Vincamine
- CAS No.
- 1617-90-9
- Chemical Name:
- Vincamine
- Synonyms
- Vinca;Vraap;Perval;Equipur;Minorin;Monorin;Novicet;Pervone;Oxybral;Angiopac
- CBNumber:
- CB7344925
- Molecular Formula:
- C21H26N2O3
- Molecular Weight:
- 354.44
- MDL Number:
- MFCD00078054
- MOL File:
- 1617-90-9.mol
- MSDS File:
- SDS
- TDS File:
- TDS
| Product description | Number | Pack Size | Price |
| Vincamine European Pharmacopoeia (EP) Reference Standard | Y0002124 | 10 mg | $164 |
| Vincamine European Pharmacopoeia (EP) Reference Standard | Y0002124 | Y0002124 | $150 |
| Vincamine analytical standard | 75778 | 50mg | $108 |
| Vincamine >98.0%(T) | V0061 | 1g | $71 |
| Vincamine >98.0%(T) | V0061 | 5g | $239 |
| More product size | |||
| Melting point | 232 °C (dec.)(lit.) |
|---|---|
| Boiling point | 487.66°C (rough estimate) |
| alpha | 42.8 º (c=1 in pyridine) |
| Density | 1.1640 (rough estimate) |
| refractive index | 1.6500 (estimate) |
| storage temp. | Keep in dark place,Inert atmosphere,2-8°C |
| solubility | Chloroform (Slightly), DMSO (Slightly) |
| form | Solid |
| pka | 12.13±0.40(Predicted) |
| color | White to Off-White |
| optical activity | [α]23/D +42.8°, c = 1 in pyridine |
| Merck | 14,9983 |
| Stability | Hygroscopic |
| Major Application | food and beverages |
| InChI | 1S/C21H26N2O3/c1-3-20-10-6-11-22-12-9-15-14-7-4-5-8-16(14)23(17(15)18(20)22)21(25,13-20)19(24)26-2/h4-5,7-8,18,25H,3,6,9-13H2,1-2H3/t18-,20+,21+/m1/s1 |
| InChIKey | RXPRRQLKFXBCSJ-GIVPXCGWSA-N |
| SMILES | CC[C@@]12CCCN3CCc4c(C13)n(c5ccccc45)[C@](O)(C2)C(=O)OC |
| LogP | 3.100 (est) |
| EWG's Food Scores | 1 |
| FDA UNII | 996XVD0JHT |
| ATC code | C04AX07 |
| NIST Chemistry Reference | Vincamine(1617-90-9) |
| UNSPSC Code | 12352200 |
| NACRES | NA.24 |
SAFETY
Risk and Safety Statements
| Symbol(GHS) |  GHS07 |
|||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Signal word | Warning | |||||||||
| Hazard statements | H302 | |||||||||
| Precautionary statements | P264-P270-P301+P312-P501 | |||||||||
| Hazard Codes | Xn | |||||||||
| Risk Statements | 22 | |||||||||
| Safety Statements | 36-26 | |||||||||
| WGK Germany | 3 | |||||||||
| RTECS | YY8575000 | |||||||||
| HS Code | 29399990 | |||||||||
| Storage Class | 11 - Combustible Solids | |||||||||
| Hazard Classifications | Acute Tox. 4 Oral | |||||||||
| Hazardous Substances Data | 1617-90-9(Hazardous Substances Data) | |||||||||
| Toxicity | LD50 in mice (mg/kg): 75 i.v.; >1000 s.c. (Szporny, Szász); 1000 orally (Szabo, Nagy) | |||||||||
| NFPA 704 |
|
Vincamine price More Price(48)
| Manufacturer | Product number | Product description | CAS number | Packaging | Price | Updated | Buy |
|---|---|---|---|---|---|---|---|
| Sigma-Aldrich | Y0002124 | Vincamine European Pharmacopoeia (EP) Reference Standard | 1617-90-9 | 10 mg | $164 | 2026-03-19 | Buy |
| Sigma-Aldrich | Y0002124 | Vincamine European Pharmacopoeia (EP) Reference Standard | 1617-90-9 | Y0002124 | $150 | 2024-03-01 | Buy |
| Sigma-Aldrich | 75778 | Vincamine analytical standard | 1617-90-9 | 50mg | $108 | 2022-05-15 | Buy |
| TCI Chemical | V0061 | Vincamine >98.0%(T) | 1617-90-9 | 1g | $71 | 2026-03-19 | Buy |
| TCI Chemical | V0061 | Vincamine >98.0%(T) | 1617-90-9 | 5g | $239 | 2026-03-19 | Buy |
Vincamine Chemical Properties,Uses,Production
Description
Vincamine is an alkaloid extracted from the leaves of the Vinca minor and is a related synthetic ethyl ester of vincaminic acid. It has spasmolytic effects similar to reserpine and can potentially improve blood flow in the brain.
Chemical Properties
white to almost white fine crystalline powder
Originator
Pervancamine ,Dausse,France,1969
Uses
Vincamine is often used as a nootropic agent to combat the effects of aging, or in conjunction with other nootropics (such as piracetam) for a variety of purposes. Vincamine is a peripheral vasodilator that increases blood flow to the brain.
Definition
ChEBI: Vincamine is a vinca alkaloid, an alkaloid ester, an organic heteropentacyclic compound, a methyl ester and a hemiaminal. It has a role as an antihypertensive agent, a vasodilator agent and a metabolite. It is functionally related to an eburnamenine.
