| Identification | More | [Name]
Xylocaine | [CAS]
137-58-6 | [Synonyms]
4,4'-DIMETHOXY DIPHENYL SULFOXIDE
LIDOCAINE-D6
2-(Diethylamino)-2',6'-acetoxylidide
2-(diethylamino)-2’,6’-acetoxylidide
2-(diethylamino)-n-(2,6-dimethylphenyl)-acetamid
2',6'-Acetoxylidide, 2-(diethylamino)-
6’-acetoxylidide,2-(diethylamino)-2
Acetamide, 2-(diethylamino)-N-(2,6-dimethylphenyl)-
Acetamide,2-(diethylamino)-N-(2,6-dimethylphenyl)-
a-Diethylamino-2,6-acetoxylidide
alfa-Dietilamino-2,6-dimetilacetanilide
alpha-(Diethylamino)-2,6-acetoxylidide
alpha-diethylamino-2,6-acetoxylidide
alpha-Diethylamino-2,6-dimethylacetanilide
alpha-Diethylaminoaceto-2,6-xylidide
Anbesol
Anestacon
Broncaine
Cappicaine
Cito optadren | [EINECS(EC#)]
205-302-8 | [Molecular Formula]
C14H14O3S | [MDL Number]
MFCD08443609 | [Molecular Weight]
262.32 | [MOL File]
137-58-6.mol |
| Chemical Properties | Back Directory | [Appearance]
solid | [Melting point ]
66-69°C | [Boiling point ]
bp4 180-182°; bp2 159-160° | [density ]
0.9944 (rough estimate) | [refractive index ]
1.5110 (estimate) | [Fp ]
9℃ | [storage temp. ]
Store at RT | [solubility ]
ethanol: 4 mg/mL
| [form ]
powder
| [pka]
pKa 7.88(H2O)(Approximate) | [color ]
White to slightly yellow | [Stability:]
Stable. Incompatible with strong oxidizing agents. | [Water Solubility ]
practically insoluble | [Merck ]
5482 | [BCS Class]
1 | [Major Application]
cleaning products
cosmetics
food and beverages
personal care
pharmaceutical (small molecule) | [Cosmetics Ingredients Functions]
NOT REPORTED | [InChI]
1S/C14H22N2O/c1-5-16(6-2)10-13(17)15-14-11(3)8-7-9-12(14)4/h7-9H,5-6,10H2,1-4H3,(H,15,17) | [Contact allergens]
Lidocaine is an anesthetic of the amide group, like articaine
or bupivacaine. Immediate-type IgE-dependent
reactions are rare, and delayed-type contact dermatitis
is exceptional. Cross-reactivity between the different
amide anesthetics is not systematic. | [InChIKey]
NNJVILVZKWQKPM-UHFFFAOYSA-N | [SMILES]
CCN(CC)CC(=O)Nc1c(C)cccc1C | [LogP]
2.440 | [CAS DataBase Reference]
137-58-6(CAS DataBase Reference) | [NIST Chemistry Reference]
Lidocaine(137-58-6) | [EPA Substance Registry System]
137-58-6(EPA Substance) |
| Hazard Information | Back Directory | [Chemical Properties]
solid | [Definition]
ChEBI: The monocarboxylic acid amide resulting from the formal condensation of N,N-diethylglycine with 2,6-dimethylaniline. | [Biological Activity]
Anasthetic and class Ib antiarrhythmic agent.? Blocks voltage-gated sodium channels in the inactivated state. | [Originator]
Xylocaine,Astra,US,1949 | [Indications]
Experimentally, lidocaine has been found to prevent VF arising during myocardial ischemia or infarction by preventing the fragmentation of organized largewavefronts into heterogeneous wavelets. Although lidocaine is of proven benefit in preventing VF early after clinical myocardial infarction, there is no evidence that it reduces mortality. To the contrary, lidocaine may increase mortality after myocardial infarction by approximately 40% to 60%.There are no controlled studies of lidocaine in secondary prevention of recurrence of VT or VF.
