| Identification | More | [Name]
Sodium hydroxide | [CAS]
1310-73-2 | [Synonyms]
BETZ 0234
IODINE SOLUTION
KALII IODIDUM
LUGOL'S SOLUTION
POTASSIUM IODIDE
POTASSIUM IODIDE STANDARD
POTASSIUM IODIDE TS
Aetznatron
Augus hot rod
causticflake
causticsoda,bead
causticsoda,bead(dot)
causticsoda,dry(dot)
causticsoda,flake(dot)
causticsoda,granular(dot)
causticsoda,solid(dot)
causticsoda,solution
Collo-Grillrein
Collo-Tapetta
Fuers Rohr | [EINECS(EC#)]
231-659-4 | [Molecular Formula]
HNaO | [MDL Number]
MFCD00011405 | [Molecular Weight]
40 | [MOL File]
1310-73-2.mol |
| Chemical Properties | Back Directory | [Definition]
The most important commercial
caustic.
| [Appearance]
NaOH is a white, odorless, deliquescent material sold as pellets, flakes, lumps, or sticks. Aqueous solutions are known as soda lye | [Melting point ]
681 °C(lit.)
| [Boiling point ]
145 °C | [density ]
1.515 g/mL at 20 °C
| [vapor density ]
<1 (vs air)
| [vapor pressure ]
1 mm Hg ( 745 °C)
| [refractive index ]
1,473-1,475 | [Fp ]
176-178°C | [storage temp. ]
2-8°C
| [solubility ]
H2O: 1 M at 20 °C, clear, colorless
| [form ]
beads
| [color ]
White | [Specific Gravity]
2.13 | [Odor]
Odorless | [PH]
>14 (100g/l, H2O, 20℃) | [PH Range]
13 - 14 | [Water Solubility ]
SOLUBLE | [Decomposition ]
176-178 ºC | [Sensitive ]
Air Sensitive & Hygroscopic | [λmax]
λ: 260 nm Amax: 0.015
λ: 280 nm Amax: 0.01 | [Merck ]
14,8627 | [Dielectric constant]
57.5(25℃) | [Exposure limits]
TLV-TWA air 2 mg/m3 (OSHA); ceiling
2 mg/m3 (ACGIH) and 2 mg/m3/15 min
(NIOSH). | [Stability:]
hygroscopic | [Cosmetics Ingredients Functions]
DENATURANT
BUFFERING | [InChI]
1S/Na.H2O/h;1H2/q+1;/p-1 | [InChIKey]
HEMHJVSKTPXQMS-UHFFFAOYSA-M | [SMILES]
[OH-].[Na+] | [Uses]
Sodium Hydroxide is an alkali that is soluble in water, having a
solubility of 1 g in 1 ml of water. it is used to destroy the bitter
chemicals in olives that are to become black olives. it also functions
to neutralize acids in various food products. | [CAS DataBase Reference]
1310-73-2(CAS DataBase Reference) | [NIST Chemistry Reference]
Sodium hydroxide(1310-73-2) | [EPA Substance Registry System]
1310-73-2(EPA Substance) |
| Safety Data | Back Directory | [Symbol(GHS) ]

GHS05 | [Signal word ]
Danger | [Hazard statements ]
H290-H314 | [Precautionary statements ]
P234-P260-P280-P303+P361+P353-P304+P340+P310-P305+P351+P338 | [Hazard Codes ]
C,Xi | [Risk Statements ]
R36/38:Irritating to eyes and skin .
R35:Causes severe burns.
R34:Causes burns. | [Safety Statements ]
S26:In case of contact with eyes, rinse immediately with plenty of water and seek medical advice .
S45:In case of accident or if you feel unwell, seek medical advice immediately (show label where possible) .
S37/39:Wear suitable gloves and eye/face protection .
S24/25:Avoid contact with skin and eyes .
