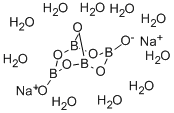
붕산나트륨10수화물
|
|
붕산나트륨10수화물 속성
- 녹는점
- 75 °C
- 끓는 점
- 320°C
- 밀도
- 1.73 g/mL at 25 °C(lit.)
- 증기압
- 0.213 hPa (20 °C)
- 저장 조건
- 2-8°C
- 용해도
- H2O: 0.1 M at 20 °C, 투명, 무색
- 물리적 상태
- 분말/고체
- Specific Gravity
- 1.73
- 색상
- 흰색, 회색, 푸르스름한 또는 녹색을 띤 흰색
- 수소이온지수(pH)
- 9.21(1 mM solution);9.17(10 mM solution);9.05(100 mM solution);
- 냄새
- 냄새 없는
- pH 범위
- 9.2
- 수용성
- 60g/L(20℃)
- 최대 파장(λmax)
- λ: 260 nm Amax: 0.012
λ: 280 nm Amax: 0.010
- Merck
- 14,8590
- 노출 한도
- ACGIH: TWA 2 mg/m3; STEL 6 mg/m3
NIOSH: TWA 5 mg/m3
- InChIKey
- CDMADVZSLOHIFP-UHFFFAOYSA-N
- CAS 데이터베이스
- 1303-96-4
안전
- 위험 및 안전 성명
- 위험 및 사전주의 사항 (GHS)
| 위험품 표기 | Xn,T | ||
|---|---|---|---|
| 위험 카페고리 넘버 | 62-61-60 | ||
| 안전지침서 | 22-24/25-36/37-45-53 | ||
| 유엔번호(UN No.) | UN 1458 | ||
| WGK 독일 | 1 | ||
| RTECS 번호 | VZ2275000 | ||
| TSCA | Yes | ||
| HS 번호 | 28401990 | ||
| 유해 물질 데이터 | 1303-96-4(Hazardous Substances Data) | ||
| 독성 | LD50 orally in rats: 5.66 g/kg (Smyth) | ||
| 기존화학 물질 | KE-03483 |
붕산나트륨10수화물 C화학적 특성, 용도, 생산
존재
천연으로는 온천의 침전물, 화산지대의 호수 침전물 등으로 산출되며, 틴카르라는 이름으로 티베트에서 산출되고, 미국 캘리포니아의 염호(鹽湖) 등에서 붕산염의 중요 자원으로 산출된다.개요
천연산을 정제하거나, 커나이트를 물에 넣어 끓인 후 재결정(再 結晶)시키거나, 코르만석 ·울렉사이트 ·브라이세아이트 등에 탄산나트륨을 직접 작용시키거나 일단 붕산을 만든 다음 중화 결정시켜서 얻는다.화학적 성질
White cryst. powder물리적 성질
White monoclinic crystal; density 1.73 g/cm3; decomposes at 75°C; soluble in water; the vapor pressure of the pure compound 1.6 torr at 20°C and that of a saturated solution 130 torr at 58°C; the pH of a 1% aqueous solution 9.24 (the pH is nearly independent of concentration); readily dissolves in alcohols.출처
Borax decahydrate occurs in nature as mineral, borax (tincal). It is one of the most common sodium borate ores. The compound has several industrial applications. The refined material is mostly used in household cleaning products. It is used to make pyrex and other borosilicate glasses. Borax is added to fertilizers in small quantities as a source of boron, as a trace nutrient for plants. High purity grade borax is used in cosmetics, toilet products and electrolytic capacitors. It also is used in fire retardants, adhesives and herbicides.용도
Sodium Tetraborate Decahydrate is used as a buffer in antigetn-retrieval procedure for bromodeoxyuridine immunolabeling with concurrent labeling of nuclear DNA and antigens damaged by hydrochloric acid pretreatment.생산 방법
Sodium borate can be prepared from minerals such as borosodium calcite, pandermite, or tinkal; these are natural sodium or calcium borates. Treatment of the mineral with sodium carbonate and sodium hydrogencarbonate yields the sodium borate decahydrate. In the USA, brine from salt lakes is also an important source of sodium borate.위험도
Toxic by inhalation.건강위험
Borates are irritants of the eyes, nose, and throat; at high concentrations ingestion of the compounds can result in gastrointestinal irritation, kidney injury, and even death from central nervous system depression or cardiovascular collapse.Pharmaceutical Applications
Sodium borate is used in pharmaceutical applications similarly to boric acid (see Boric Acid). It has been used externally as a mild astringent and as an emulsifying agent in creams. It has also been used in lozenges, mouthwashes, otic preparations (0.3% w/v), and ophthalmic solutions (0.03–1.0% w/v). Sodium borate has additionally been investigated in the prevention of crystal formation in freeze-dried solutions.Preparations of sodium borate in honey have historically been used as paints for the throat, tongue, and mouth, but such use is now inadvisable because of concerns about toxicity in such applications. Sodium borate is also used in cosmetics such as moisturizers, deodorants, and shampoos.