Lidocaine terminates organized monomorphic spontaneous VT or induced sustained VT in only approximately 20% of cases and is less effective than many other antiarrhythmic drugs. In a blinded, randomized study of intravenous lidocaine versus intravenous amiodarone in out-of-hospital VF resistant to defibrillation, lidocaine was associated with half the likelihood of survival to hospital admission compared with amiodarone. | [Manufacturing Process]
One mol of 2,6-xylidine is dissolved in 800 ml glacial acetic acid. The mixture
is cooled to 10°C, after which 1.1 mol chloracetyl chloride is added at one
time. The mixture is stirred vigorously during a few moments after which
1,000 ml half-saturated sodium acetate solution, or other buffering or
alkalizing substance, is added at one time. The reaction mixture is shaken
during half an hour. The precipitate formed which consists of ω-chloro-2,6-
dimethyl-acetanilide is filtered off, washed with water and dried. The product
is sufficiently pure for further treatment. The yield amounts to 70 to 80% of
the theoretical amount.
One mole of the chloracetyl xylidide thus prepared and 2.5 to 3 mols diethyl
amine are dissolved in 1,000 ml dry benzene. The mixture is refluxed for 4 to
5 hours. The separated diethyl amine hydrochloride is filtered off. The benzene
solution is shaken out two times with 3N hydrochloric acid, the first time with
800 ml and the second time with 400 ml acid. To the combined acid extracts
is added an approximately 30% solution of sodium hydroxide until the
precipitate does not increase.
The precipitate, which sometimes is an oil, is taken up in ether. The ether
solution is dried with anhydrous potassium carbonate after which the ether is
driven off. The remaining crude substance is purified by vacuum distillation.
During the distillation practically the entire quantity of the substance is carried
over within a temperature interval of 1° to 2°C. The yield approaches the
theoretical amount. MP 68° to 69°C. BP 180° to 182°C at 4 mm Hg; 159° to
160°C at 2 mm Hg. (Procedure is from US Patent 2,441,498.) | [Brand name]
Alphacaine (Carlisle); Lidoderm (Teikoku); Xylocaine
(AstraZeneca). | [Therapeutic Function]
Local anesthetic, Antiarrhythmic | [General Description]
Lidocaine was the first amino amide synthesized in 1948and has become the most widely used local anesthetic. Thetertiary amine has a pKa of 7.8 and it is formulated as thehydrochloride salt with a pH between 5.0 and 5.5. When lidocaineis formulated premixed with epinephrine the pH ofthe solution is adjusted to between 2.0 and 2.5 to prevent the hydrolysis of the epinephrine. Lidocaine is also availablewith or without preservatives. Some formulations of lidocainecontain a methylparaben preservative that maycause allergic reactions in PABA-sensitive individuals. Thelow pKa and medium water solubility provide intermediateduration of topical anesthesia of mucous membranes.Lidocaine can also be used for infiltration, peripheral nerveand plexus blockade, and epidural anesthesia. | [Biochem/physiol Actions]
Na+ channel blocker; class IB antiarrhythmic that is rapidly absorbed after parenteral administration. | [Pharmacokinetics]
Lidocaine is administered intravenously because extensive first-pass transformation by the liver prevents clinically effective plasma concentrations orally. The drug is dealkylated and eliminated almost entirely by the liver; therefore, dosage adjustments are necessary in the presence of hepatic disease or dysfunction. Lidocaine clearance exhibits the time dependency common to high-clearance agents. With a continuous infusion lasting more than 24 hours, there is a decrease in total lidocaine clearance and an increase in elimination half-life compared with a single dose. Lidocaine free plasma levels can vary in certain patients owing to binding with albumin and the acutephase reactant a1-acid glycoprotein. Levels of a1-acid glycoprotein are increased in patients after surgery or acute myocardial infarction, whereas levels of both a1-acid glycoprotein and serum albumin are decreased in chronic hepatic disease or heart failure and in those who are malnourished. This is an essential consideration because it is the unbound fraction that is pharmacologically active. | [Pharmacology]
Lidocaine is the most widely used local anaesthetic. It has a rapid onset and
short duration of action. Lidocaine is rapidly and extensively metabolised in
the liver and is safe at recommended doses. Efficacy is enhanced markedly
and duration of action prolonged by addition of adrenaline. Lidocaine is less
toxic than bupivacaine; a testament to this relative safety is that lidocaine is
used intravenously as a class 1b antiarrhythmic and as an i.v. infusion to treat
refractory chronic pain. Lidocaine solutions for injection are available in
concentrations of 1% and 2%, with or without adrenaline. It is also available
as a spray (4% or 10%), cream (2% or 4%), ointment or medicated plaster
(both 5%) for topical application. | [Clinical Use]
The metabolism of lidocaine is typical of the amino amideanesthetics . The liver is responsiblefor most of the metabolism of lidocaine and any decreasein liver function will decrease metabolism. Lidocaineis primarily metabolized by de-ethylation of the tertiary nitrogento form monoethylglycinexylidide (MEGX). At lowlidocaine concentrations, CYP1A2 is the enzyme responsiblefor most MEGX formation. At high lidocaine concentrations,both CYP1A2 and CYP3A4 are responsible for the formationof MEGX. | [Side effects]
Central nervous system side effects such as drowsiness, slurred speech, paresthesias, agitation, and confusion predominate. These symptoms may progress to convulsions and respiratory arrest with higher plasma concentrations. A rare adverse effect is malignant hyperthermia.
