N-뷰틸 메타크릴레이트 C화학적 특성, 용도, 생산
개요
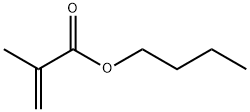
Butyl methacrylate (BMA) is the organic compound with the formula C4H9O2CC(CH3)=CH2. A colorless liquid, it is a common monomer for the preparation of methacrylate polymers. It is typically polymerized under free-radical conditions.
Butyl methacrylate is a versatile, plasticizing methacrylate monomer that acts as an important building block in many copolymer compositions. Serving as a base for acrylic resins, BMA provides exceptional weather resistance, high gloss, color retention, and durability. BMA contributes more intermediate glass transition and hardness values, with a glass transition temperature (Tg) of 20 °C.
화학적 성질
Butyl methacrylate is a flammable, colorless
liquid with a mild odor.
용도
n-Butyl methacrylate is used as a monomer for resins,
solvent coatings, adhesives, oil additives, emulsions for
textiles, leather, and paper finishing and in the manufacture
of contact lenses . It is also used in dental resins,
oil dispersible pesticides, and acrylic surface coatings
and as copolymers, for example, in paraffin embedding
media .
생산 방법
n-Butyl methacrylate is produced by the reaction of
methacrylic acid or methyl methacrylate with butanol .
일반 설명
A clear colorless liquid. Flash point 130°F. Less dense (7.5 lb / gal) than water and insoluble in water. Hence floats on water. Vapors heavier than air. Used to make resins adhesives, and oil additives.
공기와 물의 반응
Flammable. Sensitive to moisture. Insoluble in water.
반응 프로필
Butyl methacrylate reacts exothermically with acids. Reacts with oxidizing agents. Strong oxidizing acids may cause a reaction that is sufficiently exothermic to ignite the reaction products. Heat is generated with caustic solutions. Generates flammable hydrogen with alkali metals and hydrides. Polymerizes easily .
위험도
Toxic by ingestion. Moderate fire risk.
건강위험
Inhalation may cause nausea because of offensive odor. Contact with liquid causes irritation of eyes and mild irritation of skin. Ingestion causes irritation of mouth and stomach.
화재위험
Behavior in Fire: Containers may explode due to polymerization.
화학 반응
Reactivity with Water No reaction; Reactivity with Common Materials: No reactions; Stability During Transport: Stable; Neutralizing Agents for Acids and Caustics: Not pertinent; Polymerization: May occur upon exposure to heat; Inhibitor of Polymerization: 9-15 ppm monomethyl ether of hydroquinone; 90-120 ppm hydroquinone.
Safety Profile
Moderately toxic by
intraperitoneal route. Mddly toxic by
ingestion, inhalation, and skin contact. An
experimental teratogen. Experimental
reproductive effects. A skin irritant.
Flammable liquid when exposed to heat or
flame. Explosive in the form of vapor when exposed to heat or flame. Violent
polymerization can be caused by heat,
moisture, oxidizers. To fight fire, use foam,
dry chemical, CO2. When heated to
decomposition it emits acrid smoke and
irritating fumes.
잠재적 노출
Forms an explosive mixture with air.
Unless inhibitor is maintained at the proper level, oxidizers,
heat, ultraviolet light, contamination, or moisture may
cause polymerization. May accumulate static electrical
charges and cause ignition of its vapors.
운송 방법
UN2227 Butyl methacrylate, stabilized, Hazard
Class: 3; Labels: 3—Flammable liquid.
Purification Methods
Purify as for butyl acrylate. [Beilstein 2 IV 1525.]
비 호환성
Forms an explosive mixture with air.
Unless inhibitor is maintained at the proper level, oxidizers,
heat, ultraviolet light, contamination, or moisture may
cause polymerization. May accumulate static electrical
charges and cause ignition of its vapors
폐기물 처리
Dissolve or mix the material
with a combustible solvent and burn in a chemical incinerator equipped with an afterburner and scrubber. All federal,
state, and local environmental regulations must be
observed.
N-뷰틸 메타크릴레이트 준비 용품 및 원자재
원자재
준비 용품