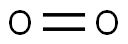Oxygen
- CAS No.
- 7782-44-7
- Chemical Name:
- Oxygen
- Synonyms
- O2;lox;Dioxygen;Sauerstoff;Pure oxygen;Liquid-oxygen-;Molecular oxygen;AAT10;OXYGEN;oxigeno
- CBNumber:
- CB5159244
- Molecular Formula:
- O2
Lewis structure
- Molecular Weight:
- 32
- MDL Number:
- MFCD00011434
- MOL File:
- 7782-44-7.mol
| Product description | Number | Pack Size | Price |
| Anti-LOX antibody produced in rabbit | SAB5700990 | 100μL | $448 |
| Anti-LOX antibody produced in rabbit Prestige Antibodies? Powered by Atlas Antibodies, affinity isolated antibody | HPA043930 | 100μL | $607 |
| Anti-LOX antibody produced in rabbit Prestige Antibodies? Powered by Atlas Antibodies, affinity isolated antibody | HPA043930 | 25μL | $282 |
| Anti-LOX antibody produced in rabbit | SAB5700990 | 100UL | $389 |
| Oxygen (99.6%) 99.6%, cylinder of 14?L, analytical standard | 300500-U | 1ea | $219 |
| More product size | |||
| Melting point | −218 °C(lit.) |
|---|---|
| Boiling point | −183 °C(lit.) |
| Density | 1.429(0℃) |
| vapor density | 1.11 (vs air) |
| vapor pressure | >760 mmHg at 20 °C |
| storage temp. | -20°C |
| solubility | At 20 °C and at a pressure of 101 kPa, 1 volume dissolves in about 32 volumes of water. |
| form | colorless gas |
| color | Colorless gas, liquid, or hexagonal crystals |
| Odor | Odorless gas |
| biological source | rabbit |
| Water Solubility | one vol gas dissolves in 32 volumes H2O (20°C), in 7 volumes alcohol (20°C); soluble other organic liq, usually higher solubility than in H2O [MER06] |
| Thermal Conductivity | 0.02658 W/(m·K) |
| Merck | 13,7033 |
| Henry's Law Constant | 1.3×10-5 mol/(m3Pa) at 25℃, Burkholder et al. (2019) |
| Dielectric constant | 1.5(-193℃) |
| Stability | Stable. Vigorously supports combustion. Incompatible with phosphorus, organic materials, many powdered metals. |
| Cosmetics Ingredients Functions | SKIN CONDITIONING |
| InChI | 1S/O2/c1-2/i1+0,2+0 |
| InChIKey | MYMOFIZGZYHOMD-ZCWHFVSRSA-N |
| SMILES | [16O]=[16O] |
| Surface tension | 15.85 mN/m at 80.0K |
| CAS DataBase Reference | 7782-44-7(CAS DataBase Reference) |
| Indirect Additives used in Food Contact Substances | OXYGEN |
| FDA 21 CFR | 177.2800 |
| EWG's Food Scores | 1 |
| FDA UNII | S88TT14065 |
| NCI Dictionary of Cancer Terms | hyperbaric oxygen; oxygen |
| NCI Drug Dictionary | hyperbaric oxygen |
| ATC code | V03AN01 |
| NIST Chemistry Reference | Oxygen(7782-44-7) |
| EPA Substance Registry System | Oxygen (7782-44-7) |
| UNSPSC Code | 12352203 |
| NACRES | NA.43 |
SAFETY
Risk and Safety Statements
| Symbol(GHS) |   GHS03,GHS04 |
|||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Signal word | Danger | |||||||||
| Hazard statements | H270-H280 | |||||||||
| Precautionary statements | P220-P244-P370+P376-P410+P403 | |||||||||
| PPE | Eyeshields, Gloves, multi-purpose combination respirator cartridge (US) | |||||||||
| Hazard Codes | O,C | |||||||||
| Risk Statements | 8-52/53-34-48/20/22-37 | |||||||||
| Safety Statements | 17-45-36/37/39-26-61 | |||||||||
| RIDADR | UN 1072 2.2 (OXYGEN, COMPRESSED) | |||||||||
| RIDADR | UN 1073 2.2 (OXYGEN, REFRIGERATED LIQUID) | |||||||||
| WGK Germany | - | |||||||||
| RTECS | RS2060000 | |||||||||
| F | 4.5-31 | |||||||||
| TSCA | TSCA listed | |||||||||
| HazardClass | 2.2 | |||||||||
| Storage Class | 2A - Gases | |||||||||
| Hazard Classifications | Ox. Gas 1 Press. Gas Compr. Gas |
|||||||||
| Hazardous Substances Data | 7782-44-7(Hazardous Substances Data) | |||||||||
| Toxicity | OSHA recommends a minimum oxygen concentration of 19.5% for human occupancy. | |||||||||
| NFPA 704 |
|
Oxygen price More Price(9)
| Manufacturer | Product number | Product description | CAS number | Packaging | Price | Updated | Buy |
|---|---|---|---|---|---|---|---|
| Sigma-Aldrich | SAB5700990 | Anti-LOX antibody produced in rabbit | 100μL | $448 | 2026-04-30 | Buy | |