Cimetidine significantly reduces the systemic clearance of lidocaine as well as the volume of distribution at steady state and the degree of plasma protein binding. Beta blockers also reduce lidocaine clearance owing to a decrease in hepatic blood flow. For the same reason, clearance is reduced in congestive heart failure or low-output states.
Amiodarone may also influence the pharmacokinetics of lidocaine. In patients receiving amiodarone, single doses of intravenous lidocaine do not influence the pharmacokinetics of either agent. When amiodarone treatment is started in patients who are already receiving lidocaine infusion, there is a decrease in lidocaine clearance, which can result in toxic lidocaine levels. | [Synthesis]
Lidocaine, 2-(diethylamino)-N-(2,6-dimethylphenyl)acetamide (2.2.2), is
synthesized from 2,6-dimethylaniline upon reaction with chloroacetic acid chloride, which
gives |á-chloro-2,6-dimethylacetanilide (2.1.1), and its subsequent reaction with diethy�lamine [11].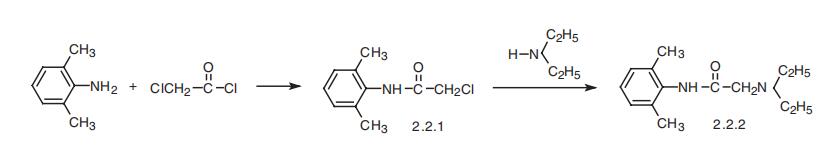
| [Veterinary Drugs and Treatments]
Besides its use as a local and topical anesthetic agent, lidocaine is
used to treat ventricular arrhythmias, principally ventricular tachycardia
and ventricular premature complexes in all species. Cats may
be more sensitive to the drug and some clinicians feel that it should
not be used in this species as an antiarrhythmic, but this remains
controversial. In horses, lidocaine may be useful to prevent postoperative
ileus and reperfusion injury. | [Electrophysiologic Effects]
Experimentally, lidocaine has been found to prevent VF arising during myocardial ischemia or infarction by preventing the fragmentation of organized largewavefronts into heterogeneous wavelets. Although lidocaine is of proven benefit in preventing VF early after clinical myocardial infarction, there is no evidence that it reduces mortality. To the contrary, lidocaine may increase mortality after myocardial infarction by approximately 40% to 60%.There are no controlled studies of lidocaine in secondary prevention of recurrence of VT or VF.
Lidocaine terminates organized monomorphic spontaneous VT or induced sustained VT in only approximately 20% of cases and is less effective than many other antiarrhythmic drugs. In a blinded, randomized study of intravenous lidocaine versus intravenous amiodarone in out-of-hospital VF resistant to defibrillation, lidocaine was associated with half the likelihood of survival to hospital admission compared with amiodarone. | [Drug interactions]
The concurrent administration of lidocaine with cimetidine
but not ranitidine may cause an increase (15%) in
the plasma concentration of lidocaine. This effect is a
manifestation of cimetidine reducing the clearance and
volume of distribution of lidocaine. The myocardial depressant
effect of lidocaine is enhanced by phenytoin
administration. | [Metabolism]
Lidocaine is extensively metabolized in the liver by N-dealkylation and aromatic hydroxylations
catalyzed by CYP1A2 isozymes. Lidocaine also possesses a weak inhibitory activity
toward the CYP1A2 isozymes and, therefore, may interfere with metabolism of other medications. | [storage]
Store at RT | [Toxicity evaluation]
The potency of lidocaine depends on various factors including
age of the subject, weight, physique including obesity, vascularity
of the site, and indication for use, as this would determine
the absorption and excretion rate. Physiologically,
lidocaine blocks neuronal transmission by interfering with the
flow of sodium across excitable membranes. A single lidocaine
molecule binds to a single voltage-gated sodium channel
impeding the movement of sodium ions across neuronal
membranes. Consequently repolarization is prevented and
further depolarization is not possible. Toxicity is dose related
and results from excessive quantities of lidocaine. | [Precautions]
Contraindications include hypersensitivity to local
anesthetics of the amide type (a very rare occurrence),
severe hepatic dysfunction, a history of grand mal
seizures due to lidocaine, and age 70 or older. Lidocaine
is contraindicated in the presence of second- or thirddegree
heart block, since it may increase the degree of
block and can abolish the idioventricular pacemaker responsible
for maintaining the cardiac rhythm. |
| Safety Data | Back Directory | [Symbol(GHS) ]

GHS07 | [Signal word ]
Warning | [Hazard statements ]
H302 | [Precautionary statements ]
P301+P312+P330 | [Hazard Codes ]
Xn | [Risk Statements ]
R22:Harmful if swallowed. | [Safety Statements ]
S22:Do not breathe dust .
