- Albendazole
-

- $6.00 / 1kg
-
2024-09-23
- CAS:54965-21-8
- Min. Order: 1kg
- Purity: 99%
- Supply Ability: 2000KG/Month
- Albendazole
-

- $100.00 / 1KG
-
2024-09-23
- CAS:54965-21-8
- Min. Order: 1KG
- Purity: 99%
- Supply Ability: 10000kg
- Albendazole
-

- $100.00/ kg
-
2024-09-23
- CAS:54965-21-8
- Min. Order: 1kg
- Purity: 99%
- Supply Ability: 5000
Question and answer - Q:What is albendazole used for?
- A:Albendazole is a synthetic nitroimidazole with a broad spectrum of antinematodal activity similar to mebendazole but with anti....
- Jul 10,2024
|
| | Albendazole Chemical Properties |
| Melting point | 208-210 °C | | density | 1.2561 (rough estimate) | | refractive index | 1.6740 (estimate) | | storage temp. | 2-8°C | | solubility | Practically insoluble in water, freely soluble in anhydrous formic acid, very slightly soluble in methylene chloride, practically insoluble in ethanol (96 per cent). | | form | Solid | | pka | 10.72±0.10(Predicted) | | color | White to Off-White | | Water Solubility | 0.75mg/L(209 ºC) | | Merck | 14,210 | | BCS Class | 4 | | InChI | InChI=1S/C12H15N3O2S/c1-3-6-18-8-4-5-9-10(7-8)14-11(13-9)15-12(16)17-2/h4-5,7H,3,6H2,1-2H3,(H2,13,14,15,16) | | InChIKey | HXHWSAZORRCQMX-UHFFFAOYSA-N | | SMILES | C(OC)(=O)NC1NC2=CC(SCCC)=CC=C2N=1 | | CAS DataBase Reference | 54965-21-8(CAS DataBase Reference) |
| | Albendazole Usage And Synthesis |
| description | Albendazole (ALBENZA) is an orally administered broad-spectrum anthelmintic. Albendazole chewable tablet is included in the World Health Organization (WHO) list of essential medicines as an intestinal anthelminthic and antifilarial medicine. Albendazole tablet was developed by SmithKline Animal Health Laboratories and approved by U.S. Food and Drug Administration (FDA) in 1996.
Albendazole has capability of completely killing the eggs of whipworm and hookworm as well as partially killing Ascaris’ eggs; it can also get rid of various kinds of nematodes parasitizing inside animal bodies, and has effects on either getting rid of or directly killing tapeworms and cysticerci. It is thus useful in the treatment of hydatid and the nervous system (cysticercosis) caused by infection of pork worm, and also in the treatment of hookworm, roundworm, pinworm, nematode trichinella, tapeworm, whipworm and stercoralis nematode. | | Pharmacology and mechanism of action | Albendazole is a benzimidazole carbamate derivative which is structurally related to mebendazole. It was originally introduced as a veterinary drug in 1975 and later as a human anthelminthic drug. It has a wide spectrum of activity against intestinal nematodes (hook worm, Ascaris lumbricoides, Enterobius vermicularis, Strongyloides stercoralis, Trichuris trichiura and Capillaria philippinensis), systemic nematodes (Trichinella spiralis and cutaneous larva migrans) and cestodes (Echinococcus granulosis, E. multilocularis and neurocysticercosis) [1]. Albendazole is active against both larval and adult stages of intestinal nematodes and ovicidal against Ascaris lumbricoides and Trichuris trichiura [1]. Its main metabolite, albendazole sulphoxide, may largely be responsible for the pharmacological effects of the drug.
The mechanism of action of albendazole is similar to that of other benzimidazoles (see mebendazole). | | Indications | Single or mixed infections caused by Ascaris lumbricoides, Enterobius vermicularis, Ancylostoma duodenale, Trichuris trichiura. Albendazole may be effective against cutaneous larva migrans and Strongyloides stercoralis, but controlled studies are needed to confirm its advantage over thiabendazole. Limited data indicate that albendazole is useful in neurocysticercosis [2,3]. Albendazole seems to be the drug of choice for the treatment of inoperable hydatid cases, but its long term benefit needs further assessment. | | Side effects | After a single dose treatment of albendazole 400 mg, minor and transient side effects such as epigastric pain and diarrhoea were seen. Less than 6% of treated patients experience these effects [4]. During the treatment of hydatid disease, where higher doses are used for longer time periods, side effects were more common and severe. In two randomized double-blind multicentre phase I and II studies [5,6] involving 139 patients given high doses of the drug, about 20% of the patients showed side effects. These included elevation of serum transaminases (6 patients), leucopenia (3 patients), gastrointestinal symptoms (8 patients), severe headache (4 patients), loss of hair (3 patients), urticaria and itching (2 patients), fever and fatigue (1 patient), and thrombocytopenia (1 patient).
| | Contraindications and precautions | There are no known contraindications to the drug during single dose treatment of intestinal nematodes. During treatment against hydatid disease, liver transaminases, leukocyte and platelet counts must be monitored regularly. | | Preparations | • Zentel® (SmithKline Beecham). Tablets 400 mg. Suspension 2%.