| Sigma-Aldrich | HPA043930 | Anti-LOX antibody produced in rabbit Prestige Antibodies? Powered by Atlas Antibodies, affinity isolated antibody | 7782-44-7 | 100μL | $607 | 2026-04-30 | Buy |
| Sigma-Aldrich | HPA043930 | Anti-LOX antibody produced in rabbit Prestige Antibodies? Powered by Atlas Antibodies, affinity isolated antibody | 7782-44-7 | 25μL | $282 | 2023-06-20 | Buy |
| Sigma-Aldrich | SAB5700990 | Anti-LOX antibody produced in rabbit | 7782-44-7 | 100UL | $389 | 2023-06-20 | Buy |
| Sigma-Aldrich | 300500-U | Oxygen (99.6%) 99.6%, cylinder of 14?L, analytical standard | 7782-44-7 | 1ea | $219 | 2022-05-15 | Buy |
Oxygen Chemical Properties,Uses,Production
Discovery
Oxygen was discovered by Joseph Priestley in 1774 by the thermal decomposition of mercuric oxide and published his findings the same year, three years before Scheele published. In 1775–80, French chemist Antoine-Laurent Lavoisier, with remarkable insight, interpreted the role of oxygen in respiration as well as combustion, discarding the phlogiston theory, which had been accepted up to that time; he noted its tendency to form acids by combining with many different substances and accordingly named the element oxygen (oxygène) from the Greek words for“acid former.”
Description
Oxygen is a very prevalent and important element and is necessary for sustaining life on this planet. This element is the third most abundant in mass behind helium and hydrogen in the universe. The diatomic form (O2) is the most common pure form. With a boiling point at -183 °C, O2 exists as a colorless and odorless gas at standard temperature and pressure. In the process of cellular respiration, the highly reactive O2 is used as the oxidant in breaking down food molecules to produce energy. In turn, photosynthetic organisms generate O2 by using energy from the sun and water. Other allotropes of pure oxygen exist, including the trioxygen (O3) form known as ozone, as well as other, less common allotropes of oxygen such as O4 and O8. These oxygen allotropes are formed under high pressure and low temperatures and are solid.
Chemical Properties
Oxygen, O2, is a colorless, tasteless, gaseous element essential to almost all forms of life. It promotes respiration and combustion. Oxygen comprises 20% of the earth's atmosphere and is the most abundant element in seawater and in the earth's crust. It is slightly soluble in water and alcohol, but combines readily with most other elements to form oxides. The electrolysis of water produces both oxygen and hydrogen.
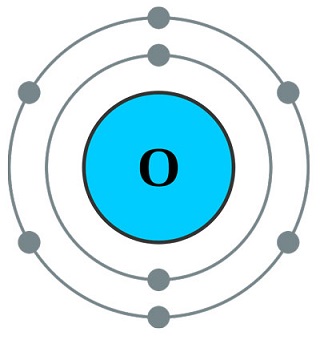
Physical properties
There are three allotropes (different forms) of oxygen: (1) atomic oxygen (O), sometimesreferred to as nascent or “newborn” oxygen; (2) diatomic oxygen (O2), or molecular oxygen(gas); and (3) ozone (O3), also a gas.
The atmospheric oxygen that we breathe is a very reactive nonmetal and is colorless, odorless,and tasteless, but it is essential to all living organisms. It readily forms compounds withmost other elements. With six electrons in its outer valence shell, it easily gains two moreelectrons to form a negative (–2) ion; or as covalent, it can share electrons with other elementsto complete its outer shell.
Almost all the oxygen in the atmosphere (21%) is the allotropic form of molecular oxygen(O2). This essential gas we breathe is the result of photosynthesis, which is how green plants(with chlorophyll) use the energy of the sun to convert carbon dioxide (CO2) and water tostarches and sugars with molecular oxygen as the by-product.
Liquid oxygen has a slightly bluish cast to it. As it boils, pure oxygen gas is released. Themelting point for oxygen is –218.79°C, its boiling point is –182.95°C, and its density is0.001429 g/cm3.
Isotopes
There are a total of 15 isotopes of oxygen, three of which are stable. The stableones are O-16, which accounts for 99.762% of all the oxygen on Earth; O-17, whichcontributes only 0.038% of the Earth’s oxygen; and O-18, which makes up just 0.200%of Earth’s oxygen.
