인돌-3-아세트산
|
|
인돌-3-아세트산 속성
- 녹는점
- 165-169 °C (lit.)
- 끓는 점
- 306.47°C (rough estimate)
- 밀도
- 1.1999 (rough estimate)
- 굴절률
- 1.5460 (estimate)
- 인화점
- 171°C
- 저장 조건
- -20°C
- 용해도
- DMSO:30.0(Max Conc. mg/mL);171.25(Max Conc. mM)
- 물리적 상태
- 수정 같은
- 산도 계수 (pKa)
- 4.75(at 25℃)
- 색상
- 회백색부터 황갈색까지
- 수용성
- 에탄올(50 mg/ml), 메탄올, DMSO 및 클로로포름(드물게)에 용해됩니다. 물에 불용성.
- 분해온도
- 167 ºC
- 감도
- Light Sensitive
- Merck
- 14,4964
- BRN
- 143358
- 안정성
- 안정적인. 강한 산화제와 호환되지 않습니다. 빛에 민감합니다.
- LogP
- 1.410
- CAS 데이터베이스
- 87-51-4(CAS DataBase Reference)
안전
- 위험 및 안전 성명
- 위험 및 사전주의 사항 (GHS)
| 위험품 표기 | Xi | ||
|---|---|---|---|
| 위험 카페고리 넘버 | 36/37/38 | ||
| 안전지침서 | 22-24/25 | ||
| WGK 독일 | 3 | ||
| RTECS 번호 | NL3150000 | ||
| F 고인화성물질 | 8-10-23 | ||
| 위험 참고 사항 | Irritant | ||
| TSCA | Yes | ||
| HS 번호 | 29339990 | ||
| 독성 | LD50 intraperitoneal in mouse: 150mg/kg | ||
| 기존화학 물질 | KE-21008 |
인돌-3-아세트산 C화학적 특성, 용도, 생산
화학적 성질
white to tan crystals용도
Plant growth regulator.정의
ChEBI: A monocarboxylic acid that is acetic acid in which one of the methyl hydrogens has been replaced by a 1H-indol-3-yl group.Biosynthesis
3-Indolylacetic acid is biosynthesised in plants from tryptophan by two pathways, the indolylpyruvic acid pathway being quantitatively the more important. Experiments with tomato shoots have shown the existence of a tryptophan transaminase, which catalyses the formation of indolylpyruvic acid, and a tryptophan decarboxylase, which catalyses the formation of tryptamine. The decarboxylation of indolylpyruvic acid is catalysed by indolylpyruvate decarboxylase, while indolylacetaldehyde dehydrogenase catalyses the oxidation of indolylacetaldehyde to indolylacetic acid.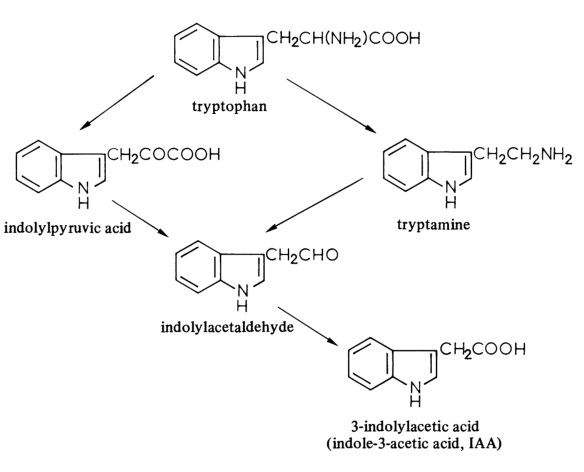
The biosynthesis of 3-indolylacetic acid
Biological Functions
3-Indolylacetic acid (indole-3-acetic acid, IAA) is one of the auxins, which together with the gibberellins and abscisic acid, cyto- kinins and ethylene are hormones regulating the growth and development of plants. IAA is a ubiquitous constituent of higher plants and the most important auxin. Some other, non-indolic compounds, including phenyl- acetic acid biosynthesised in plants from phenylalanine, have similar properties and synthetic auxins have also been prepared.In the plant, IAA conjugates with many compounds, including glucose and other sugars, and with aspartic and glutamic acids. This is probably a way of storing the hormone for future use.