S36/37/39:Wear suitable protective clothing, gloves and eye/face protection . | [OEL]
Ceiling: 2 mg/m3 | [RIDADR ]
UN 1824 8/PG 2 | [WGK Germany ]
1 | [RTECS ]
TT2975000 | [F ]
8 | [TSCA ]
Yes | [REACH Registrations]
Active | [HazardClass ]
8 | [PackingGroup ]
II | [HS Code ]
2815 11 00 | [Storage Class]
8B - Non-combustible corrosive hazardous materials | [Hazard Classifications]
Eye Dam. 1
Met. Corr. 1
Skin Corr. 1A | [Safety Profile]
Poison by intraperitoneal route. Moderately toxic by ingestion. Mutation data reported. A corrosive irritant to skin, eyes, and mucous membranes. When heated to decomposition it emits toxic fumes of NanO. | [Hazardous Substances Data]
1310-73-2(Hazardous Substances Data) | [Toxicity]
LD orally in rabbits: 500 mg/kg (10% soln) (Fazekas) | [IDLA]
10 mg/m3 |
| Raw materials And Preparation Products | Back Directory | [Raw materials]
Sodium carbonate-->Sodium chloride-->Calcium chloride-->Calcium oxide-->CALCIUM CARBONATE-->Barium chloride dihydrate-->Barium chloride-->Boron tribromide-->Calcium chloride hexahydrate-->Sodium polyacrylate-->Caustic Soda(liquid)30%-48% | [Preparation Products]
6-AMINO-5-BROMOPYRIMIDIN-2(1H)-ONE-->DL-4-HYDROXY-3-METHOXYMANDELIC ACID-->BENZYL 1-PIPERAZINECARBOXYLATE-->1-[(Benzyloxy)carbonyl]piperidine-4-carboxylic acid-->3-(Acetylamino)thiophene-2-carboxylic acid-->2-(4,6-diamino-1,3,5-triazin-2-yl)acetic acid-->4-METHYL-2-PHENYL-1,3-THIAZOLE-5-CARBOXYLIC ACID-->SAFFLOWER YELLOW-->STRONTIUM HYDROXIDE OCTAHYDRATE-->Sodium isoamylxanthate-->C^{12~18^} fatty alcohol polyoxyethylene (35) ether-->dodecyl phenyl polyoxyethylene (12) ether-->additive AC1210-->Sodium pyroantimonate-->Sodium O-isobutyl dithiocarbonate-->Tricobalt tetraoxide-->Sodium lactate-->SODIUM PHOSPHOTUNGSTATE-->QUINUCLIDINE HYDROCHLORIDE-->2,3-DIPHENYLPROPIONIC ACID-->2-HYDROXY-1-NAPHTHOIC ACID-->3-Fluoro-4-hydroxybenzaldehyde-->Disodium tartrate dihydrate-->11-Oxahexadecan-16-olide-->emulsifier C^{8~10^} OPE-10-->calcium disodium bis[2-chloro-5-[(2-hydroxy-1-naphthyl)azo]-4-sulphonatobenzoate]-->C^{8~9^} alkyl phenyl polyoxyethylene (18) ether-->Peregal O-25-->SODIUM STANNATE TRIHYDRATE-->2-THIOPHENECARBOXYLIC ACID SODIUM SALT-->5,5-DIETHYLBARBITURIC ACID SODIUM SALT-->emulsifier SOPE-20-->dibenzyl biphenyl polyoxyethylene ether-->Soda lime-->2', 3'-ribonucleotide-->2-(1-Naphthalenyloxy)propanoic acid-->castor oil poloxyethylene (30) ether-->2-AMINO-4,6-DIMETHOXY-1,3,5-TRIAZINE-->thiourea-formaldehyde resin-->Alkaline Treated Starch |
| Hazard Information | Back Directory | [General Description]
A white solid. Corrosive to metals and tissue. Used in chemical manufacturing, petroleum refining, cleaning compounds, drain cleaners. | [Reactivity Profile]
CAUSTIC SODA (Sodium hydroxide) is a strong base. Reacts rapidly and exothermically with acids, both organic and inorganic. Readily absorbs moisture from the air to give caustic semi-solids that attack aluminum and zinc with the evolution of flammable hydrogen gas. Catalyzes the polymerization of acetaldehyde and other polymerizable compounds; these reactions can occur violently, for example, acrolein polymerizes with extreme violence when put in contact with alkaline materials such as sodium hydroxide [Chem. Safety Data Sheet SD-85 1961]. Reacts with great violence with phosphorus pentaoxide when initiated by local heating [Mellor 8 Supp.3:406 1971]. Contact (as a drying agent) with tetrahydrofuran, which often contains peroxides, may be hazardous---explosions have occurred in such a use of the chemically similar potassium hydroxide [NSC Newsletter Chem. Soc. 1967]. Mixing with any of the following substances in a closed container caused the temperature and pressure to increase: glacial acetic acid, acetic anhydride, acrolein, chlorohydrin, chlorosulfonic acid, ethylene cyanohydrin, glyoxal, hydrochloric