• Eskazole® (SmithKline Beecham). Tablets 400 mg.
| | Solubility | It is slightly soluble in acetone or chloroform, but insoluble in water; it is also slightly soluble in hot diluted hydrochloric acid, and soluble in methanol, ethanol, and acetic acid.
| | Pharmacodynamics | Albendazole is a kind of benzimidazole derivatives. It is rapidly metabolized in vivo into the sulfoxide, sulfone and 2-polyamine sulfone alcohol. It can selectively and irreversibly suppress the glucose uptake of intestinal nematodes, thus resulting in endogenous glycogen depletion of the worm; at the same time, it also inhibit the activity of fumarate reductase, and thus preventing the generation of adenosine triphosphate, finally causing death of the parasites.
Similar as mebendazole, through causing the denaturation of cytoplasmic microtubules of intestinal parasites and binding to the tubulin, it causes clogging of intracellular transport, causing the accumulation of Golgi endocrine particles; cytoplasm is further gradually dissolved, causing the final death of the parasites.
This product can completely kill hookworm eggs, pinworm eggs, spin wool eggs, tapeworm eggs and cysticercosis whip eggs and partially kill Ascaris’ eggs.
| | Usage and dosage | Roundworm disease and pinworm disease: take 400mg daily per time.
Hookworm disease, whipworm disease, stercoralis disease: take 400mg each time, 2 times a day, continue for 3 days.
Trichinella spirallis disease, take 600mg or 800mg daily in 2 times; a course of treatment is one week.
Neurocysticercosis take daily 18mg/kg in 2 times of oral administration; 10 days is a course of treatment; you can also extend the course to one month according to the specific disease situation.
Hydatid disease: take 20mg/kg daily in 2 times of oral administration with the course being 1 month; it usually needs multiple times of treatment.
| | Chemical Properties | It is white to light yellow crystalline powder and is odorless. Its melting point is 207-211°C (decomposition). It is slightly soluble in organic solvents but insoluble in water. Rats by oral administration: LD50: 2.4g/kg; sheep by oral administration: LD50:100mg/kg.
| | Uses | Albendazole is a drug used to treat infections caused by parasites. It can be given to treat a rare brain infection (neurocysticercosis) or it can be given to treat a parasitic infection that causes important diarrhea (microsporidiosis). | | synthesis | The synthesis of albendazole can be performed in four steps starting with 4-nitro aniline. In the first step ortho-nitro aniline (2) is substituted with thiocyanate in para position in the presence of an oxidant such as chlorine. The reaction follows an aromatic substitution in which 2-nitrothiocyanoaniline (3) is produced. Afterwards bromopropane reacts with the product from step one to obtain 4-(Propylthio)-2-nitroaniline (4). This is achieved by the mechanism of a nucleophile substitution. In the third step the nitro-group is reduced to the amine to prepare the molecule for the imidazoleformation. Following the methylcyancarbamate is added. Now the two amine groups of 4-(propylthio) benzene-1,2-diamine (5) react with the carbamate while building an heteroaromatic system. Simultaneously ammonia is condensed and albendazole is generated.
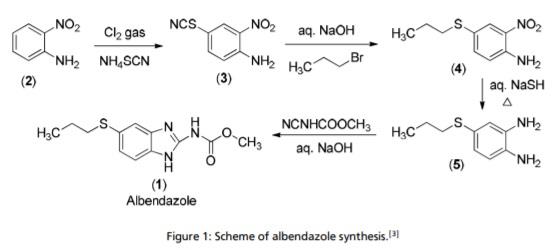
The synthesis of albendazole | | References | 1. Rossignol JF, Mausonneuve H (1984). Albendazole: a new concept in the control of intestinal helminthiasis. Gastroenterol Clin Biol, 8, 569–576.
2. Cruz M, Cruz I, Horton J (1991). Albendazole versus praziquantel in the treatment of cerebral cysticercosis: Clinical evaluation. Trans R Soc Trop Med Hyg, 85, 244–247.
3. Takayanagui OM, Jardim E (1992). Therapy of neurocysticercosis. Comparison between albendazole and praziquantel. Arch Neurol, 49, 290–294.