Occurrence
Oxygen is the third most abundant element in the universe, making up nearly half themass of the Earth’s crust and nine-tenths of the total mass of water. Even the mass of ourbodies consists of two-thirds oxygen. Oxygen is also the most abundant element in the Earth’satmosphere at 20.947% by volume.
Oxygen is produced commercially by liquefying air under reduced temperatures andincreased pressure. Then oxygen (and other gases) can be collected as the temperature rises inthe liquid air, allowing the various gases to boil off at their specific boiling points. This processis known as fractional distillation. Liquid air can be transported in vacuum vessels in theliquid form as long as there is a small vent to allow the escape of some of the gas that boilsaway as temperatures rise above the boiling point.
Fractional distillation is based on the principle that each element has its own temperatureat which it changes from a liquid to a gas. Thus, any gas can be separated from other liquefiedcomponents of air and then collected. The same process is used in the petroleum industry toseparate various fractions from the crude oil.
There are several methods of producing oxygen gas in the laboratory.
Oxygen can be produced by electrolysis of water using a salt as an electrolyte that produceshydrogen at the opposite electrode. When potassium chlorate (KClO3) is heated in a testtube with a small amount of manganese dioxide (MnO2) as a catalyst, the chemical reactionthat releases the oxygen from potassium chlorate will be accelerated. Use of potassium nitrate(KNO3) will also produce small amounts of oxygen.
A recent, and more productive, method is to pass air through fine molecular-size sievesof material that will absorb the nitrogen gas of air, which then allows the oxygen gas to passthrough the sieve to be collected.
History
Oxygen, as a gaseous element, forms 21% of the atmosphere by volume from which it can be obtained by liquefaction and fractional distillation. The atmosphere of Mars contains about 0.15% oxygen. The element and its compounds make up 49.2%, by weight, of the Earth’s crust. About two thirds of the human body and nine tenths of water is oxygen. In the laboratory it can be prepared by the electrolysis of water or by heating potassium chlorate with manganese dioxide as a catalyst. The gas is colorless, odorless, and tasteless. The liquid and solid forms are a pale blue color and are strongly paramagnetic. Ozone (O3), a highly active compound, is formed by the action of an electrical discharge or ultraviolet light on oxygen. Ozone’s presence in the atmosphere (amounting to the equivalent of a layer 3 mm thick at ordinary pressures and temperatures) is of vital importance in preventing harmful ultraviolet rays of the sun from reaching the Earth’s surface. There has been recent concern that pollutants in the atmosphere may have a detrimental effect on this ozone layer. Ozone is toxic and exposure should not exceed 0.2 mg/m3 (8-hour time-weighted average — 40-hour work week). Undiluted ozone has a bluish color. Liquid ozone is bluish black, and solid ozone is violet- black. Oxygen is very reactive and capable of combining with most elements. It is a component of hundreds of thousands of organic compounds. It is essential for respiration of all plants and animals and for practically all combustion. In hospitals it is frequently used to aid respiration of patients. Its atomic weight was used as a standard of comparison for each of the other elements until 1961 when the International Union of Pure and Applied Chemistry adopted carbon 12 as the new basis. Oxygen has thirteen recognized isotopes. Natural oxygen is a mixture of three isotopes. Oxygen 18 occurs naturally, is stable, and is available commercially. Water (H2O with 1.5% 18O) is also available. Commercial oxygen consumption in the U.S. is estimated to be 20 million short tons per year and the demand is expected to increase substantially in the next few years. Oxygen enrichment of steel blast furnaces accounts for the greatest use of the gas. Large quantities are also used in making synthesis gas for ammonia and methanol, ethylene oxide, and for oxy-acetylene welding. Air separation plants produce about 99% of the gas, electrolysis plants about 1%. The gas costs 5¢/ft3 ($1.75/cu. meter) in small quantities.
Characteristics
Oxygen is, without a doubt, the most essential element on Earth. It is required to supportall plant and animal life, and it forms more compounds with other elements than any otherelement.
Oxygen is soluble in both water and alcohol. Contrary to what many people believe, oxygenis NOT combustible (it will not burn), but rather it actively supports the combustion ofmany other substances. After all, if oxygen burned, every time a fire was lit, all the O2 in theatmosphere would be consumed!
Burning is a form of oxidation wherein oxygen chemically combines with a substance rapidlyenough to produce adequate heat to cause fire and light, or to maintain a fire once started.The oxidation of iron is called rusting. Rusting in an example of “slow oxidation,” which isthe reaction of O2 with Fe to form Fe2O3 or Fe3O4. This chemical reaction is so slow that theheat it produces is dissipated; thus, there is no fire.
Recently a new allotrope of oxygen was discovered. When O2 is subjected to great pressure,it is converted intoO4, which is a deep red solid that is a much more powerful oxidizer thanthe other forms of oxygen.
