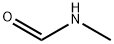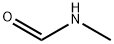N-Methylformamide
- CAS No.
- 123-39-7
- Chemical Name:
- N-Methylformamide
- Synonyms
- Methylformamide;N-Methylmethanamide;x188;X 188;ek7011;EK 7011;nsc3051;HCONHCH3;NSC 3051;Formylmethylamine
- CBNumber:
- CB9109065
- Molecular Formula:
- C2H5NO
- Molecular Weight:
- 59.07
- MDL Number:
- MFCD00003280
- MOL File:
- 123-39-7.mol
- MSDS File:
- SDS
| Melting point | -4 °C (lit.) |
|---|---|
| Boiling point | 198-199 °C (lit.) |
| Density | 1.011 g/mL at 25 °C (lit.) |
| vapor pressure | 20Pa at 20℃ |
| refractive index |
n |
| Flash point | >230 °F |
| storage temp. | Sealed in dry,Room Temperature |
| solubility | soluble in water, chloroform, alcohols, acetone, ethylacetate but not in diethyl ether and petroleum ether |
| pka | 16.48±0.23(Predicted) |
| form | Liquid |
| color | Clear colorless |
| Odor | strong odor |
| Relative polarity | 6.2 |
| Water Solubility | miscible |
| Merck | 14,6076 |
| BRN | 1098352 |
| Exposure limits | ACGIH: TWA 1 ppm (Skin) |
| Dielectric constant | 171.0 |
| InChIKey | ATHHXGZTWNVVOU-UHFFFAOYSA-N |
| LogP | -0.97 |
| CAS DataBase Reference | 123-39-7(CAS DataBase Reference) |
| EWG's Food Scores | 3 |
| FDA UNII | XPE4G7Y986 |
| NCI Drug Dictionary | N-methylformamide |
| NIST Chemistry Reference | Formamide, N-methyl-(123-39-7) |
| EPA Substance Registry System | N-Methylformamide (123-39-7) |
SAFETY
Risk and Safety Statements
| Symbol(GHS) |   GHS07,GHS08 |
|||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Signal word | Danger | |||||||||
| Hazard statements | H312-H360D | |||||||||
| Precautionary statements | P201-P280-P302+P352+P312-P308+P313 | |||||||||
| Hazard Codes | T | |||||||||
| Risk Statements | 61-21-R61-R21 | |||||||||
| Safety Statements | 53-45-S53-S45 | |||||||||
| WGK Germany | 1 | |||||||||
| RTECS | LQ3000000 | |||||||||
| TSCA | Yes | |||||||||
| PackingGroup | II | |||||||||
| HS Code | 29241990 | |||||||||
| Toxicity | LD50 in mice (mg/kg): 2300 i.p.; 2600 orally; 1580 i.v.; 2700 i.m. (Langdon) | |||||||||
| NFPA 704 |
|
N-Methylformamide price More Price(26)
| Manufacturer | Product number | Product description | CAS number | Packaging | Price | Updated | Buy |
|---|---|---|---|---|---|---|---|
| Sigma-Aldrich | 473936 | N-Methylformamide 99% | 123-39-7 | 1l | $129 | 2024-03-01 | Buy |
| Sigma-Aldrich | 473936 | N-Methylformamide 99% | 123-39-7 | 2.5l | $259 | 2024-03-01 | Buy |
| TCI Chemical | F0059 | N-Methylformamide >99.0%(GC) | 123-39-7 | 25g | $18 | 2024-03-01 | Buy |
| TCI Chemical | F0059 | N-Methylformamide >99.0%(GC) | 123-39-7 | 500g | $39 | 2024-03-01 | Buy |
| Alfa Aesar | L03908 | N-Methylformamide, 99% | 123-39-7 | 50g | $24.65 | 2024-03-01 | Buy |
N-Methylformamide Chemical Properties,Uses,Production
Chemical Properties
N-methylformamide is a clear colorless liquid with a slight amine odor. It is a water-soluble organic solvent. As an adjuvant antineoplastic agent, N-methylformamide depletes cellular glutathione, a key molecule involved in the antioxidation of reactive oxygen species (ROS) and other free radicals, thereby enhancing ionizing radiation-induced DNA cross-linking in and terminal differentiation of tumor cells. (NCI04)
Uses
N-Methylformamide is used in amidation or transamidation chemical reactions where formamide is insufficient.
