Uses of Fleroxacin
Mar 25,2022
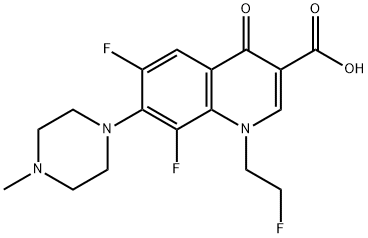
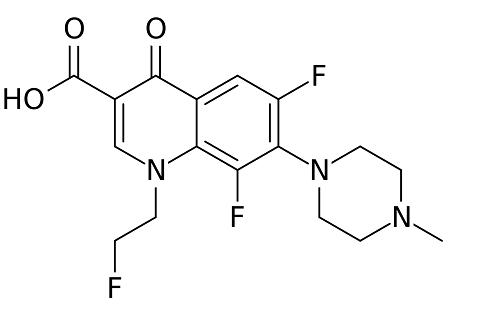
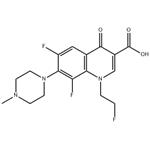
Fleroxacin (Ro 23-6240, AM 833) is different from fluoroquinolones such as ciprofloxacin or ofloxacin, since it is a trifluorinated quinolone, having three fluorine atoms rather than one attached to the quinolone ring system. It is a 6,8-difluoro-1-(2-fluorethyl)-1,4-dihydro-7- (4-methyl-1-piperazinyl)-4-oxo-3-quinolonine carboxylic acid. A major advantage of fleroxacin is its long half-life, allowing once-daily dosing. However, the extra fluorine atoms have led to a higher rate of dose-related adverse side-effects than other fluoroquinolones, leading to the withdrawal of the drug in some countries.
Uses
Fleroxacin has been used in a variety of clinical conditions, but there have been few studies in recent years owing to the availability of other fluoroquinolones with improved toxicity profiles. Nevertheless, for regions where fleroxacin remains available and adverse reactions can be readily monitored, fleroxacin has activity in a number of conditions.
Bioavailability
Oral absorption is 95–100% in healthy volunteers, substantially better than that of many other early fluoroquinolones. Owing to this excellent bioavailability, serum fleroxacin concentrations are nearly identical for oral and i.v. doses. Fleroxacin is 32% protein bound. The oral bioavailability of fleroxacin is not substantially affected by the intake of a light meal, or a fat- or calcium-rich breakfast. Although co-administration of calcium-containing antacids does not alter fleroxacin bioavailability, aluminum- and magnesium-containing antacids and sucralfate result in approximately a 25% reduction in maximum serum fleroxacin concentrations and AUC – less than for other fluoroquinolones.
Toxicity
Fleroxacin was withdrawn from the market in Europe by Roche in the late 1990s due to two significant dose-related adverse events: phototoxicity and central nervous system toxicity. These adverse events were noted in premarketing trials, but increasing reports in the postmarketing period led to withdrawal of the drug in Europe.
- Related articles
- Related Qustion
Tosufloxacin was developed by Taisho-Toyama Chemistry (Ozexs) and became commercially available in 1990. It is a newer-generation oral fluoroquinolone with a broad spectrum of activity against Gramnegative, Gram-positive, and anaerobic orga....
Mar 25,2022APITemafloxacin (6-fluoro-7-piperazino-4-quinolone)was discontinued in June 1992, only five months after gaining marketing approval. This was due to a high rate of reported adverse reactions, including several deaths.....
Mar 25,2022APIFleroxacin
79660-72-3You may like
- Fleroxacin
-
- $0.00 / 1kg
- 2025-12-13
- CAS:79660-72-3
- Min. Order: 1kg
- Purity: 98%
- Supply Ability: Customise
- Fleroxacin
-

- $40.00 / 1g
- 2025-12-12
- CAS:79660-72-3
- Min. Order:
- Purity: 99.79%
- Supply Ability: 10g
- Fleroxacin
-

- $40.00 / 1g
- 2025-12-12
- CAS:79660-72-3
- Min. Order:
- Purity: 99.79%
- Supply Ability: 10g