IAA initiates many growth effects in plants, including geotropism and phototropism, development of the ovary, division of cells, enlargement in callus tissue, root formation and apical dominance. When fed to plants, the hormone causes growth up to a maximum, which depends on the type of tissue being fed, and thereafter inhibits further growth, probably through the formation of ethylene, which is growth-inhibitory. Stern tissues tolerate the highest levels of IAA and root tissues the lowest. In the plant, the most active sites of IAA synthesis are the young, expanding leaves.
일반 설명
3-Indoleacetic acid is a highly effective, growth promotor in lower plant life, formed by a variety of fungi, including yeast and has been isolated from Aspergillus niger and Rhizopus sp. It is commonly employed in horticulture and industry.농업용
Indoleacetic acid (IAA), synthesized in the plant shoot tips, is a naturally occurring auxin. It is a plant growth promoter.Purification Methods
Recrystallise heteroauxin from EtOH/water [James & Ware J Phys Chem 89 5450 1985]. [Beilstein 22 III/IV 65.] Alternatively recrystallise 30g of the acid with 10g of charcoal in 1L of hot water, filter and cool when 22g of colourless acid separate. Dry it and store it in a dark bottle away from direct sunlight [Johnson & Jacoby Org Synth Coll Vol V 654 1973]. The picrate has m 178-180o. [Beilstein 22 H 66, 22 I 508, 22 II 50, 22 III/IV 1088.] It is a plant growth substance.인돌-3-아세트산 준비 용품 및 원자재
원자재
준비 용품
1-METHYL-3-INDOLEACETIC ACID
3-(2-chloroethyl)-1H-indole
Tryptophol
4-Quinolinecarboxaldehyde
1H-Indole-3-acetic acid, α-methyl-, methyl ester
1-(1H-Benzotriazol-1-yl)-2-(1H-indol-3-yl)ethanone
Ethyl N-Boc-(indol-3-yl)acetate/tert-butyl 3-(2-ethoxy-2-oxoethyl)-1H-indole-1-carboxylate
N-2-(indol-3-yl)ethyl-indole-3-acetamide
인돌-3-아세트산 공급 업체
글로벌( 791)공급 업체
| 공급자 | 전화 | 이메일 | 국가 | 제품 수 | 이점 |
|---|---|---|---|---|---|
| Wuhu Nuowei chemistry Co., Ltd. | +86-0553-2911116-802 +undefined17756524438 |
sales1@nuowei-chem.com | China | 1642 | 58 |
| Hebei Mojin Biotechnology Co., Ltd | +8613288715578 |
sales@hbmojin.com | China | 12456 | 58 |
| Shaanxi Haibo Biotechnology Co., Ltd | +undefined18602966907 |
qinhe02@xaltbio.com | China | 1000 | 58 |
| Capot Chemical Co.,Ltd. | 571-85586718 +8613336195806 |
sales@capotchem.com | China | 29797 | 60 |
| Henan Tianfu Chemical Co.,Ltd. | +86-0371-55170693 +86-19937530512 |
info@tianfuchem.com | China | 21691 | 55 |
| Hefei TNJ Chemical Industry Co.,Ltd. | +86-0551-65418679 +86-18949832763 |
info@tnjchem.com | China | 2989 | 55 |
| Shanghai Zheyan Biotech Co., Ltd. | 18017610038 |
zheyansh@163.com | CHINA | 3620 | 58 |
| career henan chemical co | +86-0371-86658258 |
sales@coreychem.com | China | 29914 | 58 |
| NINGBO INNO PHARMCHEM CO., LTD. | 13867897135 |
sales@nbinno.com | CHINA | 925 | 58 |
| Hubei Jusheng Technology Co.,Ltd. | 18871490254 |
linda@hubeijusheng.com | CHINA | 28180 | 58 |