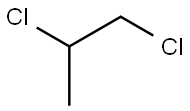
1,2-디클로로프로판
|
|
1,2-디클로로프로판 속성
- 녹는점
- −100 °C(lit.)
- 끓는 점
- 95-96 °C(lit.)
- 밀도
- 1.156 g/mL at 25 °C(lit.)
- 증기 밀도
- 3.89 (vs air)
- 증기압
- 40 mm Hg ( 19.4 °C)
- 굴절률
- n
20/D 1.439(lit.)
- 인화점
- 40 °F
- 저장 조건
- Refrigerator
- 용해도
- 유기용매와 혼합 가능(US EPA, 1985).
- 물리적 상태
- 액체
- 색상
- 무색투명~연황색
- 냄새
- 달콤한.
- 폭발한계
- 14.5%
- 수용성
- 3g/L(20℃)
- Merck
- 14,7854
- BRN
- 1718880
- Henry's Law Constant
- 0.81, 1.06, 1.32, 2.01, and 2.74 at 2.0, 6.0, 10.0, 18.0, and 25.0 °C, respectively (EPICS-SPME, Dewulf et al., 1999)
- 노출 한도
- Potential occupational carcinogen. NIOSH REL: IDLH 400 ppm; OSHA PEL: TWA 75 ppm (350 mg/m3); ACGIH TLV: TWA 75 ppm, STEL 110 ppm (adopted).
- Dielectric constant
- 9.0(Ambient)
- 안정성
- 탁한. 가연성이 높습니다. 강한 산화제와 호환되지 않습니다.
- InChIKey
- KNKRKFALVUDBJE-UHFFFAOYSA-N
- LogP
- 2-2.25 at 20-25℃
- CAS 데이터베이스
- 78-87-5(CAS DataBase Reference)
- IARC
- 1 (Vol. 41, Sup 7, 71, 110) 2017
안전
- 위험 및 안전 성명
- 위험 및 사전주의 사항 (GHS)
| 위험품 표기 | F,Xn,T | ||
|---|---|---|---|
| 위험 카페고리 넘버 | 11-20/22-39/23/24/25-23/24/25-23/25 | ||
| 안전지침서 | 16-24-45-36/37-7 | ||
| 유엔번호(UN No.) | UN 1279 3/PG 2 | ||
| WGK 독일 | 3 | ||
| RTECS 번호 | TX9625000 | ||
| 자연 발화 온도 | 1035 °F | ||
| TSCA | Yes | ||
| 위험 등급 | 3 | ||
| 포장분류 | II | ||
| HS 번호 | 29031990 | ||
| 유해 물질 데이터 | 78-87-5(Hazardous Substances Data) | ||
| 독성 | LD50 orally in rats: 1.19 ml/kg (Smyth) | ||
| IDLA | 400 ppm | ||
| 기존화학 물질 | KE-10202 | ||
| 유해화학물질 필터링 | 2017-1-763 | ||
| 중점관리물질 필터링 | 별표1-23 | ||
| 암, 돌연변이성물질 필터링 | 2 | ||
| 함량 및 규제정보 | 물질구분: 유독물질; 혼합물(제품)함량정보: 1,2-디클로로프로판 및 이를 25%이상 함유한 혼합물 |
1,2-디클로로프로판 C화학적 특성, 용도, 생산
화학적 성질
colourless liquid물리적 성질
Clear, colorless liquid with a sweet, chloroform-like odor. The average least detectable odor threshold concentration in water at 60 °C was 0.10 mg/L (Alexander et al., 1982). Experimentally determined detection and recognition odor threshold concentrations were 1.2 mg/m3 (260 ppbv) and 2.4 mg/m3 (520 ppbv), respectively (Hellman and Small, 1974).용도
1,2-Dichloropropane is a byproduct in the production of epichlorohydrin (E582310), an important industrial chemical, is a bifunctional alkylating agent with the potential to form DNA cross-links.일반 설명
Colorless watery liquid with a sweet odor. Sinks in water. Produces an irritating vapor.공기와 물의 반응
Highly flammable. Insoluble in water.반응 프로필
1,2-Dichloropropane reacts with strong oxidizers and strong acids. 1,2-Dichloropropane also reacts with aluminum. When confined, this reaction can lead to an explosion. 1,2-Dichloropropane is incompatible with bases and aluminum alloys. 1,2-Dichloropropane will attack some forms of plastics, rubber and coatings.위험도
Flammable, dangerous fire risk, explosive limits in air 3.4–14.5%. Toxic by ingestion and inhalation. Upper respiratory tract irritant and body weight effects. Questionable carcinogen.건강위험
Contact with skin or eyes may cause irriation.화재위험
Special Hazards of Combustion Products: Toxic and irritating gases may be generated.화학 반응
Reactivity with Water No reaction; Reactivity with Common Materials: No reaction; Stability During Transport: Stable; Neutralizing Agents for Acids and Caustics: Not pertinent; Polymerization: Not pertinent; Inhibitor of Polymerization: Not pertinent.Safety Profile
Suspected carcinogen with experimental carcinogenic data. Moderately toxic by inhalation and ingestion. Mddly toxic by skin contact. An eye irritant. Mutation data reported. Can cause liver, kidney, and heart damage. Can cause dermatitis. One of the more toxic chlorinated hydrocarbons. A suggested order of increasing toxicity is dichloromethane, trichloroethylene, carbon tetrachloride, dichloropropane, dichloroethane. Animals exposed to hgh concentrations often showed marked visceral congestion, fatty degeneration of the liver, kidney, and, less frequently, of the heart. They also showed areas of coagulation and necrosis of the liver. There was found to be a heavy mortality among mice exposed to 400 ppm concentrations. A flammable liquid and very dangerous fire hazard when exposed to heat or flame. Reacts with aluminum to form aluminum chloride. This reaction, when confined, can lead to explosion. Can react vigorously with oxidizing materials. To fight fire, use water, foam, CO2, dry chemical. When heated to decomposition it emits toxic fumes of Cl-. See also CHLORINATED HYDROCARBONS , ALIPH AT1 C잠재적 노출
Dichloropropane is used as a chemical intermediate in perchloroethylene and carbon tetrachloride synthesis, and as a lead scavenger for antiknock fluids. It is also used as a solvent for fats, oils, waxes, gums and resins; and in solvent mixtures for cellulose esters and ethers. Other applications include the use of dichloropropane; as a fumigant, alone and in combination with dichloropropane, as a scouring compound; and a metal degreasing agent. It is also used as an insecticidal fumigantCarcinogenicity
The EPA states, “This substance has not undergone a complete evaluation and determination under US EPA’s IRIS program for evidence of human carcinogenicity potential.”An epidemiological study of 71 Italian florists, who were found to have used an average of 162 kg/year of 1,2- dichloropropane, showed that the mean frequency of peripheral lymphocyte micronuclei for the florists was higher than the mean for 75 controls, but these findings are questionable.
