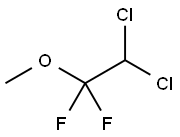
메톡시플루란
|
|
메톡시플루란 속성
- 녹는점
- -36°C
- 끓는 점
- 103 °C
- 밀도
- 1.4262
- 굴절률
- 1.386
- 인화점
- 37°C
- 저장 조건
- -70°C
- 용해도
- 클로로포름(소량으로 용해)
- 물리적 상태
- 액체
- 색상
- 투명한
- 수용성
- 알코올, 아세톤, 클로로포름, 에테르, 고정 오일 및 벤젠과 혼합됩니다. 물과 섞이지 않습니다.
- Merck
- 14,5994
- BRN
- 1737766
- 노출 한도
- No exposure limit is set. Based on comparison with related compounds, a TLV-TWA of 675 mg/m3 (100 ppm) is recommended.
- 안정성
- 안정
- CAS 데이터베이스
- 76-38-0(CAS DataBase Reference)
안전
- 위험 및 안전 성명
- 위험 및 사전주의 사항 (GHS)
| 위험품 표기 | Xi | ||
|---|---|---|---|
| 위험 카페고리 넘버 | 10 | ||
| 안전지침서 | 23-24/25 | ||
| 유엔번호(UN No.) | 3271 | ||
| RTECS 번호 | KN7820000 | ||
| 위험 참고 사항 | Irritant | ||
| 위험 등급 | 3 | ||
| 포장분류 | III | ||
| HS 번호 | 2909191800 | ||
| 유해 물질 데이터 | 76-38-0(Hazardous Substances Data) | ||
| 독성 | One of the most potent of the inhalational anesthetics, having a very high blood-gas partition coefficient and low vapor pressure at room temperature. Methoxyflurane is metabolized to a great extent (about 50-70%) in the liver and, as a consequence, there may be release of high concentrations of fluoride, sufficient to exceed the threshold for renal damage. Its use for sustained anesthesia is limited because of this renal toxicity and was discontinued around 1980. |
메톡시플루란 C화학적 특성, 용도, 생산
화학적 성질
colourless liquid용도
Methoxyflurane is a very potent and highly lipid soluble anesthetic agent. Methoxyflurane causes deep sedation and it has been used as a patient controlled analgesic for painful procedures in children. Methoxyflurane is a significant respiratory depressant.정의
ChEBI: An ether in which the two groups attached to the central oxygen atom are methyl and 2,2-dichloro-1,1-difluoroethyl.Biological Functions
Methoxyflurane (Penthrane) is the most potent inhalational agent available, but its high solubility in tissues limits its use as an induction anesthetic. Its pharmacological properties are similar to those of halothane with some notable exceptions. For example, since methoxyflurane does not depress cardiovascular reflexes, its direct myocardial depressant effect is partially offset by reflex tachycardia, so arterial blood pressure is better maintained. Also, the oxidative metabolism of methoxyflurane results in the production of oxalic acid and fluoride concentrations that approach the threshold of causing renal tubular dysfunction. Concern for nephrotoxicity has greatly restricted the use of methoxyflurane.일반 설명
Methoxyflurane is a volatile liquid (bp=105°C) with a highblood:gas partition coefficient and thus a slow induction andprolonged recovery. Approximately 75% of the drug undergoesmetabolism yielding dichloroacetate, difluoromethoxyacetate,oxalate, and fluoride ions. The intrarenal inorganicfluoride concentration, as a result of renal defluorination, maybe responsible for the nephrotoxicity seen with methoxyflurane.Both the concentration of F- generated and the durationfor which it remained elevated were factors in the developmentof methoxyflurane nephrotoxicity. Methoxyfluranewas removed from the U.S. market in 2000 because of saferalternatives. Both isoflurane and enflurane produce less fluorideion upon metabolism than methoxyflurane.공기와 물의 반응
Insoluble in water.반응 프로필
2,2-DICHLORO-1,1-DIFLUOROETHYL METHYL ETHER may be sensitive to prolonged exposure to light.건강위험
Methoxyflurane exhibited low to very lowacute toxicity via inhalation, slightly lowerthan that of ethrane. Oral toxicity was low tomoderate depending on the species. Inhala tion of its vapors at 1.5–2% by volumeconcentrations in air can cause anesthesia inhumans. The toxic symptoms are similar tothose of ethrane, and the target organs areprimarily the central nervous system, kidney,and liver. At subanesthetic concentrations of0.3–0.5% by volume in air, its exposure tohumans for 1 hour resulted in the onset oflow toxicity. The sites of biological effectswere in the kidney.LC50 value, inhalation (mice): 17,500 ppm/2 hr
LD50 value, oral (mammals): 3600 mg/kg
The liquid may be an irritant to theeyes. The teratomeric properties of this com pound were observed in rats and mice. Thesymptoms were embryo deaths and develop mental abnormalities in the urogenital andmusculoskeletal systems.
