amanita phalloides;Phallacidin from Amanita phalloides;Cyclo(Ala-
2- Mercapto- Trp- 4;5-DIHYDROXY-LEU-VAL-ERYTHRO-3-HYDROXY-D-ASP-CYS-CIS-4-HYDROXY-PRO);-3-hydroxy-d-alpha-aspartyl-l-cysteinyl-cis-4-hydroxy-l-prolyl)cyclic(2-6)-s;cyclic(l-alanyl-2-mercapto-l-tryptophyl-4,5-dihydroxy-l-leucyl-l-valyl-erythro
- CBNumber:
- CB6136097
- Molecular Formula:
- C37H50N8O13S
- Molecular Weight:
- 846.9
- MDL Number:
- MFCD00063359
- MOL File:
- 26645-35-2.mol
- MSDS File:
- SDS
- TDS File:
- TDS
| Product description | Number | Pack Size | Price |
| Phallacidin from Amanita phalloides ≥85% | P8157 | 1mg | $389 |
| Phallacidin ≥90% | 18146 | 1mg | $236 |
| Phallacidin ≥90% | 18146 | 1mg | $214 |
| PHALLACIDIN 95.00% | PEP0003782 | 1MG | $354.9 |
| PHALLACIDIN 95.00% | PEP0003782 | 5MG | $1062.38 |
| More product size | |||
| Density | 1.1678 (rough estimate) |
|---|---|
| refractive index | 1.7400 (estimate) |
| solubility | DMF: Soluble; DMSO: Soluble; Methanol: Soluble; Water: Soluble |
| form | White to off-white powder. |
| color | White to off-white |
| Appearance | Solid Powder |
| biological source | Amanita phalloides |
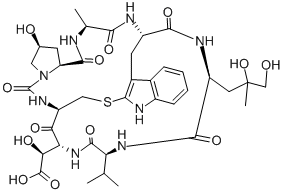
| Sequence | Ala-Trp-Leu-Val-{D-Asp}-Cys-{Hyp} (Sulfide bridge: Trp2- Cys6) |
| InChIKey | KUBDTFZQCYLLGC-UHFFFAOYSA-N |
| SMILES | CC(C)C1NC(=O)C(CC(C)(O)CO)NC(=O)C2Cc3c(SCC(NC(=O)C(NC1=O)C(O)C(O)=O)C(=O)N4CC(O)CC4C(=O)NC(C)C(=O)N2)[nH]c5ccccc35 |
| UNSPSC Code | 12352202 |
| NACRES | NA.56 |
SAFETY
Risk and Safety Statements
| Symbol(GHS) |  GHS06 |
|||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Signal word | Danger | |||||||||
| Hazard statements | H300+H310+H330 | |||||||||
| Precautionary statements | P260-P262-P264-P280-P302+P352+P310-P304+P340+P310 | |||||||||
| PPE | Eyeshields, Faceshields, Gloves, type P3 (EN 143) respirator cartridges | |||||||||
| Hazard Codes | T+ | |||||||||
| Risk Statements | 26/27/28 | |||||||||
| Safety Statements | 22-36/37/39-45 | |||||||||
| RIDADR | 1544 | |||||||||
| WGK Germany | 3 | |||||||||
| RTECS | GT8943000 | |||||||||
| HazardClass | 6.1(a) | |||||||||
| PackingGroup | I | |||||||||
| HS Code | 2934999090 | |||||||||
| Storage Class | 6.1A - Combustible acute toxic Cat. 1 and 2 very toxic hazardous materials |
|||||||||
| Hazard Classifications | Acute Tox. 1 Inhalation Acute Tox. 2 Dermal Acute Tox. 2 Oral |
|||||||||
| NFPA 704 |
|
PHALLACIDIN price More Price(5)
| Manufacturer | Product number | Product description | CAS number | Packaging | Price | Updated | Buy |
|---|---|---|---|---|---|---|---|
| Sigma-Aldrich | P8157 | Phallacidin from Amanita phalloides ≥85% | 26645-35-2 | 1mg | $389 | 2024-03-01 | Buy |
| Cayman Chemical | 18146 | Phallacidin ≥90% | 26645-35-2 | 1mg | $236 | 2026-04-30 | Buy |
| Cayman Chemical | 18146 | Phallacidin ≥90% | 26645-35-2 | 1mg | $214 | 2024-03-01 | Buy |
| American Custom Chemicals Corporation | PEP0003782 | PHALLACIDIN 95.00% | 26645-35-2 | 1MG | $354.9 | 2021-12-16 | Buy |
| American Custom Chemicals Corporation | PEP0003782 | PHALLACIDIN 95.00% | 26645-35-2 | 5MG | $1062.38 | 2021-12-16 | Buy |
PHALLACIDIN Chemical Properties,Uses,Production
Description
Phallacidin is a natural mycotoxin first isolated from the death cap mushroom, A. phalloides. It binds filamentous actin (F-actin) like phalloidin , but contains a carboxyl group for coupling reactions.
