1999-00-4
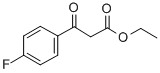 1999-00-4 结构式
1999-00-4 结构式
基本信息
3-(4'-氟苯基)-3-羰基-丙酸乙酯
(4-FLUOROBENZOYL)ACETIC ACID ETHYL ESTER
ETHYL 3-(4-FLUOROPHENYL)-3-OXOPROPANOATE
ETHYL 3-(4'-FLUOROPHENYL)-3-OXOPROPNOATE
ETHYL 4-FLUOROBENZOYLACETATE
ETHYL (P-FLUOROBENZOYL)ACETATE
3-(4'-Fluorophenyl)-3-oxopropanoate
3-Oxo-3-(4-fluorophenyl)propanoic acid ethyl ester
p-Fluorobenzoylacetic acid ethyl ester
β-Oxo-4-fluorobenzenepropionic acid ethyl ester
物理化学性质
安全数据
制备方法
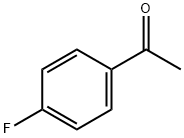
403-42-9
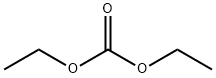
105-58-8

1999-00-4
(步骤1)在60℃条件下,向四氢呋喃(THF)悬浮液中依次加入碳酸二乙酯(47.4 mL,391 mmol)和氢化钠(55 wt%石蜡油分散体,23.7 g,543 mmol),随后缓慢加入4'-氟苯乙酮(26.4 mL,271 mmol)。将反应混合物加热回流1小时,冷却至室温后,缓慢倒入乙酸(33.6 mL,586 mmol)和冰水(1 L)的混合液中。用乙醚(2×500 mL)萃取反应混合物,有机层依次用饱和碳酸氢钠水溶液和饱和盐水洗涤,经无水硫酸钠干燥后,减压浓缩溶剂,得到黄色油状物3-(4-氟苯基)-3-氧代丙酸乙酯(45.7 g,收率100%,酮式:烯醇式=5:1)。产物通过1H-NMR(400 MHz,CDCl3)表征:酮式:δ 1.26(3H,t,J=7.2 Hz),3.97(3H,s),4.22(2H,q,J=7.2 Hz),7.13-7.19(2H,m),7.96-8.01(2H,m);烯醇式:δ 1.34(0.6H,t,J=7.2 Hz),4.27(0.4H,q,J=7.2 Hz),5.61(0.2H,s),7.08-7.12(0.4H,m),7.76-7.80(0.4H,m),12.62(0.2H,s)。
参考文献:
[1] Molecules, 2004, vol. 9, # 3, p. 135 - 157
[2] Patent: EP2738170, 2014, A1. Location in patent: Paragraph 0465
[3] Tetrahedron Letters, 2014, vol. 55, # 14, p. 2212 - 2216
[4] Patent: US2014/213571, 2014, A1. Location in patent: Paragraph 0780
[5] Patent: EP1362846, 2003, A1. Location in patent: Page/Page column 41