acid (36%), hydrofluoric acid (48.7%), nitric acid (70%), oleum, propiolactone, sulfuric acid (96%) [NFPA 1991]. Accidental contact between a caustic cleaning solution (probably containing sodium hydroxide) and Pentol caused a violent explosion. [MCA Case History 363(1964)]. Heating with a mixture of methyl alcohol and trichlorobenzene during an attempted synthesis led to a sudden increase in pressure and an explosion [MCA Guide for Safety Appendix 3 1972]. Hot and/or concentrated NaOH can cause hydroquinone to decompose exothermically at elevated temperature. (NFPA Pub. 491M, 1975, 385) | [Air & Water Reactions]
Soluble in water. Dissolution can liberate enough heat to cause steaming and spattering and ignite adjacent combustible material [Haz. Chem. Data 1966]. | [Hazard]
Corrosive to tissue in presence of mois-
ture, strong irritant to tissue (eyes, skin, mucous
membranes, and upper respiratory tract), poison by
ingestion.
| [Health Hazard]
Strong corrosive action on contacted tissues. INHALATION: dust may cause damage to upper respiratory tract and lung itself, producing from mild nose irritation to pneumonitis. INGESTION: severe damage to mucous membranes; severe scar formation or perforation may occur. EYE CONTACT: produces severe damage. | [Potential Exposure]
NaOH is utilized to neutralize acids and make sodium salts in petroleum refining, viscose rayon; cellophane, plastic production; and in the reclamation of solutions of their salts. It is used in the manufacture of mercerized cotton, paper, explosives, and dyestuffs in metal cleaning; electrolytic extraction of zinc; tin plating; oxide coating; laundering, bleaching, dishwashing; and it is used in the chemical industries. | [Fire Hazard]
Non-combustible, substance itself does not burn but may decompose upon heating to produce corrosive and/or toxic fumes. Some are oxidizers and may ignite combustibles (wood, paper, oil, clothing, etc.). Contact with metals may evolve flammable hydrogen gas. Containers may explode when heated. | [Shipping]
UN1823 NaOH, solid, Hazard class: 8; Labels: 8-Corrosive material. UN1824 NaOH, solution, Hazard class: 8; Labels: 8-Corrosive material | [Incompatibilities]
A strong base and a strong oxidizer. Violent reaction with acid. Incompatible with water; flammable liquids; organic halogens, nitromethane, and nitrocompounds, combustibles. Rapidly absorbs carbon dioxide and water from air. Contact with moisture or water may generate heat. Corrosive to metals. Contact with zinc, aluminum, tin and lead in the presence of moisture, forming explosive hydrogen gas. Attacks some forms of plastics, rubber or coatings. | [Waste Disposal]
Discharge into tank containing water, neutralize, then flush to sewer with water. | [Electrical Conductivity ]
| Concentration |
0.50% |
1% |
2% |
5% |
10% |
15% |
20% |
25% |
30% |
40% |
50% |
| mS/cm |
24.8 |
48.6 |
93.1 |
206 |
– |
– |
– |
– |
– |
– |
– |
| [Physical properties]
White orthorhombic crystals, produced in the form of pellets, lumps, sticks, beads, chips, flakes or solutions; hygroscopic; very corrosive; rapidly absorbs CO2 and water from the air; density 2.13 g/cm3; melts at 323°C; vaporizes at 1388°C; vapor pressure 1 torr at 739°C and 5 torr at 843°C; very soluble in water (110 g/100mL at room temperature), generating heat on dissolution; aqueous solutions highly alkaline, pH of 0.5% solution about 13 and 0.05% solution about 12; soluble in methanol, ethanol and glycerol (23.8 g/100 mL methanol and 13.9 g/100 mL ethanol at ambient temperatures.). | [Production Methods]
Sodium hydroxide is manufactured by electrolysis of brine using
inert electrodes. Chlorine is evolved as a gas at the anode and
hydrogen is evolved as a gas at the cathode. The removal of chloride
and hydrogen ions leaves sodium and hydroxide ions in solution.