Manufacturing Process
The following route is described in US Patent 4,145,552: At ambient temperature, over a period of thirty minutes, a solution of 33.8g (0.1mol) of (-)-vincadiformine in a mixture of 140 ml of anhydrous dimethylformamide and 140 ml of anhydrous toluene is added to a suspension of 2.64 g (0.11 mol) of sodium hydride in a mixture of 200 ml of anhydrous tetrahydrofuran, 20 ml of anhydrous hexamethylphosphotriamide (EMPT) and 18.7 ml (0.14 mol) of trimethyl phosphite. When the release of hydrogen has finished (about two hours later), the solution is cooled to -10°C and then stirred under an oxygen atmosphere until absorption ceases (duration: 3 hours). Still at -10°C, 136 ml of glacial acetic acid are added, and the mixture is then left at ambient temperature for two hours. After the addition of 500 ml of 1 N sulfuric acid, the aqueous phase is isolated, reextracted with 150 ml of isopropyl ether, made alkaline with 350 ml of 11 N ammonia, then extracted 3 times with 300 ml aliquots of methylene chloride. After drying over calcium chloride and evaporating the solvent, 30.2 g of crude product are obtained which, when chromatographed on a column of silica gel (1.5 kg) yield, 9.9 g of vincamine (yield: 28%) melting point (decomp.): 250°C.
brand name
Cerebroxine;Cetal;Ocu-vinc;Oxygeron;Pervincamine;Vadicate;Vinca minor;Vincacen;Vincapront;Vincavix;Vincimax.
Therapeutic Function
Vasodilator
World Health Organization (WHO)
Vincamine, an alkaloid derived from Vinca minor, is claimed to increase cerebral circulation and utilization of oxygen. It is used in a variety of cerebral disorders and is widely marketed for this purpose.
benefits
Vincamine is a naturally occurring indole alkaloid showing antioxidant activity and has been used clinically for the prevention and treatment of cerebrovascular disorders and insufficiencies. It has been well documented that antioxidants may contribute to cancer treatment, and thus, vincamine has been investigated recently for its potential antitumor activity. Vincamine was found to show cancer cell cytotoxicity and to modulate several important proteins involved in tumor growth, including acetylcholinesterase (AChE), mitogen-activated protein kinase (MAPK), nuclear factor-κB (NF-κB), nuclear factor erythroid 2-related factor 2 (Nrf2), and T-box 3 (TBX3).
General Description
Vincamine is a monoterpenoid indole alkaloid found in the leaves of Vinca minor L., belonging to the Apocynaceae family.
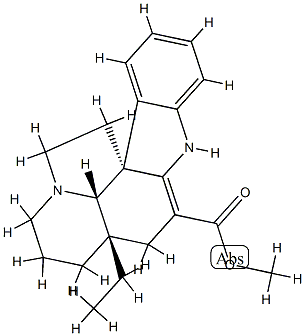
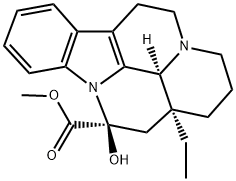
Vincamine Preparation Products And Raw materials
Raw materials
1of2
Preparation Products
| Supplier | Tel | Country | ProdList | Advantage | |
|---|---|---|---|---|---|
| Vinsce Bio pharm (Suzhou) Co Ltd | +undefined13962217456 | info@vinsce.com | China | 38 | 58 |
| Hebei Chuanghai Biotechnology Co., Ltd | +8615350571055 | Sibel@chuanghaibio.com | China | 8738 | 58 |
| Shaanxi Dideu Medichem Co. Ltd | +8617392709771 | 1097@dideu.com | China | 3996 | 58 |
| Hebei Chuanghai Biotechnology Co,.LTD | +86-86-13131129325 +8613131129325 | sales1@chuanghaibio.com | China | 5235 | 58 |
| Anhui Ruihan Technology Co., Ltd | +8617756083858 | daisy@anhuiruihan.com | China | 973 | 58 |
| Shaanxi Xianhe Biotech Co., Ltd | +86-17709210191; +8617709210191 | Jerry@xhobio.com | China | 906 | 58 |
| Hebei Zhuanglai Chemical Trading Co Ltd | +86-16264648883 | niki@zlchemi.com | China | 7245 | 58 |
| Capot Chemical Co.,Ltd. | +86-(0)57185586718; +8613336195806 | sales@capot.com | China | 29640 | 60 |
| Beijing Cooperate Pharmaceutical Co.,Ltd | 010-60279497 | sales01@cooperate-pharm.com | CHINA | 1803 | 55 |
| Henan Tianfu Chemical Co.,Ltd. | +86-0371-55170693 +86-19937530512 | info@tianfuchem.com | China | 21592 | 55 |
View Lastest Price from Vincamine manufacturers
| Image | Update time | Product | Price | Min. Order | Purity | Supply Ability | Manufacturer | |
|---|---|---|---|---|---|---|---|---|
 |
2026-04-24 | Vincamine
1617-90-9
|
US $0.00 / Kg/Bag | 1KG | 98.5%-101.5% | 100kg | WUHAN FORTUNA CHEMICAL CO., LTD | |
 |
2026-04-23 | Vincamine
1617-90-9
|
US $40.00 / mg | 99.52% | 10g | TargetMol Chemicals Inc. | ||
 |
2026-04-23 | Vincamine
1617-90-9
|
US $40.00 / mg | 99.52% | 10g | TargetMol Chemicals Inc. |
1617-90-9(Vincamine)Related Search:
1of4





