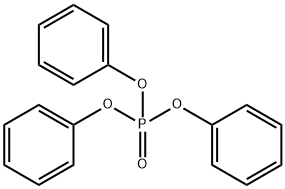
Triphenyl phosphate
- CAS No.
- 115-86-6
- Chemical Name:
- Triphenyl phosphate
- Synonyms
- TPP;TPPA;Tripheyl phosphate;TRIPHENYL PHOSPHATE (TPP);PHOSPHORIC ACID TRIPHENYL ESTER;Phosflex TPP;Triphenyl hosphate;Altal;Dymel;Wako TPP
- CBNumber:
- CB9233809
- Molecular Formula:
- C18H15O4P
- Molecular Weight:
- 326.28
- MDL Number:
- MFCD00003031
- MOL File:
- 115-86-6.mol
- MSDS File:
- SDS
| Melting point | 48-50 °C (lit.) |
|---|---|
| Boiling point | 244 °C/10 mmHg (lit.) |
| Density | 1.2055 |
| vapor density | 11.3 (vs air) |
| vapor pressure | 1.3 mm Hg ( 200 °C) |
| refractive index | 1.563 |
| Flash point | 435 °F |
| storage temp. | Store below +30°C. |
| solubility | H2O: soluble0.0019g/L at 20°C |
| form | Crystalline Flakes |
| color | White |
| Odor | Odorless |
| Viscosity | 11mm2/s |
| Water Solubility | insoluble |
| Merck | 14,9742 |
| BRN | 1888236 |
| Henry's Law Constant | 5.88 at 20 °C (approximate - calculated from water solubility and vapor pressure) |
| Exposure limits | TLV-TWA air 3 mg/m3 (ACGIH, OSHA, and NIOSH). |
| Stability | Stable. |
| InChIKey | XZZNDPSIHUTMOC-UHFFFAOYSA-N |
| LogP | 4.6 at 20℃ |
| Indirect Additives used in Food Contact Substances | TRIPHENYL PHOSPHATE |
| FDA 21 CFR | 175.105 |
| CAS DataBase Reference | 115-86-6(CAS DataBase Reference) |
| EWG's Food Scores | 5 |
| FDA UNII | YZE19Z66EA |
| NIST Chemistry Reference | Triphenylphosphate(115-86-6) |
| EPA Substance Registry System | Triphenyl phosphate (115-86-6) |
SAFETY
Risk and Safety Statements
| Symbol(GHS) |   GHS06,GHS08 |
|||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Signal word | Danger | |||||||||
| Hazard statements | H302-H315-H319-H331-H336-H351-H361d-H372 | |||||||||
| Precautionary statements | P201-P301+P312-P302+P352-P304+P340+P311-P305+P351+P338-P308+P313 | |||||||||
| Hazard Codes | Xn,N,F,Xi | |||||||||
| Risk Statements | 22-38-40-48/20/22-50/53-52/53-36/37/38-67-66-36-11-51/53 | |||||||||
| Safety Statements | 36/37-61-60-24/25-22-36-26-16-24-9 | |||||||||
| OEB | B | |||||||||
| OEL | TWA: 3 mg/m3 | |||||||||
| RIDADR | UN 3077 9/PG 3 | |||||||||
| WGK Germany | 2 | |||||||||
| RTECS | TC8400000 | |||||||||
| Autoignition Temperature | >500 °C | |||||||||
| TSCA | Yes | |||||||||
| HazardClass | 9 | |||||||||
| PackingGroup | III | |||||||||
| HS Code | 29190010 | |||||||||
| Toxicity | LD50 orally in Rabbit: 3500 mg/kg LD50 dermal Rabbit > 7900 mg/kg | |||||||||
| IDLA | 1,000 mg/m3 | |||||||||
| NFPA 704 |
|
Triphenyl phosphate price More Price(28)
| Manufacturer | Product number | Product description | CAS number | Packaging | Price | Updated | Buy |
|---|---|---|---|---|---|---|---|
| Sigma-Aldrich | 8.21197 | Triphenyl phosphate for synthesis | 115-86-6 | 100g | $40 | 2024-03-01 | Buy |
| Sigma-Aldrich | 8.21197 | Triphenyl phosphate for synthesis | 115-86-6 | 1kg | $77.2 | 2024-03-01 | Buy |
| Sigma-Aldrich | 756911 | Triphenyl phosphate solution NMR reference standard, 3?mM in chloroform-d (99.8 atom % D), NMR tube size 5?mm × 8?in. | 115-86-6 | 1pkg | $87.4 | 2024-03-01 | Buy |