The solution is dried to produce the solid sodium hydroxide.
A second method uses the Kellner–Solvay cell. Saturated sodium
chloride solution is electrolyzed between a carbon anode and a
flowing mercury cathode. In this case the sodium is produced at the
cathode rather than the hydrogen because of the readiness of
sodium to dissolve in the mercury. The sodium–mercury amalgam is
then exposed to water and a sodium hydroxide solution is
produced. | [Reactions]
Sodium hydroxide is strongly alkaline and can react with acids to form salts and water.
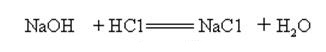
Sodium hydroxide reacts with acidic oxides to form salt and water, so sodium hydroxide can be used to absorb acid gases in the laboratory or industrially.
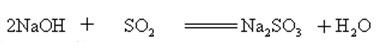
Sodium hydroxide can react with aqueous solutions of many metal salts to form sodium salts and metal hydroxides
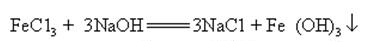
When sodium hydroxide and ammonia salt are heated together, it can release ammonia
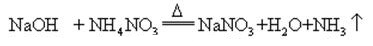
Sodium hydroxide is highly corrosive, so that the glass bottles storing sodium hydroxide solutions must be rubber stoppers, and glass stoppers should not be used to prevent a chemical reaction from opening. Sodium hydroxide is an important industrial raw material, and can be produced by electrolysis of saline solution industrially
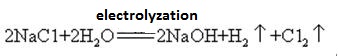
| [Flammability and Explosibility]
Sodium hydroxide and potassium hydroxide are not flammable as solids or aqueous
solutions. | [reaction suitability]
reaction type: Acid-base reactions | [Pharmaceutical Applications]
Sodium hydroxide is widely used in pharmaceutical formulations to
adjust the pH of solutions. It can also be used to react with weak
acids to form salts. | [Industrial uses]
Caustic soda (NaOH) is regarded as the strongest alkaline pH regulator. Caustic soda
is a very active substance and is highly corrosive. The bulk of caustic soda is manufactured
by electrolysis of saturated brines (NaCl). Caustic soda has a very strong pHregulating
capability (i.e. from pH 7 to pH 14) at a relatively low dosage compared to
other alkaline substances. Commercially, caustic soda is available in anhydrous form,
but in most mining applications the caustic soda is supplied as a 50% solution.
In the mineral processing industry, sodium hydroxide is mostly used for alkalinity control
during the processing of non-metallic minerals. In base metal flotation, the use of
sodium hydroxide is rare. | [Safety]
Sodium hydroxide is widely used in the pharmaceutical and food
industries and is generally regarded as a nontoxic material at low
concentrations. At high concentrations it is a corrosive irritant to
the skin, eyes, and mucous membranes.
LD50 (mouse, IP): 0.04 g/kg
LD50 (rabbit, oral): 0.5 g/kg | [storage]
Sodium hydroxide should be stored in an airtight nonmetallic
container in a cool, dry place. When exposed to air, sodium
hydroxide rapidly absorbs moisture and liquefies, but subsequently
becomes solid again owing to absorption of carbon dioxide and
formation of sodium carbonate. | [storage]
splash
goggles and impermeable gloves should be worn at all times when handling these
substances to prevent eye and skin contact. Operations with metal hydroxide
solutions that have the potential to create aerosols should be conducted in a fume
hood to prevent exposure by inhalation. NaOH and KOH generate considerable heat
when dissolved in water; when mixing with water, always add caustics slowly to the
water and stir continuously. Never add water in limited quantities to solid
hydroxides. Containers of hydroxides should be stored in a cool, dry location,
separated from acids and incompatible substances. | [Purification Methods]
Common impurities are water and sodium carbonate. Sodium hydroxide can be purified by dissolving 100g in 1L of pure EtOH, filtering the solution under vacuum through a fine sintered-glass disc to remove insoluble carbonates and halides. (This and subsequent operations should be performed in a dry, CO2-free box.) The solution is concentrated under vacuum, using mild heating, to give a thick slurry of the mono-alcoholate which is transferred to a coarse sintered-glass disc and evacuated free of mother liquor. After washing the crystals several times with purified alcohol to remove traces of water, they are dried in a vacuum, with mild heating, for about 30hours to decompose the alcoholate, leaving a fine white crystalline powder [Kelly & Snyder J Am Chem Soc 73 4114 1951]. CAUSTIC. Sodium