Uses
N-Methylformamide is used to prepare methyl isocyanate. It is used as an extraction solvent for aromatic hydrocarbons and acts as a ligand in coordination chemistry. It is employed as a solvent in oil refineries and in aluminum electrolytic capacitors. Further, it is used as a reagent in organic synthesis. In addition to this, it is used as an investigational anticancer drug and an intermediate in the production of some pharmaceutical compounds.
Production Methods
N-Methylformamide (NMF) can be synthesized by: (1) reacting methylamine, carbon monoxide, methanol and a small amount of potassium acetate at 250 atm and 160°C; (2) heating methylamine with carbon monoxide and some sodium ethoxide in ethanol at 150 atm; (3) treating methylformate with methylamine and methanol; (4) reacting hexamethylenetetramine with formamide and hydrogen in the presence of Raney nickel at 130-145°C (Beilstein's Handbuch, 1977).
Definition
ChEBI: N-methylformamide is a member of the class of formamides having a N-methyl substituent. It is functionally related to a formamide.
General Description
N-Methylformamide (NMF), a formamide derivative, is widely employed as an organic solvent. NMF molecule contains C=O and N-H groups, so it can act as a proton donor and acceptor. Due to its important physical properties such as high dielectric constant, high solubility, low conductivity and amphiprotic nature, it is suitable for use as a separation medium in capillary electrophoresis studies.
NMF is closely related to other formamides, notably formamide and dimethylformamide (DMF). However, industrial use and production of NMF are far less than for either of these other formamides. DMF is favored over NMF as a solvent due to its greater stability. Annual production of NMF can be assumed to be significantly less than the production of either formamide (100,000 tons) or DMF (500,000 tons).
Air & Water Reactions
Water soluble.
Reactivity Profile
N-Methylformamide is incompatible with benzene sulfonyl chloride. N-Methylformamide is also incompatible with strong oxidizing agents, acids, bases and acid chlorides. N-Methylformamide may react with chlorine, bromine, nitrates, nitric acid, triethylaluminum, potassium permanganate, chromic acid, chromic anhydride, chromium trioxide, borohydrides, hydrides, thionyl chloride, metallic sodium, phosphorus trioxide, diborane, (octafluoroisobutyrate + sodium nitrite) and (perchloryl fluoride + potassium methyl 4,4-dinitrobutyrate).
Health Hazard
SYMPTOMS: Symptoms of exposure to this compound include irritation of the mucous membranes and upper respiratory tract. Other symptoms include liver damage, eye irritation with discomfort, tearing or blurring of vision, skin irritation with discomfort or rash, abnormalities of liver function with jaundice, temporary nervous system depression with anesthetic effects such as dizziness, headache, confusion, incoordination and loss of consciousness.
ACUTE/CHRONIC HAZARDS: N-Methylformamide may be absorbed through the skin and cause skin irritation. It may also irritate the eyes, mucous membranes and upper respiratory tract. When heated to decomposition it emits toxic fumes of carbon monoxide, carbon dioxide and nitrogen oxides. (NTP, 1992)
Fire Hazard
N-Methylformamide is combustible.
Industrial uses
NMF possesses excellent solvent properties that are similar to those of dimethylformamide. However, NMF appears to be much less important as an industrial solvent than dimethylformamide.
Safety Profile
Moderately toxic by ingestion, intraperitoneal, intravenous, intramuscular, and subcutaneous routes. An experimental teratogen. Experimental reproductive effects. An eye irritant. A very dangerous fire hazard when exposed to heat or flame. Violent reaction with benzene sulfonyl chloride. When heated to decomposition it emits toxic fumes of NOx.
Purification Methods
Dry it over molecular sieves for 2days, then distil it under reduced pressure through a column packed with glass helices. Fractionally crystallise it by partial freezing and the solid portion is distilled in a vacuum. [Beilstein 4 IV 170.]
