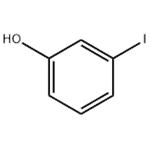
- 3-Iodophenol
-

- $0.00 / 1KG
-
2026-03-20
- CAS:626-02-8
- Min. Order: 1KG
- Purity: 99%
- Supply Ability: 20 mt
- 3-Iodophenol
-

- $750.00/ kg
-
2026-02-02
- CAS:626-02-8
- Min. Order: 1kg
- Purity: 98% GC
- Supply Ability: 10T
- 3-Iodophenol
-
- $100.00 / 1KG
-
2025-09-25
- CAS:626-02-8
- Min. Order: 1KG
- Purity: 99%
- Supply Ability: g-kg-tons, free sample is available
|
| | 3-Iodophenol Basic information |
| | 3-Iodophenol Chemical Properties |
| Melting point | 42-44 °C (lit.) | | Boiling point | 190 °C / 100mmHg | | density | 1.8665 (estimate) | | Fp | >230 °F | | storage temp. | 2-8°C | | solubility | Chloroform, Ethyl Acetate | | form | Crystalline Powder, Crystals and/or Chunks | | pka | 9.03(at 25℃) | | color | Yellow-beige to gray | | Water Solubility | slightly soluble | | Sensitive | Light Sensitive | | BRN | 2039304 | | Henry's Law Constant | 7.0×101 mol/(m3Pa) at 25℃, Hilal et al. (2008) | | InChI | 1S/C6H5IO/c7-5-2-1-3-6(8)4-5/h1-4,8H | | InChIKey | FXTKWBZFNQHAAO-UHFFFAOYSA-N | | SMILES | Oc1cccc(I)c1 | | CAS DataBase Reference | 626-02-8(CAS DataBase Reference) | | NIST Chemistry Reference | Phenol, 3-iodo-(626-02-8) | | EPA Substance Registry System | m-Iodophenol (626-02-8) |
| Hazard Codes | Xi | | Risk Statements | 36/37/38 | | Safety Statements | 26-37/39-24/25 | | RIDADR | UN 3335 | | WGK Germany | 2 | | RTECS | SL5490000 | | TSCA | TSCA listed | | HazardClass | IRRITANT | | HS Code | 29081900 | | Storage Class | 11 - Combustible Solids | | Hazard Classifications | Eye Irrit. 2
Skin Irrit. 2
STOT SE 3 |
| | 3-Iodophenol Usage And Synthesis |
| Chemical Properties | yellow-beige to grey crystalline powder, crystals | | Uses | 3-Iodophenol is used in esterification, electrophilic aromatic substitution, alkylation, nucleophilic aromatic substitution, Mitsunobu, transition metal-catalyzed cross-coupling, acylation, and oxidation reactions. | | Definition | ChEBI: 3-iodophenol is an iodophenol. | | Application | 3-Iodophenol is used in the synthesis of centrally acting H2 receptor histamine antagonists. Also used in the preparation of various cell-permeable probes for biological assays. | | Synthesis | 3-Iodophenol Synthesis:
1£Add potassium tert-butoxide (67.3 mg, 0.60 mmol, 2.0 eq.), oxime (74.5 mg, 0.60 mmol, 2.0 eq.), and 4-bromoacetophenone (59.7 mg, 0.30 mmol, 1.0 eq.) to an oven-dry screw-cap 8mL reaction vial.
2£Add anhydrous DMSO (1.5 mL) to the solids a magnetic stir bar.
3£Close the vial and sparge the reaction mixture with N2 for 15 minutes, seal with parafilm.
4£Stir the reaction mixture and heat at 100C in a metal heating block for 16 h.
5£Dilute the mixture with CH2Cl2 or EtOAc (20 mL), pour into a mixture of water (10mL) and brine (5 mL), acidify with 10% aq HCl (~1 mL).
6£Collect the organic phase and extract the aqueous phase with CH2Cl2 or EtOAc (3á20mL).
7£Combine the organics, dry (MgSO4) and concentrate under reduced pressure.
8£Purify the crude product to give 3-Iodophenol by column chromatography (30% EtOAc in hexane).
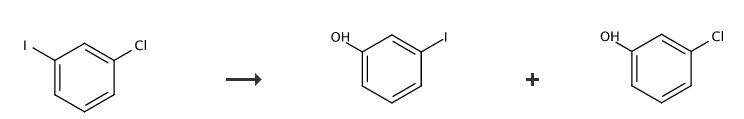
| | References | [1] Tetrahedron Asymmetry, 2013, vol. 24, # 13-14, p. 827 - 832
[2] Chemische Berichte, 1887, vol. 20, p. 3019 |
| | 3-Iodophenol Preparation Products And Raw materials |
|