Vonoprazan fumarate
|
|
Vonoprazan fumarate 속성
- 저장 조건
- Store at -20°C
- 용해도
- insoluble in H2O; insoluble in EtOH; ≥18.9 mg/mL in DMSO
- 물리적 상태
- 고체
- 물리적 상태
- 단단한 모양
- InChIKey
- ROGSHYHKHPCCJW-WLHGVMLRSA-N
- SMILES
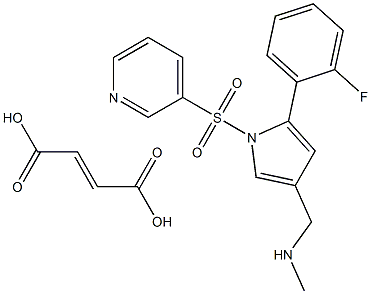
- N1(S(C2=CC=CN=C2)(=O)=O)C(C2=CC=CC=C2F)=CC(CNC)=C1.C(O)(=O)/C=C/C(O)=O
안전
- 위험 및 안전 성명
- 위험 및 사전주의 사항 (GHS)
Vonoprazan fumarate C화학적 특성, 용도, 생산
개요
Vonoprazan fumarate (Takecab®), discovered and developed by Takeda and Otsuka, was approved by the PMDA of Japan in December 2014, and is indicated for the treatment of gastric ulcer, duodenal ulcer and reflux esophagitis. Vonoprazan fumarate has a novel mechanism of action called potassium-competitive acid blockers, which competitively inhibit the binding of potassium ions to H+, K+-ATPase (also known as the proton pump) in the final step of gastric acid secretion in gastric parietal cells. Vonoprazan does not inhibit Na+, K+-ATPase activity even at concentrations 500 times higher than that of their IC50 values against gastric H+, K+-ATPase activity. Furthermore, the drug is unaffected by the gastric secretory state, unlike PPIs.용도
Vonoprazan Fumarate is a novel potassium-?competitive acid blocker for the treatment of acid-?related diseases.주요 응용
Vonoprazan fumarate is an oral, newly synthesised potassium-competitive acid blocker (P-CAB) with antisecretory activity. It is also a proton pump inhibitor (PPI) reversibly inhibiting H+/K+, ATPase. It is mainly used in the treatment of acid-related diseases such as GERD and peptic ulcer disease.Synthesis
Commercially available 2-fluoroacetophenone (283) was brominated to yield a-bromo-acetophenone derivative 284. This compound was treated with ethyl 2-cyanoacetate (285) under basic conditions to provide ketoester 286 in essentially quantitative yield. Next, intramolecular condensation of 286 upon treatment of 4 M HCl furnished the tri-substituted pyrrole 287 in 53% yield. Reduction of the chloride under hydrogenolytic conditions facilitated arrival at pyrrole 288, albeit in just 18% yield. Subsequent diisobutylaluminium hydride (DIBAL) reduction, followed by the oxidation with tetrapropylammonium perruthenate (TPAP) and 4-methylmorpholine N-oxide (NMMO) afforded the corresponding aldehyde 290 in 60% yield across the 2 steps. Next, N-pyrrole substitution with pyridine- 3-sulfonyl chloride 291 gave rise to N-sulfonylpyrrole 292 in 82% yield. Reductive amination of 292 afforded amine 293, which was treated with fumaric acid (294) via co-crystallization to provide vonoprazan fumarate (XXXVI) in 74% for the two steps.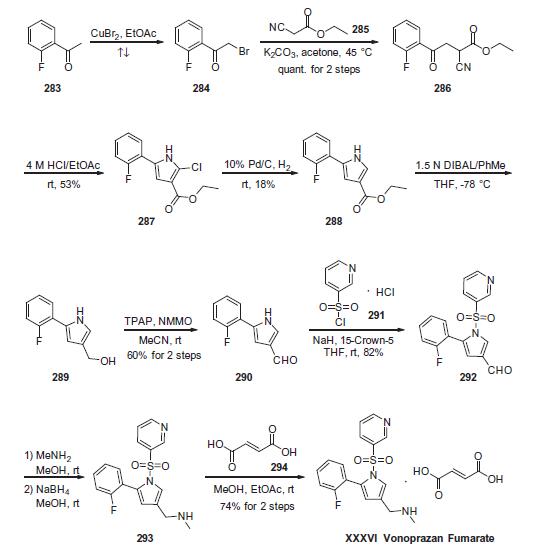
Vonoprazan fumarate 준비 용품 및 원자재
원자재
준비 용품
Vonoprazan fumarate 공급 업체
글로벌( 472)공급 업체
| 공급자 | 전화 | 이메일 | 국가 | 제품 수 | 이점 |
|---|---|---|---|---|---|
| Beijing Mesochem Technology Co.,Ltd | +8613651027935 |
rachel@mesochem.com | China | 191 | 58 |
| TAIZHOU YUXIN BIOTECHNOLOGY CO,.LTD | +86-576-88902229;+86-0576-88902229 +8613968687450 |
yuxin@yuxchem.com | China | 158 | 58 |
| Hangzhou ICH Biofarm Co., Ltd | +86-0571-28186870; +undefined8613073685410 |
sales@ichemie.com | China | 990 | 58 |
| Beijing Hope Pharmaceutical Co., Ltd. | +86-010-67886402 +8613611125266 |
market@hopelife.cn | China | 71 | 58 |
| Wuhan Fortuna Chemical Co., Ltd | +86-027-59207850 |
info@fortunachem.com | China | 5987 | 58 |
| Hebei Mojin Biotechnology Co., Ltd | +86 13288715578 +8613288715578 |
sales@hbmojin.com | China | 12446 | 58 |
| XI'AN TIANGUANGYUAN BIOTECH CO., LTD. | +86-029-86333380 18829239519 |
sales06@tgybio.com | China | 905 | 58 |
| Changzhou Rokechem Technology Co., Ltd. | 18758118018 |
sales001@rokechem.com | China | 255 | 58 |
| Firsky International Trade (Wuhan) Co., Ltd | +8615387054039 |
admin@firsky-cn.com | China | 436 | 58 |
| shandong perfect biotechnology co.ltd | +86-53169958659; +8618596095638 |
sales@sdperfect.com | China | 294 | 58 |
Vonoprazan fumarate 관련 검색:
에소메프라졸
Vonoprazan
Vonoprazan FuMarate
Donepezil Hydrochloride
ETOFIBRATE
Fosaprepitant dimeglumine
Lansoprazole
Pantoprazole
2-PYRIMIDINAMINE, 4-(3,4-DIHYDRO-1-METHYL-2(1H)-ISOQUINOLINYL)-N-(4-FLUOROPHENYL)-5,6-DIMETHYL-, MONOHYDROCHLORIDE
R-(+)-Lansoprazole
Teneligliptin Hydrobromide
Trelagliptin succinate
Trelagliptin
Alogliptin benzoate
Linagliptin
AGOMELATINE
ALOGLIPTIN(ALOGLIPTINE, ALOGLIPTINA)
5-(2-Fluorophenyl)-1-(pyridin-3-ylsulfonyl)-1H-pyrrole-3-carbaldehyde