| Sigma-Aldrich | 733792 | Triphenyl phosphate solution NMR reference standard, 0.0485?M in acetone-d6 (99.9 atom % D), NMR tube size 10?mm × 8?in. | 115-86-6 | 1pkg | $397 | 2024-03-01 | Buy |
| Sigma-Aldrich | 733741 | Triphenyl phosphate solution NMR reference standard, 0.0485?M in acetone-d6 (99.9 atom % D), NMR tube size 3?mm × 8?in. | 115-86-6 | 1pkg | $374 | 2024-03-01 | Buy |
Triphenyl phosphate Chemical Properties,Uses,Production
Chemical Properties
Triphenyl phosphate (TPP) is a colorless crystalline powder with a faint, phenol-like odor. Triphenyl phosphate is practically insoluble in water at 1.9 × 10 7 mg/l at 24 °C (Yalkowsky et al., 2010). It is very soluble in carbon tetrachloride (Haynes, 2010) and is soluble in most lacquers, solvents, thinners, and oils, as well as in alcohol, benzene, ether, chloroform, and acetone (Lewis, 1996). It begins to decompose at about 600 °C in inert gas, and in a large excess of air, complete combustion to carbon dioxide occurs in the range 800–900 °C (Lhomme et al., 1984). Hydrolysis of TPP occurs very slowly in acidic or neutral solutions, but occurs rapidly in alkaline solutions (Barnard et al., 1966).
Uses
Triphenyl phosphate is known as a product with manifold fields of applications regarding its qualities in particular as a flame retardant. One primary use is as a flame retardant in phenolic- and phenylene oxide-based resins for the manufacture of electrical and automobile components, for auto upholstery, and as a nonflammable plasticizer in cellulose acetate for photographic films. It has also been used to impregnate roofing paper. Triphenyl phosphate occurs as a plasticizer in various lacquers and varnishes (O'Neil, 2006), and as a component of lubricating oil and hydraulic fluids (ACGIH, 2012).
Definition
ChEBI: Triphenyl phosphate is an aryl phosphate resulting from the formal condensation of phosphoric acid with 3 mol eq. of phenol. It has a role as a flame retardant and a plasticiser. It is functionally related to a phenol.
Application
Triphenyl Phosphate is used in the insecticidal composition. It is also use in hydraulic liquids, and adhesives, inks, coatings, as a plasticizer in lacquers and varnishes, and as a substitute for camphor in celluloid materials to make the latter stable and fireproof.
Preparation
Triphenyl phosphate is prepared by reacting phosphorus pentoxide and phenol (Budavari, 2001), or by reacting phosphorus oxychloride and phenol (Snyder, 1990). On a larger scale phosphorus oxychloride and phenol are reacted in an esterification tank with heating. The HCL formed is trapped and condensed, while the crude triphenyl phosphate runs into a large tank where it is purified.
Reactivity Profile
Organophosphates, such as Triphenyl phosphate, are susceptible to formation of highly toxic and flammable phosphine gas in the presence of strong reducing agents such as hydrides. Partial oxidation by oxidizing agents may result in the release of toxic phosphorus oxides.
Hazard
Toxic by inhalation. Cholinesterase inhibitor. Questionable carcinogen.