Uses
Oxygen has many uses due to its high electronegativity with the ability to oxidize manyother substances. Only fluorine has higher electronegativity and is thus a stronger oxidizer.Besides the essential use to support life, oxygen has many other uses.
It is used in the smelting process to free metals from their ores. It is particularly importantin the oxygen-converter process in the production of steel from iron ore.
Oxygen is used in making several important synthetic gases and in the production ofammonia, methyl alcohol, and so on.
It is the oxidizer for liquid rocket fuels, and as a gas, oxygen is used in a mixture withhelium to support the breathing of astronauts and divers and to aid patients who have difficultybreathing. It is use to treat (oxidize) sewage and industrial organic wastes.
Oxygen has many uses because of its ability to accept electrons from other elements to formionic bonds or to share electrons with other elements to form covalent bonds.
Uses
The most obvious use of oxygen is to support life. The process of aerobic respiration uses O2 in an oxidation reaction that produces H2O and energy required for metabolic reactions. Uses of commercially produced O2 gas are mainly industrial and medical. O2 is a highly reactive element that reacts readily with most other chemicals. The process of combustion uses oxygen as the oxidizing element; for instance, the combustion of hydrocarbons in cars requires O2 in a chemical reaction to produce H2O, CO2, and energy as byproducts. Similarly, rocket fuel containing H2 gas uses liquid O2 in a combustion reaction to produce water vapor and heat. O2 is used in a wide variety of commercial industries. Most commercially produced O2 is used in the production of steel from iron ore. O2 is used in the production of other metals as well, such as zinc, copper, and lead. O2 is used in the gasification of coal to promote more complete combustion in incinerators; in the production of various chemicals such as ethylene oxide, propylene oxide, and nitric acid; as a fuel component in blow torches for the welding and cutting of metals. i.e., oxyacetylene torch; and in the papermaking industry to bleach pulp. O2 is used in wastewater treatment facilities and to reduce hydrogen sulfide in sewers. O2 has many therapeutic uses in medicine. Highpurity O2 gas is used in life support in surgeries, in intensive care units to assist breathing, for premature babies, and with inhalation therapies with chronic conditions. Hyperbaric oxygen chambers provide oxygen at high partial pressures for numerous conditions, including instances of CO poisoning or in treatment for decompression sickness, or ‘the bends,’ in divers.
Uses
In oxyhydrogen or oxyacetylene flame for welding metals and for lighting (calcium light, etc); submarine work by divers, propellant for rockets. In the production of synthesis gas which can be used in the Fischer-Tropsch process for liquid fuels.
Definition
Dioxygen: the normal form of molecularoxygen, O2, used to distinguishit from oxygen atoms or fromozone (O3).
Definition
oxygen: Symbol O. A colourlessodourless gaseous element belongingto group 16 (formerly VIB) of the periodictable; a.n. 8; r.a.m. 15.9994; d.1.429 g dm–3; m.p. –218.4°C; b.p.–183°C. It is the most abundant elementin the earth’s crust (49.2% byweight) and is present in the atmosphere(28% by volume). Atmosphericoxygen is of vital importance for allorganisms that carry out aerobic respiration.For industrial purposes it isobtained by fractional distillation of liquid air. It is used in metallurgicalprocesses, in high-temperatureflames (e.g. for welding), and inbreathing apparatus. The commonform is diatomic (dioxygen, O2);there is also a reactive allotropeozone (O3). Chemically, oxygen reactswith most other elements formingoxides. The element wasdiscovered by Joseph Priestley in1774.
Preparation
Most commercial oxygen at present is obtained from air by cryogenic separation processes. Although design of oxygen manufacturing plants and process conditions may vary depending on production capacity, purity desired, and cost, basic steps are similar.
Air first is filtered to remove dust particles. Water and carbon dioxide and most trace impurities are removed by silica gel (or other effective adsorbent) at a temperature slightly above 0°C. Acetylene and other hydrocarbons also can be removed by such adsorption processes. Alternatively, clean air is compressed and cooled to freeze out water and carbon dioxide, which can be trapped and removed in reversing exchangers. Compression and cooling of air is a critical step in its liquefaction. When cooled compressed air is allowed to expand it cools further (Joule-Thomson effect), converting the gaseous air to liquid air at about -196°C. Liquefied air is subjected to fractional distillation. More volatile argon and nitrogen distill out on warming, leaving behind oxygen with trace quantities of hydrogen, helium, and other inert gases.
Oxygen may be produced by electrolysis of water. In such electrolytic procedure, small amounts of H2SO4 or NaOH may be added to water. Electrolysis methods, however, are not used as much commercially as are air liquefaction processes which cost less. However, in making hydrogen from water by electrolysis, oxygen is obtained as a by-product.
