- Teneligliptin
-

- $0.00/ kg
-
2024-09-25
- CAS:760937-92-6
- Min. Order: 1kg
- Purity: 99%, Single impurity<0.1
- Supply Ability: 1 ton
- Teneligliptin
-
- $0.00 / 1KG
-
2022-09-16
- CAS:760937-92-6
- Min. Order: 1KG
- Purity: 98%
- Supply Ability: 1ton
- Teneligliptin
-

- $15.00 / 1KG
-
2021-07-13
- CAS:760937-92-6
- Min. Order: 1KG
- Purity: 99%+ HPLC
- Supply Ability: Monthly supply of 1 ton
|
| | Teneligliptin Basic information |
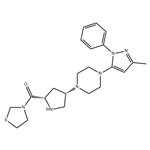
| Product Name: | Teneligliptin | | Synonyms: | teneligliptin;3-[[(2S,4S)-4-[4-(3-Methyl-1-phenyl-1H-pyrazol-5-yl)-1-piperazinyl]-2-pyrrolidinyl]carbonyl]thiazolidine;Methanone,[(2S,4S)-4-[4-(3-Methyl-1-phenyl-1H-pyrazol-5-yl)-1-piperazinyl]-2-pyrrolidinyl]-3-thiazolidinyl-;Teneligptin WS;[(2S,4S)-4-[4-(5-methyl-2-phenylpyrazol-3-yl)piperazin-1-yl]pyrrolidin-2-yl]-(1,3-thiazolidin-3-yl)methanone;API (Teneligliptin free base);Teliglietin;Teneligliptin (MP-513) | | CAS: | 760937-92-6 | | MF: | C22H30N6OS | | MW: | 426.58 | | EINECS: | | | Product Categories: | | | Mol File: | 760937-92-6.mol | 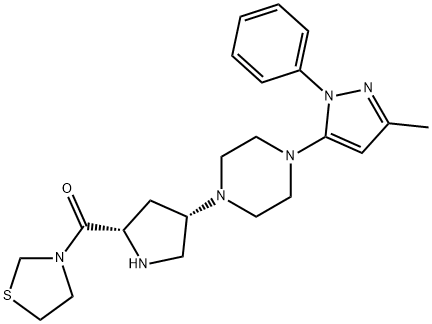 |
| | Teneligliptin Chemical Properties |
| Melting point | >100°C (dec.) | | Boiling point | 663.4±55.0 °C(Predicted) | | density | 1.38 | | storage temp. | Refrigerator | | solubility | DMSO (Slightly), Methanol (Slightly) | | form | Solid | | pka | 8.70±0.10(Predicted) | | color | Off-White | | InChIKey | WGRQANOPCQRCME-PMACEKPBSA-N | | SMILES | C([C@@H]1C[C@H](N2CCN(C3N(C4=CC=CC=C4)N=C(C)C=3)CC2)CN1)(N1CCSC1)=O |
| | Teneligliptin Usage And Synthesis |
| Description | Teneligliptin was approved in September 2012 in Japan for the treatment of patients with Type 2 diabetes mellitus (T2DM). Teneligliptin is a member of the dipeptidyl peptidase 4 (DPP-4) inhibitor class of antidiabetes agents. DPP-4 is an enzyme that degrades GLP-1, a 30-amino acid peptide that is secreted in response to food intake. GLP-1 stimulates insulin secretion and inhibits glucagon secretion, which leads to lower levels of plasma glucose. Teneligliptin is one of a growing numbers of DPP-4 inhibitors to be approved worldwide. The discovery of teneligliptin was guided by structure-based design, with a key element being binding of the phenyl group in the S2 pocket, which not only increases potency for DPP-4, but also improves selectivity versus DPP-8 and DPP- 9. Teneligliptin is a potent inhibitor of DPP-4 in the enzyme inhibition assay (IC50=0.37 nM). | | Originator | Mitsubishi Tanabe Pharma (Japan) | | Uses | Teneligliptin, is a dipeptidyl peptidase-4 (DPP-4) inhibitor that is used to treat type 2 diabetes. It is eliminated via excretion, and has a half-life of 24.2 hours in the human body. | | Definition | ChEBI: Teneligliptin is an amino acid amide. | | Brand name | Tenelia | | Pharmacokinetics | Reported evidence suggests that with teneligliptin 20 mg therapy, Tmax was 1 hour, and t1/2 was 18.9 hours. Maximum (89.7%) inhibition in plasma DPP-4 activity was noted within 2 hours and maintained >60% at 24 hours. Compared to the placebo, active plasma GLP-1 concentration was higher throughout the day and even 24 hours after administration of 20 mg of teneligliptin. Metabolism of teneligliptin was majorly mediated through CYP3A4, a cytochrome P450 isozyme, and flavin-containing monooxygenases (FMO1 and FMO3). A weak inhibitory activity of teneligliptin on CYP2D6, CYP3A4, and FMO was noted, while there was no inhibitory activity on CYP1A2, CYP2A6, CYP2B6, CYP2C8, CYP2C8/9, CYP2C19, and CYP2E1. There was no induction of expression of CYP1A2 or CYP3A4. Reported evidence suggests that teneligliptin is metabolized and eliminated via renal and hepatic routes. Approximately 34% of teneligliptin is excreted unchanged via the renal route, while 66% is metabolized and eliminated via the hepatic and renal routes[1].
| | Side effects |
The DPP-4 molecule in teneligliptin reduces the risk of hypoglycemia, although it is possible. These are some of teneligliptin’s major and minor side effects:
Hypoglycemia
Headache
Upper respiratory tract infection
Gastrointestinal symptoms
Constipation
Fatigue
Kidney problems
Nasopharyngitis
Skin rashes
Itching
Loss of appetite
Abdominal pain
| | References | [1] Surendra Kumar Sharma. “Teneligliptin in management of type 2 diabetes mellitus.” Diabetes, Metabolic Syndrome and Obesity: Targets and Therapy 9 (2016): 251–60. |
| | Teneligliptin Preparation Products And Raw materials |
|