4. Coulaud JP, Rossignol JF (1984). Albendazole: a new single dose anthelminthic. Acta Tropica (Basel), 41, 87–90.
5. Davies A, Dixon H, Pawlowski ZS (1989). Multicentre clinical trials of benzimidazole carbamates in human cystic echinococcosis (phase 2). Bull World Health Organ, 67, 503–508.
6. Davis A, Pawlski ZS, Dixon H (1986). Multicentre clinical trials of benzimidazole-carbamates in human echinococcosis. Bull WHO, 64, 383–388.
| | Chemical Properties | Albendazole occurs as a white to faintly yellowish powder. It is practically insoluble in water and alcohol, very slightly soluble in ether and dichloromethane, but freely soluble in anhydrous formic acid. | | Originator | Zentel ,SK and F ,France ,1981 | | Uses | Albendazole is a derivative of benzimidazole, is a drug with a broad antihelmintic spectrum. It exhibits an antihelmintic effect against sensitive cestodes and nematodes by blocking the process of glucose uptake by the parasites, which is expressed in the depletion of glycogen reserves and subsequent reduction in the level of adenosintriphophate. As a result, the parasite stops moving and dies. It is used upon infection of Acaris lumbricoides, Ancylostoma duodenale, Necator americanus, Enterobius vermicularis, and Trichuris trichiura. Synonyms of this drug are SKF 62979 and others. | | Indications | Albendazole appears to cause cytoplasmic microtubular
degeneration, which in turn impairs vital cellular
processes and leads to parasite death.There is some evidence
that the drug also inhibits helminth-specific ATP
generation by fumarate reductase. | | Definition | ChEBI: Albendazole is a carbamate ester that is methyl 1H-benzimidazol-2-ylcarbamate substituted by a propylsulfanyl group at position 5. It is commonly used in the treatment of parasitic worm infestations. It has a role as a tubulin modulator, a microtubule-destabilising agent and an anthelminthic drug. It is a carbamate ester, a benzimidazolylcarbamate fungicide, an aryl sulfide and a member of benzimidazoles. | | Manufacturing Process | A mixture of 6.65 g of 3-chloro-6-nitroacetanilide, 3.2 ml of propylmercaptan,
5.6 g of 50% sodium hydroxide and 100 ml of water is heated at reflux
overnight. The cooled mixture is filtered to give the desired 2-nitro-5-
propylthioaniline, MP 69.5-71.5°C after recrystallization from ethanol then
hexane-ether. NMR (CDCl3) 40%.
The aniline (2.5 g) is hydrogenated with 1.9 ml of concentrated hydrochloric
acid, 100 ml ethanol and 5% palladium-on-charcoal to give 4-propylthio-ophenylene-
diamine hydrochloride.
A mixture of 2.5 ml of 50% sodium hydroxide in 5 ml of water is added to a
mixture of 1.9 g of cyanamide, 2.2 g of methylchloroformate, 3.5 ml of water
and 3 ml of acetone over 45 minutes below 10°C, pH raised to 6.5. A molar
equivalent solution of the diamine in 100 ml of ethanol is added. The mixture
is heated until the easily volatile solvents are expelled, to about 85°C, then
maintained at this temperature with some water added for one-half hour. The
product, methyl 5-propylthio-2-benzimidazolecarbamate, is separated, washed
to give a colorless crystalline solid, MP 208-210°C. | | Brand name | Albenza (GlaxoSmithKline). | | Therapeutic Function | Anthelmintic | | Antimicrobial activity | Albendazole is active against trichostrongyles
and exhibits useful activity against tissue-dwelling larvae
of Trichinella spiralis, larvae of animal hookworms (causing
cutaneous larva migrans) and microfilariae of various filarial
species. It also exhibits some activity against cysticercosis and
hydatid stages of Echinococcus granulosus and Echinococcus
multilocularis. It has been successfully used in infections with
the protozoon Giardia lamblia and for microsporidiosis. | | General Description | Albendazole occurs as a white crystalline powder that isvirtually insoluble in water. The oral absorption of albendazoleis enhanced by a fatty meal. The drug undergoes rapidand extensive first-pass metabolism to the sulfoxide, whichis the active form in plasma. The elimination half-life ofthe sulfoxide ranges from 10 to 15 hours. Considerable biliaryexcretion and enterohepatic recycling of albendazolesulfoxide occurs. Albendazole is generally well toleratedin single-dose therapy for intestinal nematodes. The highdose,prolonged therapy required for clonorchiasis orechinococcal disease therapy can result in adverse effectssuch as bone marrow depression, elevation of hepatic enzymes,and alopecia. | | Biochem/physiol Actions | Binds to tubulin and inhibits microtubule assembly. | | Mechanism of action | Albendazole is given orally and is poorly and variably
absorbed (�5%) because of its poor water solubility.
