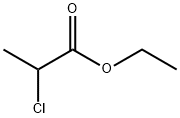
클로로(2-)프로피온산에틸에스테르 C화학적 특성, 용도, 생산
화학적 성질
clear colorless liquid
용도
Ethyl 2-chloropropionate was used as initiator during the synthesis of poly(2-(diethylamino)ethyl methacrylate) and poly(
N-isopropylacrylamide) via atom transfer radical polymerization. It was also used in the synthesis of end-functionalized poly(
N-isopropylacrylamide) with pyrenyl group.
일반 설명
A clear colorless liquid with a pungent odor. Flash point of 100°F. Denser than water and insoluble in water. Vapors are heavier than air.
공기와 물의 반응
Highly flammable. Insoluble in water.
반응 프로필
Ethyl 2-chloropropionate is a halogenated ester. Esters react with acids to liberate heat along with alcohols and acids. Strong oxidizing acids may cause a vigorous reaction that is sufficiently exothermic to ignite the reaction products. Heat is also generated by the interaction of esters with caustic solutions. Flammable hydrogen is generated by mixing esters with alkali metals and hydrides.
건강위험
May cause toxic effects if inhaled or absorbed through skin. Inhalation or contact with material may irritate or burn skin and eyes. Fire will produce irritating, corrosive and/or toxic gases. Vapors may cause dizziness or suffocation. Runoff from fire control or dilution water may cause pollution.
화재위험
HIGHLY FLAMMABLE: Will be easily ignited by heat, sparks or flames. Vapors may form explosive mixtures with air. Vapors may travel to source of ignition and flash back. Most vapors are heavier than air. They will spread along ground and collect in low or confined areas (sewers, basements, tanks). Vapor explosion hazard indoors, outdoors or in sewers. Runoff to sewer may create fire or explosion hazard. Containers may explode when heated. Many liquids are lighter than water.
잠재적 노출
This material is used in organic
synthesis.
운송 방법
UN2935 Ethyl 2-chloropropionate, Hazard
Class: 3; Labels: 3-Flammable liquid.
비 호환성
Flammable liquid; forms explosive mixture with air. Incompatible with oxidizers (chlorates,
nitrates, peroxides, permanganates, perchlorates, chlorine,
bromine, fluorine, etc.); contact may cause fires or explosions. Keep away from alkaline materials, strong bases,
strong acids, oxoacids, epoxides, and reducing agents.
Esters are generally incompatible with nitrates. Moisture
may cause hydrolysis or other forms of decomposition.
Esters react with acids releasing heat in addition to alcohols
and acids. Contact with caustic solutions generate heat.
Contact with esters with alkali metals and hydrides releases
flammable hydrogen.
클로로(2-)프로피온산에틸에스테르 준비 용품 및 원자재
원자재
준비 용품