- Fosfomycin tromethamine
-

- $0.00 / 1Kg/Bag
-
2024-09-18
- CAS:78964-85-9
- Min. Order: 1KG
- Purity: 98%-102%; EP
- Supply Ability: 100KG
- Fosfomycin Tromethamine
-

- $0.00 / 1Kg
-
2024-04-08
- CAS:78964-85-9
- Min. Order: 1Kg
- Purity: 99.9%
- Supply Ability: 200tons
|
| | Fosfomycin tromethamine Basic information |
| Product Name: | Fosfomycin tromethamine | | Synonyms: | (3-methyloxiranyl)-phosphonicaci(2r-cis)-phosphonicacicompd.with2-amino-2-(hydroxy;fosfomycincompd.withtrometamol;fosfomycintrometamolsalt;methyl)-1,3-propanediol(1:1);Tromethamine fosfosmycin;Fosfomycin Tromethamine (800 mg);FosfoMycin troMethaMine API;Fosfomycin trometamolUSP,
98.0%-102.0% | | CAS: | 78964-85-9 | | MF: | C7H18NO7P | | MW: | 259.19 | | EINECS: | 279-018-8 | | Product Categories: | API;Antibiotics | | Mol File: | 78964-85-9.mol | 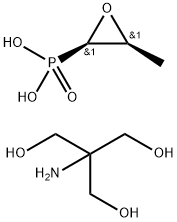 |
| | Fosfomycin tromethamine Chemical Properties |
| Melting point | 122-125oC | | storage temp. | Hygroscopic, Refrigerator, under inert atmosphere | | solubility | Very soluble in water, slightly soluble in ethanol (96 per cent) and in methanol, practically insoluble in acetone. | | form | Solid | | color | White to Off-White | | Stability: | Hygroscopic | | InChIKey | QZJIMDIBFFHQDW-LMLSDSMGSA-N |
| | Fosfomycin tromethamine Usage And Synthesis |
| Chemical Properties | White to off-white powder | | Uses | Phosphonic acid derivative; antibacterial. | | Uses | Fosfomycin Tromethamine is used as an antibiotic compound in the treatment of urinary tract infections. | | Definition | ChEBI: Fosfomycin tromethamine is a phosphonoacetic acid. | | Brand name | Monurol (Zambon). | | General Description | Fosfomycin tromethamine (Monurol) is a phosphonic acidepoxide derivative that was initially isolated from fermentationsof Streptomyces spp. Making the tromethamine salt greatly expanded the therapeutic utility of this antibacterial because water solubilityincreased enough to allow oral administration.
Fosfomycin is a broad-spectrum, bactericidal antibacterialthat inhibits the growth of E. coli, S. aureus, andSerratia, Klebsiella, Citrobacter, Enterococcus, andEnterobacter spp. at a concentration less than 64 mg/L.Currently fosfomycin is recommended as single-dose therapyfor uncomplicated urinary tract infections. It possessesin vitro efficacy similar to that of norfloxacin and trimethoprim-sulfamethoxazole.
Fosfomycin covalently inactivates the first enzyme in thebacterial cell wall biosynthesis pathway, UDP-N-acetylglucosamineenolpyruvyl transferase (MurA) by alkylation ofthe cysteine-115 residue. The inactivation reaction occursthrough nucleophilic opening of the epoxide ring. Resistanceto fosfomycin can occur through chromosomal mutationsthat result in reduced uptake or reduced MurA affinity for theinhibitor. Plasmid-mediated resistance mechanisms involveconjugative bioinactivation of the antibiotic with glutathione.The frequency of resistant mutants in in vitro studies hasbeen low, and there appears to be little cross-resistance betweenfosfomycin and other antibacterials. |
| | Fosfomycin tromethamine Preparation Products And Raw materials |
|