농업용
Solubor is a type of borate containing 20.3% boron. It is chemically a polyborate, similar to borax, and is represented as Na2B2O7?5H2O +Na2B10O16?10H2O. is a finely-ground, white product specially designed for foliar, liquid or dust applications, to correct boron deficiency.Safety Profile
Experimental poison by subcutaneous route. Moderately toxic to humans by ingestion. Moderately toxic experimentally by ingestion, intravenous, and intraperitoneal routes. Experimental reproductive effects. Mutation data reported. Ingestion of 5-10 g of borax by children can cause severe vomiting, diarrhea, shock, death. Incompatible with acids, metallic salts. When heated to decomposition it emits toxic fumes of Na2O, boron. See also BORON COMPOUNDS. Used in ant poisons, for fly control around refuse and manure piles, as a larvicide, in manufacture of glazes, enamels, cleaning compounds, and in soldering metals.Safety
Sodium borate has weak bacteriostatic and astringent properties. Historically, sodium borate has been used as a disinfectant in skin lotions and eye-, nose-, and mouthwashes. However, boric acid is easily absorbed via mucous membranes and damaged skin, and severe toxicity has been observed, especially in babies and children. Consequently, the use of sodium borate as a disinfectant is now considered somewhat obsolete and careful use is recommended. The toxic effects of sodium borate include vomiting, diarrhea, erythema, CNS depression, and kidney damage. The lethal oral intake is approximately 20 g in adults and 5 g in children.LD50 (guinea pig, oral): 5.33 g/kg
LD50 (mouse, IP): 2.711 g/kg
LD50 (mouse, IV): 1.320 g/kg
LD50 (mouse, oral): 2.0 g/kg
LD50 (rat, oral): 2.66 g/kg
잠재적 노출
Borax is used as a soldering flux, preservative against wood fungus; and as an antiseptic. Used in ant poisons, for fly control around refuse and manure piles, as a larvicide. It is used in the manufacture of enamels and glazes, fiberglass insulation; sodium perborate bleach; in tanning, cleaning compounds; for fireproofing fabrics and wood; and in artificial aging of wood.Carcinogenicity
Sodium borate tested negatively in the Ames bioassay but was found to be cytotoxic to cultured human fibroblasts.저장
Sodium borate should be stored in a well-closed container in a cool, dry, place.운송 방법
UN3077 Environmentally hazardous substances, solid, n.o.s., Hazard class: 9; Labels: 9—Miscellaneous hazardous material, Technical Name Required.Purification Methods
Crystallise the borate from water (3.3mL/g), keeping below 55o to avoid formation of the pentahydrate. Filter it off at the pump, wash it with water and equilibrate it for several days in a desiccator containing an aqueous solution saturated with respect to sucrose and NaCl. Borax can be prepared more quickly (but its water content is somewhat variable) by washing the recrystallised material at the pump with water, followed by 95% EtOH, then Et2O, and dried in air at room temperature for 12-18hours on a clock glass. [Becher in Handbook of Preparative Inorganic Chemistry (Ed. Brauer) Academic Press Vol I pp 794-795 1963.]비 호환성
Dissolves in water forming a basic solution. Boron dust may form explosive mixture with air. Contact with strong oxidizers may be violent. Boron is incompatible with ammonia, bromine tetrafluoride, cesium carbide, chlorine, fluorine, interhalogens, iodic acid, lead dioxide, nitric acid, nitric oxide, nitrosyl fluoride, nitrous oxide, potassium nitrite, rubidium carbide, silver fluoride.폐기물 처리
Borax, dehydrated: The material is diluted to the recommended provisional limit (0.10 mg/L) in water. The pH is adjusted to between 6.5 and 9.1 and then the material can be discharged into sewers or natural streams.Regulatory Status
Accepted for use as a food additive in Europe. Included in the FDA Inactive Ingredients Database (otic preparations; ophthalmic solutions and suspensions). Included in nonparenteral medicines licensed in the UK, Italy, France, Germany, and Japan. Included in the Canadian List of Acceptable Non-medicinal Ingredients.붕산나트륨10수화물 준비 용품 및 원자재
원자재
준비 용품
붕산나트륨10수화물 공급 업체
글로벌( 523)공급 업체
| 공급자 | 전화 | 이메일 | 국가 | 제품 수 | 이점 |
|---|---|---|---|---|---|
| Hebei Mojin Biotechnology Co., Ltd | +8613288715578 |
sales@hbmojin.com | China | 12456 | 58 |
| Henan Bao Enluo International TradeCo.,LTD | +86-17331933971 +86-17331933971 |
deasea125996@gmail.com | China | 2503 | 58 |
| Hebei Xinsheng New Material Technology Co., LTD. | +86-16632316109 |
xinshengkeji@xsmaterial.com | China | 1100 | 58 |
| Hebei Anlijie Biotechnology Co., Ltd | +8619031013551 |
ably@aljbio.com | China | 177 | 58 |
| Hebei Saisier Technology Co., LTD | +86-18400010335 +86-13102810335 |
admin@hbsaisier.cn | China | 747 | 58 |
| Shaanxi TNJONE Pharmaceutical Co., Ltd | +8618740459177 |
sarah@tnjone.com | China | 893 | 58 |
| Henan Tianfu Chemical Co.,Ltd. | +86-0371-55170693 +86-19937530512 |
info@tianfuchem.com | China | 21691 | 55 |
| Shanghai Time Chemicals CO., Ltd. | +86-021-57951555 +8617317452075 |
jack.li@time-chemicals.com | China | 1807 | 55 |
| Hefei TNJ Chemical Industry Co.,Ltd. | +86-0551-65418679 +86-18949832763 |
info@tnjchem.com | China | 2989 | 55 |
| Shanghai Zheyan Biotech Co., Ltd. | 18017610038 |
zheyansh@163.com | CHINA | 3620 | 58 |