An oral cancer bioassay conducted in rats and mice by NTP, concluded that there was “some evidence of carcinogenicity in both male and female mice based on incidences of liver tumors (primarily adenomas, equivocal evidence of carcinogenicity in female rats based on mammary gland adenocarcinomas and no evidence of carcinogenicity in male rats.” Additional detail is provided below. Propylene dichloride was fed by gavage to rats and mice, 5 days/week for 103 weeks. Dosageswere 0 (corn oil controls), 125, and 250 mg/kg for mice and 0, 62, and 125 mg/kg for rats. Survival was reduced in high-dose female rats and mice (possibly due to infection in female mice). Body weight was reduced in high-dose rats of both sexes, and clear-cell changes and necrosis of the livers were found in high-dose female rats. The investigators concluded that dose-related increases were observed for adenomas of the liver in both male (control, 7/50; low dose, 10/50; high dose, 17/50) and female mice. The increase in the frequency of liver carcinomas supported the evidence that there was a neoplastic response in the mouse liver for both sexes (males: 11/50, 17/50, 16/50; females: 1/50, 3/50, 4/50). Hepatocytomegaly and hepatic necrosis were increased in male mice, but not in female mice.
신진 대사 경로
1,2-Dichloropropane is quite stable to hydrolytic and microbial degradation in soils. It is metabolised rapidly and extensively in animals via a combination of gluthathione conjugation, reductive dechlorination, hydrolytic displacement of the halogen and oxidation and hydroxylation reactions (Scheme 1).운송 방법
UN1279 1,2-Dichloropropane, Hazard Class: 3; Labels: 3-Flammable liquid.Purification Methods
Distil the propane from CaH2. It has a limited shelf life. [Beilstein 1 IV 195.]비 호환성
May form explosive mixture with air. May accumulate static electrical charges, and may cause ignition of its vapors. Contact with strong oxidizers, powdered aluminum may cause fire and explosion hazard. Strong acids can cause decomposition and the formation of hydrogen chloride vapors. Reacts with strong bases; o-dichlorobenzene, 1,2-dichloroethane. Corrosive to aluminum and its alloys. Attacks some plastics, rubber, and coatings.폐기물 처리
Incineration, preferably after mixing with another combustible fuel. Care must be exercised to assure complete combustion to prevent the formation of phosgene. An acid scrubber is necessary to remove the halo acids produced. Consult with environmental regulatory agencies for guidance on acceptable disposal practices. Generators of waste containing this contaminant (≧100 kg/mo) must conform with EPA regulations governing storage, transportation, treatment, and waste disposal.1,2-디클로로프로판 준비 용품 및 원자재
원자재
준비 용품
1,2-디클로로프로판 공급 업체
글로벌( 277)공급 업체
| 공급자 | 전화 | 이메일 | 국가 | 제품 수 | 이점 |
|---|---|---|---|---|---|
| Henan Fengda Chemical Co., Ltd | +86-371-86557731 +86-13613820652 |
info@fdachem.com | China | 7845 | 58 |
| Henan Bao Enluo International TradeCo.,LTD | +86-17331933971 +86-17331933971 |
deasea125996@gmail.com | China | 2503 | 58 |
| Shanghai Daken Advanced Materials Co.,Ltd | +86-371-66670886 |
info@dakenam.com | China | 15928 | 58 |
| Henan Tianfu Chemical Co.,Ltd. | +86-0371-55170693 +86-19937530512 |
info@tianfuchem.com | China | 21691 | 55 |
| Hangzhou FandaChem Co.,Ltd. | 008657128800458; +8615858145714 |
fandachem@gmail.com | China | 9348 | 55 |
| career henan chemical co | +86-0371-86658258 |
sales@coreychem.com | China | 29914 | 58 |
| Hubei Jusheng Technology Co.,Ltd. | 18871490254 |
linda@hubeijusheng.com | CHINA | 28180 | 58 |
| Xi'an Kono chem co., Ltd., | 029-86107037 13289246953 |
info@konochemical.com | China | 2995 | 58 |
| Hubei xin bonus chemical co. LTD | 86-13657291602 |
linda@hubeijusheng.com | CHINA | 22968 | 58 |
| Shanghai Longyu Biotechnology Co., Ltd. | +8615821988213 |
info@longyupharma.com | China | 2531 | 58 |