In the laboratory oxygen may be prepared by several chemical methods that involve thermal decomposition of solid oxides or oxo salts. The most convenient method of preparing oxygen is to heat potassium chlorate in the presence of manganese dioxide catalyst:
Early preparation of oxygen involved thermal dissociation of metal oxides, notably mercury(II) oxide, which was used independently by both Priestley and Scheele. Also, oxides of lead, silver, and barium or potassium nitrate and permanganate were used by these and later investigators to prepare oxygen. Some reactions that yield oxygen by thermal decomposition of metal oxides and metal oxo salts, are highlighted below:
2HgO (s) → 2Hg (l) + O2 (g)
2BaO2 (s) → 2BaO (s) + O2 (g)
2PbO2 (s) → 2PbO (s) + O2 (g)
2KNO3 (s) → 2KNO2 (s) + O2 (g)
2Ag2O (s) → 4Ag (s) + O2 (g)
2KMnO4 (s) → K2MnO4 (s) + MnO2 (s) + O2 (g)
2K2S2O8 (s) → 2K2SO4 (s) + 2SO2 (g) + O2 (g)
Barium peroxide was used in commercial production of oxygen in the past. Heating barium oxide in air at 500°C forms barium peroxide, which decomposes at 800°C to yield oxygen:
Oxygen can be prepared chemically at ordinary temperatures. Several reactions in solution are known that may produce small quantities of oxygen at room temperatures. One such convenient method of producing oxygen is to slowly add water to sodium peroxide. The reaction is exothermic; therefore, the addition of water must be done cautiously.
2Na2O2 (s) + 2H2O (l) → 4NaOH (aq) + O2 (g)
Oxygen also is liberated when an acidified solution of potassium permangnate, acidified with sulfuric acid, is treated with a solution of hydrogen peroxide:
2MnO4ˉ (aq) + 5H2O2 (aq) + 6H+ (aq) → 2Mn2+ (aq) + 8H2O (l) + 5O2 (g).
Definition
The most abundant elementon earth, making up about 47% of the earth’s mass,and essential for respiration.
Production Methods
Oxygen is the most prevalent element in the Earth’s crust, making up 49.2% by weight. It accounts for 20.95% by volume of the Earth’s atmosphere and approximately 65% by weight of the human body.
General Description
Oxygen is a colorless, odorless and tasteless gas. Oxygen will support life. Oxygen is noncombustible, but will actively support the burning of combustible materials. Some materials that will not burn in air will burn in Oxygen. Materials that burn in air will burn more vigorously in Oxygen. As a non-liquid gas Oxygen is shipped at pressures of 2000 psig or above. Pure Oxygen is nonflammable. Under prolonged exposure to fire or intense heat the containers may rupture violently and rocket. Oxygen is used in the production of synthesis gas from coal, for resuscitation and as an inhalant.
Reactivity Profile
Propellant; ignites upon contact with alcohols, alkali metals, amines, ammonia, beryllium alkyls, boranes, dicyanogen, hydrazines, hydrocarbons, hydrogen, nitroalkanes, powdered metals, silanes, or thiols [Bretherick 1979. p.174]. Heat of water will vigorously vaporize liquid Oxygen, pressures may build to dangerous levels if this occurs in a closed container. Liquid Oxygen gives a detonable mixture when combined with powdered aluminum [NFPA 491M. 1991].
Hazard
Although oxygen itself is not flammable or explosive, as is sometimes believed, its mainhazard is that, in high concentrations, oxygen can cause other materials to burn much morerapidly.
Oxygen is toxic and deadly to breathe when in a pure state at elevated pressures. In addition,such pure oxygen promotes rapid combustion and can produce devastating fires, such asthe fire that killed the Apollo 1 crew on a test launch pad in 1967. It spread rapidly because thepure oxygen was at normal pressure rather than the one-third pressure used during flight.
Oxygen used for therapeutic purposes in adults can cause convulsions if the concentrationis too high. At one time, high levels of oxygen were given to premature infants to assist theirbreathing. It was soon discovered that a high concentration of O2 caused blindness in some ofthe infants. This practice has been abandoned, or the oxygen levels have since been reduced,and this is no longer a medical problem.
Oxygen involved in metabolic processes are prone to form “free radicals,” which arethought to cause damage to cells and possibly be associated with cancer and aging.
Health Hazard
Oxygen is nontoxic under the usual conditions of laboratory use. Breathing pure oxygen at one atmosphere may produce cough and chest pains within 8 to 24 h, and concentrations of 60% may produce these symptoms in several days. Liquid oxygen can cause severe "burns" and tissue damage on contact with the skin due to extreme cold.
Health Hazard
Inhalation of 100% Oxygen can cause nausea, dizziness, irritation of lungs, pulmonary edema, pneumonia, and collapse. Liquid may cause frostbite of eyes and skin.