hydroxide solutions (caustic), 14.77. Carbonate ion can be removed by passage through an anion-exchange column (such as Amberlite IRA-400; OH--form). The column should be freshly prepared from the chloride form by slow prior passage of sodium hydroxide solution until the effluent gives no test for chloride ions. After use, the column can be regenerated by washing with dilute HCl, then water. Similarly, other metal ions are removed when a 1M (or more dilute) NaOH solution is passed through a column of Dowex ion-exchange A-1 resin in its Na+-form. Alternatively, carbonate contamination can be reduced by rinsing sticks of NaOH (analytical reagent quality) rapidly with H2O, then dissolving in distilled H2O, or by preparing a concentrated aqueous solution of NaOH and drawing off the clear supernatant liquid. (Insoluble Na2CO3 is left behind.) Carbonate contamination can be reduced by adding a slight excess of concentrated BaCl2 or Ba(OH)2 to a NaOH solution, shaking well and allowing the BaCO3 precipitate to settle. If the presence of Ba in the solution is unacceptable, an electrolytic purification can be used. For example, sodium amalgam is prepared by the electrolysis of 3L of 30% NaOH with 500mL of pure mercury for cathode, and a platinum anode, passing 15 Faradays at 4Amps, in a thick-walled polyethylene bottle. The bottle is then fitted with inlet and outlet tubes, the spent solution being flushed out by CO2-free N2. The amalgam is then washed thoroughly with a large volume of deionised water (with the electrolysis current switched on to minimize loss of Na). Finally, a clean steel rod is placed in contact in the solution with the amalgam (to facilitate hydrogen evolution), reaction being allowed to proceed until a suitable concentration is reached, before being transferred to a storage vessel and diluted as required [Marsh & Stokes Aust J Chem 17 740 1964]. | [Toxics Screening Level]
The Initial Threshold Screening Level (ITSL) for sodium hydroxide is 8 μg/m3 with a
1-hour averaging time. The previous ITSL established on 9/14/1992 was 20 μg/m3
(1-hour averaging time). | [Regulatory Status]
GRAS listed. Accepted for use as a food additive in Europe.
Included in the FDA Inactive Ingredients Database (dental
preparations; injections; inhalations; nasal, ophthalmic, oral, otic,
rectal, topical, and vaginal preparations). Included in nonparenteral
and parenteral medicines licensed in the UK. Included in the
Canadian List of Acceptable Non-medicinal Ingredients. |
| Questions And Answer | Back Directory | [Chemical Properties]
Sodium hydroxide is a white, odorless, nonvolatile alkaline material marketed in solid form as pellets, flakes, clumps, or sticks. Its solubility in water is 111% by weight and a vapor pressure of 0mmHg (NIOSH, 1994).
It can react with tricholoethylene (TCE) to form flammable dichloroacetylene and with metals to form hydrogen gas (OEHHA, 1993). Its reactivity with metals should be considered in regards to storage units and containers.

Sodium hydroxide is commonly available as an aqueous solution known as caustic soda, soda lye, or simple as lye. It has various uses, including neutralization of acid; the manufacture of paper, textiles, plastics, corrosives, dyestuffs, paint, paint remover, and soap; refining of petroleum; electroplating; metal cleaning; laundering; and dish washing. A burgeoning use has been in the illegal manufacture of methamphetamine. | [Uses]
Sodium hydroxide (NaOH) is one of the most useful industrial sodium compounds. It
is also known as lye or caustic soda and is one of the strongest base alkalis (high pH value)
on the household market. It is used as a drain and oven cleaner, and it saponifies fats in the
manufacture of soap. It must be used with care because it is also capable of producing serious
skin burns. |
| Questions and Answers (Q&A) | Back Directory | [description]
Sodium hydroxide, also known as lye and caustic soda, is a highly caustic substance that is used used in small amounts in cosmetics to establish and hold the pH of a product.Sodium Hydroxide is a extremely important compound in our lives because it has so many uses. It is a very common base used in the chemical industry and is used for many things, many of which occur in our daily lives. One of the most well known uses of Sodium Hydroxide is its use in unclogging drains. It comes in many different brands of drain cleaners, but one of the most common is Drano. It also comes in the form of lye soap which can be used to wash practically anything, from the dishes to your face.