Health effects
Non-industrial:
An allergic reaction in a 67-year old woman to spectacle frames containing triphenyl phosphate was reported. Patch tests with analytical grade triphenyl phosphate in that individual indicated a reaction at concentrations as low as 0.05%. This observation was confirmed in another male patient (Carlsen et al 1986).
Industrial:
Occupational exposure of men engaged in manufacturing triphenyl phosphate produced a statistically significant reduction in erythrocyte acetylcholinesterase activity and plasma cholinesterase activity. There was no evidence of adverse clinical effects in men exposed to triphenyl phosphate for as long as 10 years. Exposure was to triphenyl phosphate mist, vapor, and dust at a weighted average air concentration of 3.5 mg/m3 (Sutton et al 1960).
Fire Hazard
Noncombustible solid. Incompatibility— none.
Safety Profile
Poison by subcutaneous route. Moderately toxic by ingestion. Absorbed slowly, particularly by skin contact. Not a potent cholinesterase inhibitor. Combustible when exposed to heat or flame. To fight fire, use CO2, dry chemical. When heated to decomposition it emits toxic fumes of POx. See also TRITOLYL PHOSPHATE.
Potential Exposure
Triphenyl phosphate is used to impregnate roofing paper and as a fire-resistant plasticizer in plastics; for cellulose esters in lacquers and varnishes. Used in making adhesives, gasoline additives; flotation agents; insecticides, surfactants, antioxidants, and stabilizers. A substitute for camphor.
Source
Triphenyl phosphate was identified as a component in outer covers of brand-new computer video display units. Concentrations were estimated to be 8 to 10 and 0.3 to 0.5 wt % in 4 and 6 video display units, respectively. The concentrations of triphenyl phosphate in the remaining 8 video display units were <0.02 wt % (Carlsson et al., 2000).
Environmental Fate
Chemical/Physical. When an aqueous solution containing triphenyl phosphate (0.1 mg/L) and
chlorine (3 to 1,000 mg/L) was stirred in the dark at 20 °C for 24 h, the benzene ring was
substituted with one to three chlorine atoms (Ishikawa and Baba, 1988). The reported hydrolysis
half-lives at pH values of 8.2 and 9.5 were 7.5 and 1.3 d, respectively (Howard and Doe, 1979).
Decomposes at temperatures greater than 410 °C (Dobry and Keller, 1957)
Metabolism
Rat liver microsomal enzymes degraded triphenyl phosphate in the presence of NADPH, but also in the absence of NADPH. The product of incubation was diphenyl phosphate. It was clear that the reaction was cytochrome P-450-linked since the reaction was inhibited by carbon monoxide (Sasaki et al 1984). Goldfish liver microsomes metabolized only about 10% of triphenyl phosphate (Sasaki et al 1985). Houseflies treated with triphenyl phosphate were analyzed after 24 h and the presence of diphenyl p-hydroxyphenyl phosphate was confirmed (Eto et al 1975).
Shipping
UN3077 Environmentally hazardous substances, solid, n.o.s., Hazard class: 9; Labels: 9-Miscellaneous hazardous material, Technical Name Required.
Purification Methods
Crystallise the phosphate from EtOH or pet ether (b 60-80o)/EtOH. [Cox & Westheimer J Am Chem Soc 80 5441 1958, Krishnakumar & Sharma Synthesis 558 1983, Cherbuliez in Organo Phosphorus Compounds (Kosolapoff & Maier eds) Wiley Vol 6 pp 211-577 1973, Beilstein 6 III 658, 6 IV 720.]
Toxicity evaluation
Triphenyl phosphate(TPP) is neurotoxic, causing paralysis at high dosages. Like tri-o-cresyl phosphate (TOCP), it is a cholinesterase inhibitor. The acute oral toxicity is low. The acute toxicity via subcutaneous administration is low to moderate. The toxic symptoms from high dosages in test animals were tremor, diarrhea, muscle weakness, and paralysis.