Fire Hazard
Oxygen itself is nonflammable, but at concentrations greater than 25% supports and vigorously accelerates the combustion of flammable materials. Some materials (including metals) that are noncombustible in air will burn in the presence of oxygen.
Fire Hazard
Behavior in Fire: Increases intensity of any fire. Mixtures of liquid Oxygen and any fuel are highly explosive.
Flammability and Explosibility
Oxygen itself is nonflammable, but at concentrations greater than 25% supports and vigorously accelerates the combustion of flammable materials. Some materials (including metals) that are noncombustible in air will burn in the presence of oxygen.
Agricultural Uses
Oxygen (O) is an odorless, colorless, gaseous element
that belongs to group 16 (formerly group VI) of the
Periodic Table. It is the most abundant element
in the earth's crust (49.2% by weight), is present in the
atmosphere (20% by volume) and is a constituent of
water. It exists in three isotopes 16, 17 and 18. Oxygen is
essential for respiration of most living organisms and for
combustion. It is used in metallurgical processes, in high
temperature flames (welding) and in medical treatment.
The common form of oxygen is di-atomic oxygen
(O2) There is also another form - reactive allotrope
ozone (O3)C.h emically, oxygen reacts with most other
elements forming oxides. For industrial use, it is
obtained by fractional distillation of liquid air. This has
been replaced by a process which utilizes ambient
temperature separation by means of a pressure cycle in
which molecular sieves of synthetic zeolite preferentially
absorb nitrogen from air, giving 95 % oxygen and 5 %
argon.
The most popular industrial use of oxygen is in
oxygen enrichment of steel blast furnaces. Large
quantities of oxygen are used in the synthesis of nitric
acid from ammonia, methanol and ethylene oxide, as also
in oxy-acetylene welding.
Safety Profile
Human systemic effects by inhalation: cough and other pulmonary changes. Human teratogenic effects by inhalation: developmental abnormalities of the fetal cardovascular system. Mutation data reported. Not toxic as gas. In liquid form it can cause severe "burns" and tissue damage on contact with the slun due to extreme cold. An oxidant. Though itself nonflammable,it is essential to combustion. Even a slight increase in the oxygen content of the air above the normal 21% greatly increases the oxidation or burning rate (and the hazard) of many materials. Exclusion of O2 from the neighborhood of a fire is one of the principal methods of extinguishment. Avoid smoking, flames, electric sparks. Liquid O2 can explode on contact with readdy oxidizable materials, especially at high temperatures. Under the proper condltions of temperature, pressure, and reagent concentration it can react violently with acetaldehyde, acetylene, acetone, secondary alcohols (e.g., 2-propanol, 2-butanol) aluminum, Al(BH4)3, AH3, aluminumtitanium alloys, alkali metals @hum, cesium, potassium, rubidlum, sodlum, potassium), ammonia, ammonia + platinum, asphalt, ccl4, chlorinated hydrocarbons, cyanogen, barium, benzene, 1,4-benzenediol + 1-propanol, benzoic acid, Be(BH4)2, biological materials + ether, BAszBr3, B2H10, diH6, boron tribromide, boron trichloride, bromine + chlorotrifluoroethylene, butane + Ni(CO)4, carbon disulfide, carbon disulfide + mercury + anthracene, carbon monoxide, CsH, calcium, calcium hosphide, copper + hydrogen sulfide, Cl0H14, cyclohexane-l,2-done biskhenylhydrazone), cycl0℃tatetraene, dborane, diboron tetrafluoride, dimethoxymethane, dimethylketene, dimethyl sulfide, diphenyl ethylene, disilane, ethers (e.g., diethyl ether, diisopropyl ether, tetrahydro furan, dtoxane, ethyl ether), fibrous fabrics, fluorine + hydrogen, fuels, germanium, glycerol, halocarbons (e.g., l,l,l-trichloroethane, trichloroethylene, chlorotrifluoroethylene, bromotrifluoroethylene), hydrazine, hydrocarbons (e.g., 1,l-dphenylethylene, gasoline, cyclohexane, ethylene, cumene, pxylene, but-3-yne), hydrocarbons + promoters (e.g., methyl nitrate, nitromethane, ethyl nitrate, tetrafluorohydrazine), hydrogen, hydrogen sulfide, lithiated dalkylnitrosoamines, magnesium, metals, metal hydrides (e.g., sodtum hydride, uranium hydride, lithium h ydride, potassium hydride , rubidium hydride, cesium hydride, magnesium hydride), methane, methoxycycl0℃tatetraene, 4-methoxytoluene7 Ni(CO)4 + butane, nonmetal hydrides (e.g., diborane, tetraborane(lO), phosphine, pentaborane(1 l), pentaborane(9) , decaborane(l4), aluminum tetrahydroborate), oil films, organic matter, (OF2 + H20), phosphorus, phosphorus tribromide, phosphorus trifluoride, phosphorus(IⅡ) oxide, polymers [e.g., foam rubber, neoprene, polytetrafluoroethylene (teflon)], polytetrafluoroethylene + stainless steel, polyurethane, polyvinyl chloride, propylene oxide, K2O2, rhenium, trirhenium nonachloride, rubber + ozone, rubberized fabric, selenium, NaH, sodium hydroxide + tetramethyldsiloxane, strontium, tetracarbonylnickel, tetracarbonylnickel + mercury, tetrafluoroethylene, tetrafluorohydrazine, tetrasilane, titanium and alloys, trisilane, CH2Cl2, oil, paraformaldehyde, wood, charcoal. Compressed O2 is shipped in steel cylinders under hgh pressure. If these containers are broken due to shock or exposed to high temperature, an explosion and fire may result.