sodium hydroxide structure
At room temperature, sodium hydroxide is a white crystalline odorless solid that absorbs moisture from the air. It is a manufactured substance. Sodium Hydroxide is an inorganic compound used to control the pH levels or serve as a buffering agent in cosmetics and personal care products. It was historically used in the formulation of soaps, but is currently seen in a variety of formulas, including bath products, cleansing products, fragrances, foot powders, hair dyes and colors, makeup, nail products, personal cleanliness products, shampoos, shaving products, depilatories, skin care products, and suntan products, as well as chemical hair straighteners and hair wave sets. It is also a popular ingredient in industrial solvents as a chemical base for soaps, oven cleaners, detergents and drain cleaners because of its ability to dissolve grease, oils, fats and protein based deposits, according to Wikipedia. Less frequently, Sodium Hydroxide is seen as an ingredient in toothpastes.
Sodium Hydroxide is FDA approved, and has received the GRAS (Generally Recognized as Safe) rating as a direct food additive. However, it is primarily used in the washing and chemical peeling of produce. It is approved for use in cosmetics and personal care products in varying concentrations: 5% by weight in nail cuticle solvents, 2% by weight in hair straighteners for general use, 4.5% by weight in hair straighteners for professional use, up to a pH 12.7 in depilatories, and up to pH 11 in other uses as a pH adjuster.
References
https://pubchem.ncbi.nlm.nih.gov/compound/sodium_hydroxide
http://sodiumhydroxide.weebly.com/uses.html | [Uses]
Sodium hydroxide is one of the most important industrial chemicals. In volume, it is in the top ten chemicals produced in the United States. It is used in manufacturing a large number of compounds including several sodium salts, in treating cellulose for producing rayon and cellophane, and in manufacturing soaps, detergents, pulp, and paper. Sodium hydroxide is a common neutralizing agent for acids in acid-base titrations and petroleum refining. Another major application is extracting metals from their ores where alkali fusion, such as fusion with caustic soda, often is applied to open the ores. Additionally, sodium hydroxide is used to precipitate metals as hydroxides. Other uses are in reclaiming rubber, dissolving casein in plastics production, refining vegetable oils, processing textiles, as an eluant in ion chromatography, etching and electroplating, and as a laboratory reagent. Sodium hydroxide also is used as a strong base in many organic synthesis and base-catalyzed reactions.
Industry
Applications
Benefit
Paper and egenerated fibers making
Pulping process
Help to separate lignin from cellulose fibers
Brown pulp bleaching
Supply strong alkaline environment
Chemical manufacture
Production of sodium salt
Source of sodium
Praparation of formic acid, oxalic acid,phenol and other chemicals
Adjust pH, raw materials
Biodiesel manufacture
Ransesterification of methanol and triglycerides
Anhydrous sodium hydroxide acts as a catalyst
Cleaning agent
Industrial cleaning agent
Heated with water to clean process equipment, storage tanks, etc.
Cleaning waste discharge pipes and drains
Water solution can dissolve grease, oils, fats and protein-based deposits
Cleaning stainless steel, glass bakeware and oven cleaners
Used with surfactants to stabilize dissolved substances and thus prevent redeposition
Production of parts washer detergents
Major components
Drain opener and water pipes cleaners
Dissolves greases and hydrolyzes the proteins to unblock clogged drains
Food
Washing or chemical peeling of fruits and vegetables
Major components of peeling agent
Olives
Soak olives in sodium hydroxide for softening
Pretzels and German lye rolls
Help to make them crisp
Other foods processe
Used as neutralization, peeling, bleaching, deodorization, detoxification, and/or detergent agent.
