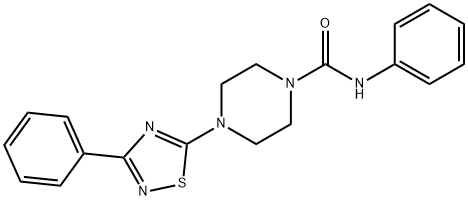
JNJ 1661010 synthesis
- Product Name:JNJ 1661010
- CAS Number:681136-29-8
- Molecular formula:C19H19N5OS
- Molecular Weight:365.45
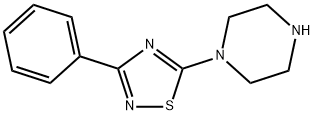
306935-14-8
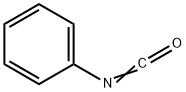
103-71-9

681136-29-8
GENERAL STEPS: A solution of 1-(3-phenyl-1,2,4-thiadiazol-5-yl)piperazine (1.00 g, 4.06 mmol) and triethylamine (0.565 mL, 4.06 mmol) in tetrahydrofuran (20 mL) was added to a 25 mL round bottom flask. The reaction mixture was cooled to 0°C in an ice bath, followed by slow dropwise addition of phenyl isocyanate (0.529 mL, 4.87 mmol). After the dropwise addition was completed, the ice bath was removed and the reaction mixture was stirred at room temperature for 3 hours. Upon completion of the reaction, diisopropyl ether (40 mL) was added to the mixture to precipitate the product. The solid was collected by filtration and recrystallized with a mixed solvent of hexane and ethyl acetate (1:1, v/v) to afford N-phenyl-4-(3-phenyl-1,2,4-thiadiazol-5-yl)piperazine-1-carboxamide 1.19 g in 80.4% yield as a white solid. The structure of the product was confirmed by 1H-NMR (CDCl3): δ 3.70 (8H, s), 6.43 (1H, s), 7.06-7.11 (1H, m), 7.29-7.46 (7H, m), 8.17-8.21 (2H, m).

306935-14-8
42 suppliers
$108.75/500mg

103-71-9
0 suppliers
$20.19/5g

681136-29-8
83 suppliers
$59.00/5mg
Yield: 80.4%
Reaction Conditions:
with triethylamine in tetrahydrofuran at 0 - 20; for 3 h;
Steps:
25
Example 25; N-Phenyl-4-(3-phenyl-l,2,4-thiadiazol-5-yl)piperazine-1-carboxamide; To a solution of 1-(3-phenyl-1,2,4-thiadiazol-5-yl)piperazine (1.00 g, 4.06 mmol) and triethylamine (0.565 ml, 4.06 mmol) in tetrahydrofuran (20 ml) was added, under ice-cooling, phenyl isocyanate (0.529 ml, 4.87 mmol), and the mixture was stirred at room temperature for 3 hours. To the reaction mixture was added diisopropyl ether (40 ml), and the solid was separated by filtration. The resulting solid was recrystallized from a mixed solvent of hexane and ethyl acetate to give 1.19 g (80.4%) of the desired product as a solid. 1H-NMR (CDCl3) δ; 3.70 (8H, s), 6.43 (1H, s), 7.06 - 7.11 (1H, m), 7.29 - 7.46 (7H, m), 8.17 - 8.21 (2H, m).
References:
Takeda Pharmaceutical Company Limited EP1813606, 2007, A1 Location in patent:Page/Page column 32-33
![Ethanamine, N-[(3,5-dichlorophenyl)methylene]-2,2-diethoxy-](/CAS/20210305/GIF/1000210-73-0.gif)