Potential Exposure
Compressed oxygen is used in various oxidation processes, for feedstock; and enrichment purposes; as a medicinal gas; a chemical intermediate; in oxyacetylene welding; in metallurgy. Liquid oxygen is used as a rocket fuel. Oxygen is naturally present at a concentration of 21% in breathing air.
First aid
Eye Contact: With liquid oxygen—immediatelyremove any contact lenses and flush with large amounts ofwater for at least 15 min, occasionally lifting upper andlower lids. Seek medical attention immediately. SkinContact: with liquid oxygen—if frostbite has occurred, seekmedical attention immediately; do NOT rub the affectedareas or flush them with water. In order to prevent furthertissue damage, do NOT attempt to remove frozen clothingfrom frostbitten areas. If frostbite has NOT occurred, immediately and thoroughly wash contaminated skin with soapand water. Seek medical attention. Breathing Pure Oxygenor Gases>40% O2; Remove the person from exposure.Begin rescue breathing (using universal precautions, including resuscitation mask) if breathing has stopped and CPR ifheart action has stopped. Transfer promptly to a medicalfacility. Medical observation is recommended for 24=48 hafter breathing overexposure, as pulmonary edema may bedelayed.
Carcinogenicity
Exposure to ionizing radiation is recognized as a cause of cancer, and the production of free radicals in the exposed cells is a part of the process. If hyperoxia causes free radical formation, it may contribute to the occurrence of cancer. Although a direct relation of hyperoxic injury and cancer has not been proven, numerous instances of association in experimental animals exist. Reactive oxidative intermediates have been shown to cause chromosome breaks and damage to DNA that can initiate carcinogenesis. Experimental work done with mouse skin tumors (as models of human tumors) has been both revealing and confusing. A substance may act as a cancer initiator or as a promoter, or sometimes both, depending on intensity and duration of exposure, and the presence of other carcinogenic materials. The same substance can also inhibit cancer growth. Hyperoxia has clearly been involved in modifying the course of tumor development, but the effects have differed under varying circumstances.
Environmental Fate
Atmospheric air contains 20.8% O2. Despite consumption of O2 through respiration and oxidative processes, this concentration remains constant, most likely due to the depleted O2 being replaced by plant-generated O2 in the photosynthetic process. O2 does not bioaccumulate in organisms as pure oxygen.
storage
Color Code—Yellow: Reactive Hazard; Store in alocation separate from other materials, especially flammables and combustibles. Prior to working with this chemical you should be trained on its proper handling andstorage. Liquid oxygen must be stored to avoid contact withorganic and combustible materials (such as oil, grease, andcoal dust) since violent reactions occur. Open storage is preferred. Oxygen gas evaporating from liquid or from oxygenenriched environments is easily absorbed into clothing andany source of ignition (such as a static spark) can causeflash burning. Compressed oxygen cylinders must besecurely stored separately from fuel cylinders. Liquid oxygen tanks should be stored outdoors. Sources of ignition,such as smoking and open flames, are prohibited whereoxygen is used, handled, or stored in a manner that couldcreate a potential fire or explosion hazard. See OSHAStandard 1910.104 and NFPA 43A Code for the Storage ofLiquid and Solid Oxidizers for detailed handling and storageregulations. Procedures for the handling, use, and storage ofcylinders should be in compliance with OSHA 1910.101and 1910.169, as with the recommendations of theCompressed Gas Association.
Shipping
UN1072 Oxygen, compressed & UN1073 Oxygen, refrigerated liquid (cryogenic liquid), Hazard Class: 2.2; Labels: 2.2-Nonflammable compressed gas; 5.1- Oxidizer. Cylinders must be transported in a secure upright position, in a well-ventilated truck. Protect cylinder and labels from physical damage. The owner of the compressed gas cylinder is the only entity allowed by federal law (49CFR) to transport and refill them. It is a violation of transportation regulations to refill compressed gas cylinders without the express written permission of the owner.