Oral bioavailability is increased as much as five
times when the drug is given with a fatty meal instead of
on an empty stomach. Concurrent treatment with corticosteroids
increases plasma concentrations of albendazole.
The drug is rapidly metabolized in the liver to an
active sulfoxide metabolite.The half life of the metabolites
is 8 to 12 hours. | | Pharmacokinetics | Albendazole is better absorbed after oral absorption than
other benzimidazole carbamates. It is extensively metabolized
to the anthelmintically active albendazole sulfoxide,
producing plasma concentrations of the metabolite
of about 1.3 mg/L 2–5 h after a 400 mg oral dose. The
half-life is about 8 h and the major route of excretion is
via the bile. Plasma protein binding of the sulfoxide is
around 70%. | | Clinical Use | Albendazole has a broad spectrum of activity against intestinal nematodes and cestodes, as well as the liver flukes Opisthorchis sinensis, Opisthorchis viverrini, and Clonorchis sinensis. It also has been used successfully against Giardia lamblia. It is widely used throughout the world for the treatment of intestinalnematode infection. It is effective as a single-dose treatmentfor ascariasis, New and Old World hookworm infections,and trichuriasis. Multiple-dose therapy with albendazole caneradicate pinworm, threadworm, capillariasis, clonorchiasis,and hydatid disease. The effectiveness of albendazole againsttapeworms (cestodes) is generally more variable and lessimpressive. It also is effective in treating cerebral and spinal neurocysticercosis, particularly when given with dexamethasone.Albendazole is recommended for treatment of gnathostomiasis. | | Side effects | Various mild intestinal and other upsets usually resolve
without treatment. With extended use, as for larval tapeworm
infections, hepatic abnormalities or leukopenia may
require discontinuation of treatment. In rare cases granulocytopenia,
pancytopenia, agranulocytosis or thrombocytopenia
may occur. It should not be given during pregnancy
since it may cause fetal harm; women should be cautioned
against becoming pregnant within a month of completing
treatment. | | Synthesis | Albendazole, methyl-[5-(propylthio)-1H-benzoimidazol-2-yl]carbamate
(38.1.18), is also made by the heterocyclization of a derivative of phenylenediamine to a
derivative of benzimidazole. In order to do that, 3-chloro-6-nitroacetanylide is reacted
with propylmercaptane to make 3-propylthio-6-nitroacetanylide (38.1.6). Reducing the
nitro group in this compound with hydrogen using a palladium on carbon catalyst
gives 4-(propylthio)-o-phenylenediamine (38.1.7). Reacting the resulting derivative of
o-phenylenediamine with cyanamide and then with the methyl chloroformate gives the
desired albendazole. 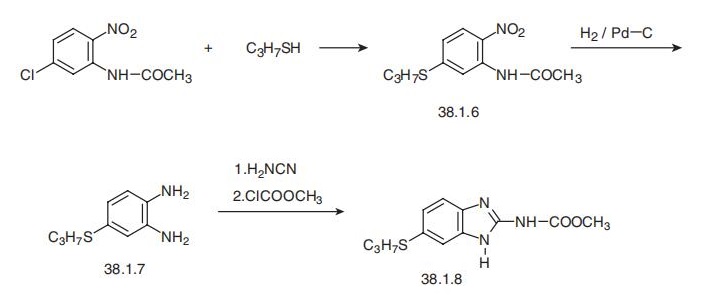
| | Veterinary Drugs and Treatments | Albendazole is labeled for the following endoparasites of cattle (not
lactating): Ostertagia ostertagi, Haemonchus spp., Trichostrongylus
spp., Nematodius spp., Cooperia spp., Bunostomum phlebotomum,
Oesphagostomum spp., Dictacaulus vivaparus (adult and 4th stage
larva), Fasciola hepatica (adults), and Moniezia spp.
In sheep, albendazole is approved for treating the following
endoparasites: Ostertagia circumcincta, Marshallagia marshalli,
Haemonchus contortus, Trichostrongylus spp., Nematodius spp.,
Cooperia spp., Oesphagostomum spp., Chibertia ovina, Dictacaulus
filaria, Fasciola hepatica, Fascioides magna, Moniezia expansa, and
Thysanosoma actinoides.
Albendazole is also used (extra-label) in small mammals, goats
and swine for endoparasite control.
In cats, albendazole has been used to treat Paragonimus kellicotti
infections. In dogs and cats, albendazole
has been used to treat
capillariasis. In dogs, albendazole has been used to treat Filaroides
infections. It has been used for treating giardia infections in small
animals, but concerns about bone marrow toxicity have diminished
enthusiasm for the drug’s use. |
| | Albendazole Preparation Products And Raw materials |
|