Uses
Phallacidin is a natural mycotoxin first isolated from the death cap mushroom. It binds filamentous actin like phalloidin.
Uses
Toxin Phallacidin is a weak fluorescent bicyclic peptide probe in aquous solution, Which is isolated from Amanita phalloides mushroom and could be used for blocking F-actin staining by labeled phallotoxins, which promotes actin polymerization.
Definition
ChEBI: A homodetic bicyclic heptapeptide having a sulfide bridge.
Biochem/physiol Actions
Phallacidin, which is a bicyclic toxin from the Amanita phalloides mushroom, has been shown to inhibit F-actin degradation by proteolytic enzymes including trypsin and α-chymotrypsin.
Enzyme inhibitor
This cyclic heptapeptide toxin (FWfree-acid = 846.92 g/mol; CAS 26645-35-2) from the poisonous green fungus Amanita phalloides stabilizes actin filaments and inhibits actin filament depolymerization. NBD phallacidin is a high-affinity, filamentous actin probe consisting of rabbit actin that is conjugated by reacting its thiol group with 4-chloro-7- nitrobenzofurazan, a yellow-green fluorescent dye. NBD phallacidin (Excitation at 465 nm; Emission at 536 nm) selectively stains F-actin un a manner that is superior to antibody staining, preferably in fixed and permeabilized samples. See Phalloidin
References
[1] L S BARAK. Fluorescence staining of the actin cytoskeleton in living cells with 7-nitrobenz-2-oxa-1,3-diazole-phallacidin.[J]. Proceedings of the National Academy of Sciences of the United States of America, 1980, 77 2: 980-984. DOI: 10.1073/pnas.77.2.980
[2] L S BARAK W W W R R Yocum. In vivo staining of cytoskeletal actin by autointernalization of nontoxic concentrations of nitrobenzoxadiazole-phallacidin.[J]. Journal of Cell Biology, 1981, 89 2: 368-372. DOI: 10.1083/jcb.89.2.368
PHALLACIDIN Preparation Products And Raw materials
Raw materials
Preparation Products
| Supplier | Tel | Country | ProdList | Advantage | |
|---|---|---|---|---|---|
| Aladdin Scientific | tp@aladdinsci.com | United States | 57505 | 58 | |
| DAYANG CHEM (HANGZHOU) CO.,LTD | +86-88938639 +86-17705817739 | info@dycnchem.com | China | 53802 | 58 |
| Shanghai Tauto Biotech Co., Ltd. | 021-51320588 | tauto@tautobiotech.com | China | 3988 | 66 |
| Sigma-Aldrich | 021-61415566 800-8193336 | orderCN@merckgroup.com | China | 51395 | 80 |
| Shanghai EFE Biological Technology Co., Ltd. | 021-65675885 18964387627 | info@efebio.com | China | 9803 | 58 |
| Chengdu Huachun Technology Co., Ltd | 400-1166-196 15982826727 | cdhxsj@163.com | China | 14603 | 60 |
| Beijing Solarbio Science & Tecnology Co., Ltd. | 010-50973130 18101056239 | 3193328036@qq.com | China | 29661 | 68 |
| Shanghai Zhen Li Biological Technology Co., Ltd | 021-16621358918; 18516310516 | zhenlipharma888@163.com | China | 19066 | 58 |
| Shanghai Jizhi Biochemical Technology Co. Ltd. | 021-4009004166/18616739031 18616739031 | 3007523370@qq.com | China | 52678 | 58 |
| Shanghai Hongye Biotechnology Co. Ltd | 400-9205774 | sales@glpbio.cn | China | 6777 | 58 |