S26:In case of contact with eyes, rinse immediately with plenty of water and seek medical advice .
S36:Wear suitable protective clothing . | [RIDADR ]
3249 | [WGK Germany ]
3
| [RTECS ]
AN7525000
| [TSCA ]
TSCA listed | [REACH Registrations]
Active | [HazardClass ]
6.1(b) | [PackingGroup ]
III | [HS Code ]
29242990 | [Storage Class]
11 - Combustible Solids | [Hazard Classifications]
Acute Tox. 4 Oral | [Safety Profile]
Poison by ingestion,
intravenous, intraperitoneal, and
subcutaneous routes. Human systemic
effects: blood pressure lowering, changes in
heart rate, coma, convulsions, dlstorted
perceptions, dyspnea, excitement,
hallucinations, muscle contraction or
spasticity, pulse rate, respiratory depression,
toxic psychosis. An experimental teratogen.
Other experimental reproductive effects. A
local anesthetic. Mutation data reported.
When heated to decomposition it emits
toxic fumes of NOx. | [Hazardous Substances Data]
137-58-6(Hazardous Substances Data) | [Toxicity]
LD50 oral in rat: 317mg/kg |
| Questions And Answer | Back Directory | [description]
Lidocaine is a local anesthetic, also known as Xylocaine, in recent years it has been replaced procaine, widely used in local infiltration anesthesia in cosmetic plastic surgery, it can block the nerve excitability and conduction by inhibiting the sodium channels of nerve cell membrane. The fat soluble and protein binding rate of lidocaine is higher than procaine, its cell penetrating ability is strong, fast onset, long duration of action, the interaction strength is 4 times of procaine.
Lidocaine is used in infiltration anesthesia, epidural anesthesia, topical anesthesia (including thoracoscopy or abdominal surgery for mucosal anesthesia) and nerve block. In order to extend the time of anesthesia, reduce the poisoning of lidocaine and other side effects, can be added in the anesthetic epinephrine.
Lidocaine can also be used for the treatment of ventricular premature beat after acute myocardial infarction, ventricular tachycardia, digitalis poisoning, cardiac surgery and cardiac catheterization-induced ventricular arrhythmias, including ventricular premature beats, ventricular tachycardia and ventricular fibrillation. Lidocaine is also used for duration status of epilepsy which other anti-seizure drugs are not effective, as well as local or spinal anesthesia. But it is usually ineffective for supraventricular arrhythmias. | [Chemical property]
Lidocaine is white needle like crystals, and its melting point is 68-69℃; boiling point is 180-182℃ (0.53kPa), soluble in ethanol in 159-160℃ (0.267kPa), ether, benzene, chloroform and oil, do not dissolve in water. In common use radical hydrochloride, lidocaine hydrochloride (C14H22N2O • HCL, [73-78-9]) is a white crystalline powder. Melting point 127-129℃, and the monohydrate melting point is 77-78℃. Easily soluble in water, 0.5% aqueous solution pHO 4.0-5.5. Odorless, bitter taste. | [Uses]
Lidocaine is an Anesthetic (local); antiarrhythmic (class IB). Long-acting, membrane stabilizing agent against ventricular arrhythmia. Originally developed as a local anesthetic. Neuroprotective & Neuroresearch Products. Lidocaine is widely used in surface anesthesia, anesthesia, conduction anesthesia and epidural anesthesia. The LD50 of oral lidocaine hydrochloride to mice was 290 mg/kg. |
|
|