Dissolving amphoteric metals and compounds
Etching, removing anodizing, or converting a polished surface to a satin-like finish
Make the solution alkaline to dissolve amphoteric metals and compounds
The refining of alumina containing ores to produce alumina
Dissolve alumina, leaving impurities less soluble at high pH
Praparation of sodium zincate and sodium plumbate
Make the solution alkaline to dissolve zinc and lead
Petroleum
Refining petroleum products
Neutralize the residual acid in the refining process
Oil-field drilling mud
Make the mud to be alkalin to inhibit microbial survival
Tissue digestion
Farm animals processing
Used with water to breaks the chemical bonds that keep the flesh intact
The process of decomposing roadkill dumped in landfills
Turns the body into a liquid with coffee-like appearance
Hairdressing
Manufacturers of chemical relaxers
Help to straighten hair
Water treatment
Removing lead, copper and other toxic metals
Increase ph of water
Soap making
Cold process soap, saponification
Used as esterification and transesterification reagent
Textile and dyeing
Cotton cloth processing
Used as desizing, scouring and mercerizing agent
Chemical analysis
Determination of acid content
Neutralize acid to make indicator coloured
Others
Asorbing slight CO2 and water
Works by its hygroscopicity and alkalinity
| [Reaction]
Sodium hydroxide is strongly alkaline and can react with acids to form salts and water.
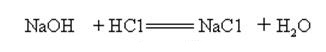 Sodium hydroxide reacts with acidic oxides to form salt and water, so sodium hydroxide can be used to absorb acid gases in the laboratory or industrially.
Sodium hydroxide reacts with acidic oxides to form salt and water, so sodium hydroxide can be used to absorb acid gases in the laboratory or industrially.
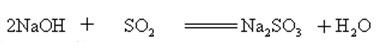 Sodium hydroxide can react with aqueous solutions of many metal salts to form sodium salts and metal hydroxides
Sodium hydroxide can react with aqueous solutions of many metal salts to form sodium salts and metal hydroxides
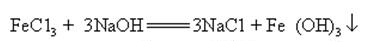 When sodium hydroxide and ammonia salt are heated together, it can release ammonia
When sodium hydroxide and ammonia salt are heated together, it can release ammonia
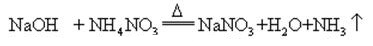 Sodium hydroxide is highly corrosive, so that the glass bottles storing sodium hydroxide solutions must be rubber stoppers, and glass stoppers should not be used to prevent a chemical reaction from opening. Sodium hydroxide is an important industrial raw material, and can be produced by electrolysis of saline solution industrially
Sodium hydroxide is highly corrosive, so that the glass bottles storing sodium hydroxide solutions must be rubber stoppers, and glass stoppers should not be used to prevent a chemical reaction from opening. Sodium hydroxide is an important industrial raw material, and can be produced by electrolysis of saline solution industrially
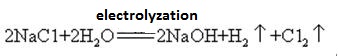
| [Preparation]
Sodium hydroxide is manufactured together with chlorine by electrolysis of sodium chloride solution. Various types of electrolytic cells are used commercially. They include the mercury cell, the diaphragm cell, and the membrane cell.
A saturated solution of brine is electrolyzed. Chlorine gas is liberated at the anode and sodium ion at the cathode. Decomposition of water produces hydrogen and hydroxide ions. The hydroxide ion combines with sodium ion forming NaOH. The overall electrolytic reactions may be represented as:
2Na+ + 2Cl_ + 2H2O → Cl2 (g) + H2 (g) + 2NaOH (aq)
The mercury cell proceeds in two stages that occur separately in two cells. The first is known as the brine cell or the primary electrolyzer in which sodium ion deposits on the mercury cathode forming amalgam, while chlorine gas is liberated at the anode:
Na+ + Cl–→ Na-Hg (cathode) + ½Cl2(g) (anode)
In the second cell, known as the decomposer cell, a graphite cathode is used while sodium amalgam serves as the anode. Water reacts with the sodium metal of the amalgam in the decomposer:
Na-Hg + H2O → Na+ + OH– + ½H2↑ + Hg
In chlor-alkali diaphragm cells, a diaphragm is employed to separate chlorine liberated at the anode from the sodium hydroxide and hydrogen generated at the cathode. Without a diaphragm, the sodium hydroxide formed will combine with chlorine to form sodium hypochlorite and chlorate. In many cells, asbestos diaphragms are used for such separation. Many types of diaphragm cells are available.
Sodium hydroxide is produced either as an anhydrous solid or as a 50% aqueous solution.
|
|
|