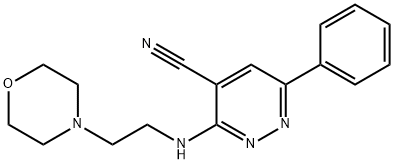
Bazinaprine
- CAS No.
- 94011-82-2
- Chemical Name:
- Bazinaprine
- Synonyms
- SR 95191;Bazinaprine;3-((2-Morpholinoethyl)aMino)-6-phenylpyridazine-4-carbonitrile;3-[(2-Morpholinoethyl)amino]-6-phenyl-4-pyridazinecarbonitrile;3-[[2-(Morpholin-4-y1)ethyl]amino]-6-phenyl-4-pyridazinecarbonitrile;4-Pyridazinecarbonitrile, 3-[[2-(4-morpholinyl)ethyl]amino]-6-phenyl-
- CBNumber:
- CB4969590
- Molecular Formula:
- C17H19N5O
- Molecular Weight:
- 309.37
- MDL Number:
- MOL File:
- 94011-82-2.mol
| Boiling point | 589.3±50.0 °C(Predicted) |
|---|---|
| Density | 1.26±0.1 g/cm3(Predicted) |
| storage temp. | 2-8°C |
| pka | 6.65±0.10(Predicted) |
| FDA UNII | NU8Y4C529J |
Bazinaprine price
| Manufacturer | Product number | Product description | CAS number | Packaging | Price | Updated | Buy |
|---|---|---|---|---|---|---|---|
| American Custom Chemicals Corporation | API0010774 | BAZINAPRINE 95.00% | 94011-82-2 | 5MG | $504.49 | 2021-12-16 | Buy |
| American Custom Chemicals Corporation | API0010774 | BAZINAPRINE 95.00% | 94011-82-2 | 1G | $1575 | 2021-12-16 | Buy |
| CSNpharm | CSN10383 | Bazinaprine | 94011-82-2 | 1g | $628 | 2021-12-16 | Buy |
| Chemenu | CM163612 | 3-((2-morpholinoethyl)amino)-6-phenylpyridazine-4-carbonitrile 95% | 94011-82-2 | 1g | $729 | 2021-12-16 | Buy |
| Crysdot | CD31000156 | Bazinaprine 95+% | 94011-82-2 | 1g | $772 | 2021-12-16 | Buy |
Bazinaprine Chemical Properties,Uses,Production
Originator
SR 95.191,Clin Midy/Sanofi
Uses
Bazinaprine can be used as a monoamine oxidase depressant in the preparation of antipsychotics.
Manufacturing Process
240.25 g of ethyl malonate, 138.0 g of potassium carbonate, 5.0 g of
potassium iodide and 154.0 g of phenacylchloride in 2 L of anhydrous acetone
are heated under reflux overnight. After the inorganic salts have been filtered
off, the filtrate is evaporated to dryness and the excess ethyl malonate is then
distilled off under reduced pressure (pressure: 0.5 mbar; temperature: about
60°C). The distillation residue is chromatographed on a silica column using a
cyclohexane/ethyl acetate mixture (9:1) as the eluent. The ethyl
phenacylmalonate is obtained in the form of a red oil. Yield: 80.3%.
40.5 g of the ethyl phenacylmalonate are dissolved in 70 ml of absolute
ethanol, and 7.25 g of hydrazine hydrate are added dropwise to the reaction
medium at 0°C, with stirring. When the reaction medium has returned to
room temperature, it is stirred for 24 h and the beige precipitate obtained,
which corresponds to the expected pyridazinone, is then filtered off. The
filtrate is treated with 3.62 g of hydrazine hydrate. After stirring for 24 h, an
additional quantity of pyridazinone can be filtered off. The same operation is
repeated once more on the filtrate. After purification by passage through a
silica column using a cyclohexane/ethyl acetate mixture (1:1) as the eluent,
the 4-ethoxycarbonyl-6-phenyl-4,5-dihydro-2H-pyridazin-3-one is obtained.
Yield: 37%.
9.0 g of the 4-ethoxycarbonyl-6-phenyl-4,5-dihydro-2H-pyridazin-3-one are
dissolved in 200 ml of acetic acid, and 11.18 g of bromine are then added to
the solution, with stirring. Decolouration of the medium occurs after 5 min.
After 2 h at room temperature, and with stirring, the medium is poured into
200 ml of water, the mixture is then extracted with methylene chloride and
the organic phase is evaporated to dryness. The residue is taken up 3 times
with cyclohexane. The beige powder obtained is chromatographed on a silica
column using a cyclohexane/ethyl acetate mixture (1:1) as the eluent. The 4-
ethoxycarbonyl-6-phenyl-2H-pyridazin-3-one, melting point 150°C is obtained.
Yield: 51%.
2.0 g of the 4-ethoxycarbonyl-6-phenyl-2H-pyridazin-3-one are added to 40
ml of concentrated ammonia solution and the mixture is stirred overnight at
room temperature. The solid is filtered off and dried to give the 6-phenyl-3-
oxo-2H-pyridazine-4-carboxamide, melting point >300°C. Yield: 86%.
1.5 g of the 6-phenyl-3-oxo-2H-pyridazine-4-carboxamide are dissolved in 20
ml of phosphorus oxychloride and the solution is then heated at 80°C for 5 h.
The mixture is poured into 50 ml of water. A precipitate appears, which is
filtered off and dried. There are obtained 58.3% of 3-chloro-4-cyano-6-
phenylpyridazine, melting point 206°C.
7.3 g of the 3-chloro-4-cyano-6-phenylpyridazine are dissolved in 60 ml of nbutanol,
and 8.0 g of N-(2-aminoethyl)-morpholine are added. The mixture is
heated under reflux for 3 h and then poured into 1000 ml of water. The
organic phase is extracted with ether and the ether solution is then extracted
with a 1 N solution of sulfuric acid. The aqueous phase is separated off,
rendered alkaline with sodium hydroxide and extracted with ether. The ether
phase is dried over magnesium sulfate and the solvent is then evaporated off
to dryness in vacuo to give Bazinaprine, as yellow solid, melting point 138°C. Yield: 81.3%.
Therapeutic Function
Antidepressant
Bazinaprine Preparation Products And Raw materials
Bazinaprine Suppliers
| Supplier | Tel | Country | ProdList | Advantage | |
|---|---|---|---|---|---|
| Alchem Pharmtech,Inc. | 8485655694 | sales@alchempharmtech.com | United States | 63711 | 58 |
| Amadis Chemical Company Limited | 571-89925085 | sales@amadischem.com | China | 131980 | 58 |
| Aikon International Limited | 025-66113011 18112977050 | cb6@aikonchem.com | China | 15495 | 58 |
| Changzhou Bojia Biomedical Technology Co., Ltd. | 2122619822 | czbjpharma@126.com | China | 18488 | 58 |
| TargetMol Chemicals Inc. | 4008200310 | marketing@tsbiochem.com | China | 24017 | 58 |
| Shanghai Haohong Scientific Co., Ltd. | 400-400-8210725 | malulu@leyan.com | China | 40041 | 58 |