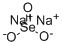
Sodium selenite
- CAS No.
- 10102-18-8
- Chemical Name:
- Sodium selenite
- Synonyms
- SodiumSelenitanhydrous;natriumselenit;SODIUM SELENITE;SODIUM SELENIATE;Disodium selenite;Sodiumselenite,99%;Sodium Selenite AR;SODIUMSELENIUMOXIDE;SODIUMSELENITE,POWDER;sodium selenite .5H2O
- CBNumber:
- CB9852600
- Molecular Formula:
- Na2O3Se
- Molecular Weight:
- 172.94
- MDL Number:
- MFCD00003489
- MOL File:
- 10102-18-8.mol
- MSDS File:
- SDS
| Melting point | >350 °C (lit.) |
|---|---|
| Density | 3.1[at 20℃] |
| vapor pressure | 0.133Pa at 20℃ |
| storage temp. | Store at RT. |
| solubility | Water (Slightly) |
| form | powder |
| color | white |
| Odor | Odorless |
| Water Solubility | 950 g/L (20 ºC) |
| Sensitive | Hygroscopic |
| Merck | 14,8673 |
| Exposure limits |
ACGIH: TWA 0.2 mg/m3 NIOSH: IDLH 1 mg/m3; TWA 0.2 mg/m3 |
| InChIKey | BVTBRVFYZUCAKH-UHFFFAOYSA-L |
| CAS DataBase Reference | 10102-18-8(CAS DataBase Reference) |
| EWG's Food Scores | 2-3 |
| FDA UNII | HIW548RQ3W |
| NCI Drug Dictionary | sodium selenite |
| ATC code | A12CE02 |
| EPA Substance Registry System | Disodium selenite (10102-18-8) |
SAFETY
Risk and Safety Statements
| Symbol(GHS) |   GHS06,GHS09 |
|||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Signal word | Danger | |||||||||
| Hazard statements | H300+H330-H315-H317-H319-H411 | |||||||||
| Precautionary statements | P260-P273-P280-P302+P352-P304+P340+P310-P305+P351+P338 | |||||||||
| Hazard Codes | T+,N,Xi | |||||||||
| Risk Statements | 23-28-31-43-51/53-36/37/38 | |||||||||
| Safety Statements | 28-36/37-45-61-28A-36-26 | |||||||||
| RIDADR | UN 2630 6.1/PG 1 | |||||||||
| WGK Germany | 2 | |||||||||
| RTECS | VS7350000 | |||||||||
| TSCA | Yes | |||||||||
| HazardClass | 6.1 | |||||||||
| PackingGroup | I | |||||||||
| HS Code | 28429010 | |||||||||
| Toxicity | LD50 orally in rats: 7 mg/kg (Cummins, Kimura) | |||||||||
| NFPA 704 |
|
Sodium selenite Chemical Properties,Uses,Production
Chemical Properties
White colored crystalline solid. It could form pentahydrate. It is stable in air and could lose water in dry air. It is soluble in water, insoluble in ethanol. The theoretical selenium content is 45.7%.
Application
- Nutrition enhancer.
- Determination of alkaloids and preparation of red colored glass and glaze.
- Feed additive.
- Selenium is a component of glutathione peroxidase, maintains the function of the cell membrane through oxidation, and increase the production of protein lipid based endogenous antioxidants. It involves in the energy conversion, effects the metabolism, plays an important role in the fat emulsification and absorption, and in the absorption of a variety of vitamins. At the same time, it involves in the synthesis of coenzyme A and coenzyme Q, effects the function of other enzyme systems. It influences the metabolism of amino acids, protein synthesis, carbohydrate metabolism, and biological oxidation. The lack of selenium in livestock and poultry seriously affect the growth and reproduction.
Preparation
(1) Selenite reacts with sodium hydroxide to produce sodium selenite. Further refinement and addition of dilution agent to obtain the final product.
(2) Selenium reacts with nitric acid to produce selenium oxide, which reacts with sodium hydroxide to produce sodium selenite. Followed drying and grinding to obtain the final product.
Category
Toxic substance
Toxicity grading
Highly toxic
Acute toxicity
Oral-rat LD50: 7 mg/kg; oral-mouse LD50: 7.08 mg/kg.
Flammability Hazardous Characteristics
Non-combustible; gives off toxic fume of selenium oxide in a fire.
Storage and Transport
Stored in a well-ventilated area, at low temperature, and dry condition; food should be stored and transported separately.
Extinguishing agent
Sand, water.
Occupational standards
TLV-TWA 0.2 mg (Selenium)/m3; STEL 0.3 mg (Selenium)/m3.
Chemical Properties
White to light beige crystals
Chemical Properties
Sodium selenite is a white crystalline substance.
Uses
Used in reducing carcinogenesis with potency greater than selenomethionene
Uses
Removing green color from glass during its manufacture; alkaloidal reagent.
Uses
Sodium selenite Na2O3Se is used in the production of red enamels and in the manufacture of clear red glass.
Definition
ChEBI: An inorganic sodium salt composed of sodium and selenite ions in a 2:1 ratio.
General Description
A white colored crystalline solid. Soluble in water and more dense than water. Contact may irritate skin, eyes and mucous membranes. Toxic by ingestion, inhalation and skin absorption.
Air & Water Reactions
Soluble in water.
Reactivity Profile
Sodium selenite is incompatible with strong acids.
Hazard
Toxic by ingestion.
Health Hazard
Toxic by ingestion. In humans, a concentration of 5 ppm in food or 0.5 ppm in milk or water has been estimated to be dangerous. Elemental selenium has low acute systemic toxicity, but dust or fumes can cause serious irritation of the respiratory tract.
Fire Hazard
May burn but will not ignite readily. When heated to decomposition, may emit toxic fumes of selenium and sodium oxide. Stable in air.
Flammability and Explosibility
Non flammable
Safety Profile
Poison by ingestion, intraperitoneal, intravenous, subcutaneous, intracervical, parenteral, and intramuscular routes. Experimental teratogenic and reproductive effects. Questionable carcinogen. Human mutation data reported. When heated to decomposition it emits toxic fumes of Se and Na2O. See also SELENIUM COMPOUNDS.
Potential Exposure
Sodium selenite is used in glass manufacturing and as an alkaloidal reagent; for removing green color from glass during its manufacture; alkaloidal reagent; reagent in bacteriology; testing germination of seeds; decorating porcelain; as a livestock feed additive.
Shipping
UN2630 Selenates or Selenites, Hazard Class: 6.1; Labels: 6.1-Poisonous materials.
Purification Methods
Crystallise sodium selenite from a saturated aqueous solution where its solubility is 68% at 20o to give the pentahydrate salt. This yields the anhydrous salt on heating at 40o. [Fehér in Handbook of Preparative Inorganic Chemistry (Ed. Brauer) Academic Press Vol I p 431 1963].
Incompatibilities
The aqueous solution is a medium strong base. Reacts with water, strong acids; hot surfaces; causing decomposition and a toxic hazard.
Waste Disposal
Liquid or solid: make a strongly acidic solution using hydrochloric acid. Slowly add sodium sulfite to the cold solution. Stir mixture producing sulfur dioxide. Heat, forming dark-gray selenium and black tellurium. Let stand overnight. Filter and dry. Ship to supplier.
Sodium selenite Preparation Products And Raw materials
Raw materials
Preparation Products
10102-18-8(Sodium selenite)Related Search:
1of4