Toxicity evaluation
N-Methylformamide (NMF) is a metabolite of dimethylformamide (DMF), a solvent with wide applications in the chemical industry.Pregnant rats and rabbits were dosed once daily by gavage on Gestation Days 6-15 and 6-18, respectively. Doses for rats were 0, 1, 5, 10, or 75 mg/kg; doses for rabbits were 0, 5, 10, or 50 mg/kg. No treatment-related maternal deaths or clinical signs occurred in either species. Body weight gain and food consumption were depressed in rats given 75 mg/kg and rabbits given 50 mg/kg. Fetal viability was reduced at both rabbits and rats. The lowest-observed-adverse-effect levels for maternal and developmental toxicity in the rat and rabbit were 75 and 50 mg/kg, respectively. he no-observed-adverse-effect level for maternal and developmental toxicity in the rat and rabbit was 10 mg/kg[1].
References
[1] Kelich S, et al. Developmental toxicity of N-methylformamide administered by gavage to CD rats and New Zealand white rabbits. Fundam Appl Toxicol, 1995.
N-Methylformamide Preparation Products And Raw materials
Raw materials
1of2
Preparation Products
1of2
| Supplier | Tel | Country | ProdList | Advantage | |
|---|---|---|---|---|---|
| Hefei TNJ Chemical Industry Co.,Ltd. | +86-0551-65418679 +86-18949832763 | info@tnjchem.com | China | 2989 | 55 |
| Hebei Guanlang Biotechnology Co., Ltd. | +86-19930503282 | alice@crovellbio.com | China | 8823 | 58 |
| Yancheng Green Chemicals Co.,Ltd | +undefined-86-25-86655873 | info@royal-chem.com | China | 114 | 58 |
| Hebei Guanlang Biotechnology Co,.LTD | +8619930503252 | daisy@crovellbio.com | China | 5964 | 58 |
| Hebei Miaoyin Technology Co.,Ltd | +86-17367732028 +86-17367732028 | kathy@hbyinsheng.com | China | 3582 | 58 |
| Hebei Jingbo New Material Technology Co., Ltd | +8619931165850 | hbjbtech@163.com | China | 1000 | 58 |
| Hebei Zhuanglai Chemical Trading Co.,Ltd | +8613343047651 | admin@zlchemi.com | China | 476 | 58 |
| Nanjing Deda New Material Technology Co., Ltd | +8613223293093 | bella@njdeda.com | China | 81 | 58 |
| Firsky International Trade (Wuhan) Co., Ltd | +8615387054039 | admin@firsky-cn.com | China | 436 | 58 |
| Anhui Yiao New Material Technology Co., Ltd | +86-199-55145978 +8619955145978 | sales8@anhuiyiao.com | China | 253 | 58 |
Related articles
- Preparation and Application of N-Methylformamide
- N-Methylformamide is an important organic chemical raw material and intermediate, and an organic solvent with good performance....
- Aug 19,2022
View Lastest Price from N-Methylformamide manufacturers
| Image | Update time | Product | Price | Min. Order | Purity | Supply Ability | Manufacturer | |
|---|---|---|---|---|---|---|---|---|
 |
2024-04-26 | N-Methylformamide
123-39-7
|
US $1.00 / kg | 1kg | 99% | 10 tons | Dorne Chemical Technology co. LTD | |
 |
2024-04-26 | N-Methylformamide/ NMF
123-39-7
|
US $10.00 / G | 1G | 99% | 20 | CONTIDE BIOTECH CO.,LTD | |
 |
2024-04-26 | N-Methylformamide
123-39-7
|
US $10.00-5.00 / kg | 1kg | 99.5 | 10 ton per month | Nanjing Deda New Material Technology Co., Ltd |
-

- N-Methylformamide
123-39-7
- US $1.00 / kg
- 99%
- Dorne Chemical Technology co. LTD
-

- N-Methylformamide/ NMF
123-39-7
- US $10.00 / G
- 99%
- CONTIDE BIOTECH CO.,LTD
-

- N-Methylformamide
123-39-7
- US $10.00-5.00 / kg
- 99.5
- Nanjing Deda New Material Technology Co., Ltd
123-39-7(N-Methylformamide)Related Search:
1of4