Purification Methods
Purify it by passing the gas over finely divided platinum at 673oK and Cu(II) oxide (see under nitrogen) at 973o, then condensed in a liquid N2-cooled trap. HIGHLY EXPLOSIVE in contact with organic matter.
Toxicity evaluation
The lack of sufficient O2 delivery to tissues of the body and thus insufficient supply of energy to support cellular processes is known as hypoxia. The partial pressure of O2 that exists in air is 160mmHg, and hypoxic effects will start to occur at partial pressures of 120 mmHg and below. Occupational Safety and Health Administration standards require 19.5% oxygen. The brain, with a high metabolic rate and no O2 reserve, is the most susceptible to hypoxia. Complex physiological changes occur in response to hypoxia, including an increase in heart and respiratory rate and artery constriction, all to try to ensure adequate tissue oxygenation. Depending on the rate and extent of hypoxia onset, different symptoms will be prevalent with loss of consciousness and cessation of respiration and heart activity in extreme states of hypoxia below about 10% O2. Hypoxia due to carbon monoxide poisoning is due to the higher relative affinity of hemoglobin in red blood cells to CO over O2, resulting in insufficient O2 delivery to metabolizing tissues, with deleterious effects on the heart and the central nervous system (CNS) being most problematic. The partial reduction of molecular oxygen in biological systems produces cytotoxic intermediates, including the superoxide, hydrogen peroxide, and hydroxyl radical. The superoxide radical especially plays a significant role in a number of pathophysiologic states associated with oxygen toxicity. Reactive oxygen species (ROS) cause lipid peroxidation in the cell membranes, leading to cellular damage and death. The ROS generated can also lead to the production of other free radicals like nitric oxide, peroxynitrite, and trioxidane, which can cause damage to DNA and other molecules. Oxygen radical scavengers such as superoxide dismutase and catalase protect the body against oxygen-free radicals produced in day-to-day metabolic reactions.
Incompatibilities
A strong oxidizer. Reacts violently with nearly every element, combustibles, organics, and reducing materials.
Waste Disposal
Return refillable compressed gas cylinders to supplier. Vent to atmosphere.
Oxygen Preparation Products And Raw materials
Raw materials
1of2
Preparation Products
1of8
| Supplier | Tel | Country | ProdList | Advantage | |
|---|---|---|---|---|---|
| Henan Tianfu Chemical Co.,Ltd. | +86-0371-55170693 +86-19937530512 | info@tianfuchem.com | China | 21588 | 55 |
| Dideu Industries Group Limited | +86-29-89586680 +86-15129568250 | 1026@dideu.com | China | 18895 | 58 |
| Hebei Fengjia New Material Co., Ltd | +86-0311-87836622 +86-18712993135 | sales01@tairunfaz.com | China | 8051 | 58 |
| PT CHEM GROUP LIMITED | +86-85511178; | peter68@ptchemgroup.com | China | 35425 | 58 |
| XIAMEN AMITY INDUSTRY AND TRADE CO., LTD. | +8618950047208 | ellena@amitychem.com | China | 43416 | 58 |
| Mainchem Co., Ltd. | +86-0592-6210733 | sale@mainchem.com | China | 32343 | 55 |
| Chemwill Asia Co.,Ltd. | 86-21-51086038 | chemwill_asia@126.com | CHINA | 23912 | 58 |
| Wuhan eastop Technology Co., ltd.6 | 18086015167 17702779303 | service@isotopechina.com | China | 310 | 58 |
| China Isotope Development Co.,Ltd | 17331008322 | 178069904@qq.com | China | 196 | 58 |
| JinYan Chemicals(ShangHai) Co.,Ltd. | 13817811078 | sales@jingyan-chemical.com | China | 9976 | 60 |
Related articles
- Oxygen:Sources,electronic configuration,uses,production
- Oxygen constitutes 49.2% of the Earth's crust by mass as part of oxide compounds such as silicon dioxide, and SiO2, and is the....
- May 27,2024
- what is the atomic number of the element located in group 16, period 2 of the periodic table?
- The atomic number of the element in Group 16, Period 2 of the Periodic Table is 8 and the element is oxygen.
- Mar 7,2024
- What is oxygen used for? Is it an element?
- Oxygen forms a molecule (O2) of two atoms which is a colorless gas at normal temperatures and pressures.Oxygen is the most abu....
- Mar 4,2024
View Lastest Price from Oxygen manufacturers
| Image | Update time | Product | Price | Min. Order | Purity | Supply Ability | Manufacturer | |
|---|---|---|---|---|---|---|---|---|
 |
2025-11-18 | Oxygen USP/EP/BP
7782-44-7
|
US $1.10 / g | 1g | 99.9% | 100 Tons min | Dideu Industries Group Limited |
-

- Oxygen USP/EP/BP
7782-44-7
- US $1.10 / g
- 99.9%
- Dideu Industries Group Limited