No carcinogenic actions in animals orhumans have been reported. The histidinereversion–Ames test for mutagenicity wasinconclusive.
화재위험
2,2-DICHLORO-1,1-DIFLUOROETHYL METHYL ETHER is combustible.Clinical Use
Methoxyflurane is seldom used because of its propensity to cause renal toxicity. It is the most potent agent, and it has the highest solubility in blood. Induction and recovery would be expected to be slow. Chemically, it is rather unstable, and as much as 50% of an administered dose can be metabolized. Toxic metabolites significantly limit its utility as a general anesthetic.메톡시플루란 준비 용품 및 원자재
원자재
준비 용품
메톡시플루란 공급 업체
글로벌( 87)공급 업체
| 공급자 | 전화 | 이메일 | 국가 | 제품 수 | 이점 |
|---|---|---|---|---|---|
| Hubei Jusheng Technology Co.,Ltd. | 18871490254 |
linda@hubeijusheng.com | CHINA | 28180 | 58 |
| Career Henan Chemica Co | +86-0371-86658258 15093356674; |
laboratory@coreychem.com | China | 30255 | 58 |
| Hefei TNJ Chemical Industry Co.,Ltd. | 0551-65418671 |
sales@tnjchem.com | China | 34572 | 58 |
| ANHUI WITOP BIOTECH CO., LTD | +8615255079626 |
eric@witopchemical.com | China | 23556 | 58 |
| Dideu Industries Group Limited | +86-29-89586680 +86-15129568250 |
1026@dideu.com | China | 29220 | 58 |
| CD Chemical Group Limited | +8615986615575 |
info@codchem.com | China | 20356 | 58 |
| Shaanxi Didu New Materials Co. Ltd | +86-89586680 +86-13289823923 |
1026@dideu.com | China | 9116 | 58 |
| ZHEJIANG JIUZHOU CHEM CO., LTD | +86-0576225566889 +86-13454675544 |
admin@jiuzhou-chem.com;jamie@jiuzhou-chem.com;alice@jiuzhou-chem.com | China | 20000 | 58 |
| Shanghai Acmec Biochemical Technology Co., Ltd. | +undefined18621343501 |
product@acmec-e.com | China | 33349 | 58 |
| Shanghai Sinch Parmaceuticals Tech. Co. Ltd. | +86-21-54098501 |
sales@sinch.com.cn | China | 11605 | 64 |
메톡시플루란 관련 검색:
1,2,3-프로페인트라이올 트라이나이트레이트 질산 칼륨 할로탄 메톡시플루란
Acetic acid
3-METHOXYPROPIONIC ACID
Pyrrole
OLIGOMYCIN
Chloral hydrate
Flumazenil
Zoledronic acid
Sildenafil
5-(1-Hydroxy-2-tert-butylamino-ethyl)benzene-1,3-diol
Dichloroethane
1,1-DIFLUORODIMETHYL ETHER
METHOXY-D3-FLURANE
1,1-DICHLORO-2,2-DIFLUOROETHANE
chloromethyl 2,2-dichloro-1,1,2-trifluoroethyl ether