LD50 value, oral (mice): 1320 mg/kg
LD50 value, subcutaneous (cats): 100 mg/kg
Cleveland et al. (1986) investigated the acute and chronic toxicity to various species of freshwater fish of phosphate ester compounds containing TPP. The adverse toxic effects occurred at exposure concentrations of 0.38–1.0 mg/L.
Incompatibilities
Incompatible with strong oxidizers; strong acids; nitrates may cause fire or explosions. Phosphates are incompatible with antimony pentachloride, magnesium, silver nitrate, zinc acetate.
Waste Disposal
Incinerate in furnace equipped with alkaline scrubber.
Triphenyl phosphate Preparation Products And Raw materials
Raw materials
1of3
Preparation Products
| Supplier | Tel | Country | ProdList | Advantage | |
|---|---|---|---|---|---|
| Tianjin Zhongxin Chemtech Co., Ltd. | +86-022-66880623 +8618622897568 | sales@tjzxchem.com | China | 564 | 58 |
| Hebei Mojin Biotechnology Co., Ltd | +86 13288715578 +8613288715578 | sales@hbmojin.com | China | 12465 | 58 |
| Hebei Chuanghai Biotechnology Co,.LTD | +86-13131129325 | sales1@chuanghaibio.com | China | 5895 | 58 |
| Changzhou waston chemical technology Co.,Ltd | +86-051985861892 +8618112881323 | info@wastonchem.com | China | 250 | 58 |
| Hebei Kingfiner Technology Development Co.Ltd | +86-15532196582 +86-15373005021 | lisa@kingfinertech.com | China | 2990 | 58 |
| Henan Fengda Chemical Co., Ltd | +86-371-86557731 +86-13613820652 | info@fdachem.com | China | 20291 | 58 |
| Capot Chemical Co.,Ltd. | 571-85586718 +8613336195806 | sales@capotchem.com | China | 29798 | 60 |
| Dalian Richfortune Chemicals Co., Ltd | 0411-84820922 8613904096939 | sales@richfortunechem.com | China | 304 | 57 |
| Henan Tianfu Chemical Co.,Ltd. | +86-0371-55170693 +86-19937530512 | info@tianfuchem.com | China | 21666 | 55 |
| Shanghai Time Chemicals CO., Ltd. | +86-021-57951555 +8617317452075 | jack.li@time-chemicals.com | China | 1807 | 55 |
Related articles
- Triphenyl Phosphate: Applications and its Reproductive Toxicity
- Triphenyl phosphate enhances fire safety in consumer goods but poses reproductive toxicity concerns, necessitating stringent r....
- Jul 18,2024
- Preparation method of triphenyl phosphate
- Triphenyl phosphate (TPhP), a typical organophosphate ester, is frequently detected in the environment and biota samples.
- Aug 16,2022
View Lastest Price from Triphenyl phosphate manufacturers
| Image | Update time | Product | Price | Min. Order | Purity | Supply Ability | Manufacturer | |
|---|---|---|---|---|---|---|---|---|
 |
2024-09-23 | Triphenyl Phosphate (TPP)
115-86-6
|
US $9.60 / KG | 1KG | 99% | 5000kg | Hebei Chuanghai Biotechnology Co,.LTD | |
 |
2024-06-20 | Triphenyl phosphate
115-86-6
|
US $0.00-0.00 / kg | 1kg | 99% | 50000kg | Hebei Kingfiner Technology Development Co.Ltd | |
 |
2024-03-16 | TPP | US $0.00 / KG | 100g | 98%+ | 100kg | WUHAN CIRCLE POWDER TECHNOLOGY CO.,LTD |
-

- Triphenyl Phosphate (TPP)
115-86-6
- US $9.60 / KG
- 99%
- Hebei Chuanghai Biotechnology Co,.LTD
-

- Triphenyl phosphate
115-86-6
- US $0.00-0.00 / kg
- 99%
- Hebei Kingfiner Technology Development Co.Ltd
-

- TPP
- US $0.00 / KG
- 98%+
- WUHAN CIRCLE POWDER TECHNOLOGY CO.,LTD