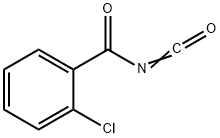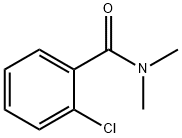
2-chloro-N,N-dimethyl-benzamide synthesis
- Product Name:2-chloro-N,N-dimethyl-benzamide
- CAS Number:6526-67-6
- Molecular formula:C9H10ClNO
- Molecular Weight:183.63
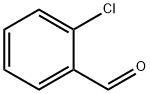
89-98-5
668 suppliers
$15.00/25g
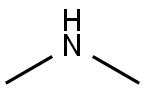
124-40-3
547 suppliers
$18.00/100ml

6526-67-6
6 suppliers
$30.00/20mg
Yield:6526-67-6 37%
Reaction Conditions:
Stage #1: dimethyl aminewith carbon dioxideGreen chemistry;
Stage #2: 2-chloro-benzaldehydewith tert.-butylhydroperoxide;iron(II) chloride tetrahydrate;tetrabutylammomium bromide in water;acetonitrile at 60; for 16 h;Inert atmosphere;Green chemistry;
Steps:
general procedure for the oxidative amidation ofaldehydes with amine:
General procedure: The amine (1.2 mmol) was taken in a reaction vial with a magnetic bar and stirred vigorously under CO2 atmosphere (balloon) about 15 to 30 minutes until corresponding carbamate ammonium salts were formed. Subsequently, CO2 atmosphere was taken off and FeCl2*4H2O (10 mol %), TBAB (10 mol%), aldehyde (1.0 mmol), TBHP (70 wt % in H2O, 1.1 mmol) and acetonitrile (2 mL) were added to the reaction vial. The reaction vial was capped under inert atmosphere and allowed to stir at 40 °C-60 °C and the progress of the reaction was monitored by TLC. After completion of reaction, the volatiles were removed under the reduced pressure, and the crude product was purified by column chromatography on silica.
References:
Bathini, Thulasiram;Rawat, Vikas S.;Bojja, Sreedhar [Tetrahedron Letters,2015,vol. 56,# 41,art. no. 46650,p. 5656 - 5660] Location in patent:supporting information
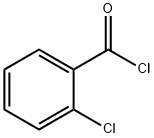
609-65-4
346 suppliers
$21.00/25g

124-40-3
547 suppliers
$18.00/100ml

6526-67-6
6 suppliers
$30.00/20mg
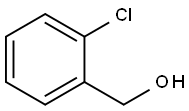
17849-38-6
291 suppliers
$6.00/10g
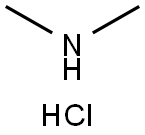
506-59-2
557 suppliers
$5.00/5G

6526-67-6
6 suppliers
$30.00/20mg
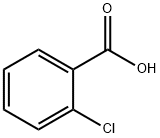
118-91-2
675 suppliers
$14.00/25g

6526-67-6
6 suppliers
$30.00/20mg
![Benzamide, 2-chloro-N-(2-chlorobenzoyl)-N-[(dimethylamino)carbonyl]-](/CAS/20210305/GIF/104417-75-6.gif)