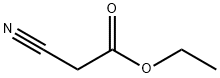
에틸 사이아노아세테이트
|
|
에틸 사이아노아세테이트 속성
- 녹는점
- -22 °C (lit.)
- 끓는 점
- 208-210 °C (lit.)
- 밀도
- 1.063 g/mL at 25 °C (lit.)
- 증기 밀도
- 3.9 (vs air)
- 증기압
- 1 mm Hg ( 67.8 °C)
- 굴절률
- n
20/D 1.418(lit.)
- 인화점
- >230 °F
- 저장 조건
- Store below +30°C.
- 용해도
- 20g/L
- 물리적 상태
- 액체
- 산도 계수 (pKa)
- 3.19±0.10(Predicted)
- 색상
- 투명한
- 수용성
- 20g/L(20℃)
- Merck
- 14,3786
- BRN
- 605871
- Dielectric constant
- 26.9(20℃)
- LogP
- -0.119-1.05 at 23-25℃ and pH6.1
- 표면장력
- 70.2mN/m at 1.034g/L and 23℃
- CAS 데이터베이스
- 105-56-6(CAS DataBase Reference)
안전
- 위험 및 안전 성명
- 위험 및 사전주의 사항 (GHS)
| 위험품 표기 | Xn,Xi | ||
|---|---|---|---|
| 위험 카페고리 넘버 | 20/21/22-36/38 | ||
| 안전지침서 | 36/37-37/39-26 | ||
| 유엔번호(UN No.) | 3276 | ||
| WGK 독일 | 1 | ||
| RTECS 번호 | AG4110000 | ||
| F 고인화성물질 | 10 | ||
| 자연 발화 온도 | 460 °C | ||
| TSCA | Yes | ||
| HS 번호 | 2926 90 70 | ||
| 유해 물질 데이터 | 105-56-6(Hazardous Substances Data) | ||
| 독성 | LD50 orally in Rabbit: > 2000 mg/kg | ||
| 기존화학 물질 | KE-09030 |
에틸 사이아노아세테이트 C화학적 특성, 용도, 생산
개요
Ethyl cyanoacetate is the ethyl ester of cyanoacetic acid. Ethyl cyanoacetate hydrolizes rapidly under neutral and alkaline conditions to cyanoacetic acid and ethanol (and so it does under most physiological and environmental conditions), while in acid pH the half life is considerably longer.Knoevenagel condensation of ethyl cyanoacetate with aldehyde is reported. Microwave enhanced Knoevenegal condensation reaction of ethyl cyanoacetate with an aldehyde, P2O5, piperidine and chlorobenzene is reported.
화학적 성질
Ethyl cyanoacetate is a colorless to straw colored liquid with a mild pleasant odor용도
Reagent used in labelled pyrimidine and purine synthesis. Ethyl cyanoacetate is an ester. It may be used in the synthesis of ethyl glyoxylate.It was used to investigate the Knoevenagel condensation reactions in microreactor using zeolite catalysts obtained by grafting amino groups onto NaX and CsNaX zeolites.제조 방법
Ethyl cyanoacetate can be prepared by the action of sodium or potassium cyanide on ethyl chloroacetate, and by the action of sodium cyanide on sodium chloroacetate, followed by esterification.일반 설명
A colorless liquid. Denser than water. Contact may irritate skin, eyes and mucous membranes. Flash point 210°F. May be toxic by ingestion. Used to make other chemicals.공기와 물의 반응
Slightly soluble in water.반응 프로필
Ethyl cyanoacetate is both a nitrile and an ester. Esters react with acids to liberate heat along with alcohols and acids. Strong oxidizing acids may cause a vigorous reaction that is sufficiently exothermic to ignite the reaction products. Heat is also generated by the interaction of esters with caustic solutions. Flammable hydrogen is generated by mixing esters with alkali metals and hydrides. Nitriles may polymerize in the presence of metals and some metal compounds. They are incompatible with acids; mixing nitriles with strong oxidizing acids can lead to extremely violent reactions. Nitriles are generally incompatible with other oxidizing agents such as peroxides and epoxides. The combination of bases and nitriles can produce hydrogen cyanide. Nitriles are hydrolyzed in both aqueous acid and base to give carboxylic acids (or salts of carboxylic acids). These reactions generate heat. Peroxides convert nitriles to amides. Nitriles can react vigorously with reducing agents. Acetonitrile and propionitrile are soluble in water, but nitriles higher than propionitrile have low aqueous solubility. They are also insoluble in aqueous acids.위험도
Toxic by ingestion and inhalation.건강위험
TOXIC; inhalation, ingestion or contact (skin, eyes) with vapors, dusts or substance may cause severe injury, burns or death. Contact with molten substance may cause severe burns to skin and eyes. Reaction with water or moist air will release toxic, corrosive or flammable gases. Reaction with water may generate much heat that will increase the concentration of fumes in the air. Fire will produce irritating, corrosive and/or toxic gases. Runoff from fire control or dilution water may be corrosive and/or toxic and cause pollution.화재위험
Combustible material: may burn but does not ignite readily. Substance will react with water (some violently) releasing flammable, toxic or corrosive gases and runoff. When heated, vapors may form explosive mixtures with air: indoors, outdoors and sewers explosion hazards. Most vapors are heavier than air. They will spread along ground and collect in low or confined areas (sewers, basements, tanks). Vapors may travel to source of ignition and flash back. Contact with metals may evolve flammable hydrogen gas. Containers may explode when heated or if contaminated with water.Safety Profile
oison by ingestion. Moderately toxic by intraperitoneal and subcutaneous routes. Combustible when exposed to heat or flame; can react with oxidzing materials. Wdl react with water or steam to produce toxic and flammable vapors. To fight fire, use CO2, dry chemical. When heated to decomposition or on contact with acid or acid fumes it emits highly toxic fumes of CN-. See also NITRILES.잠재적 노출
A nitrile used to manufacture dyes, pharmaceuticals, and other chemicals.운송 방법
UN3276 Nitriles, liquid, toxic, n.o.s., Hazard Class: 6.1; Labels: 6.1-Poisonous materials, Technical Name Required, Potential Inhalation Hazard (Special Provision 5).Purification Methods
Shake the ester several times with aqueous 10% Na2CO3, wash it well with water, dry with Na2SO4 and fractionally distil it. [Beilstein 2 IV 1889.]비 호환성
Incompatible with oxidizers (chlorates, nitrates, peroxides, permanganates, perchlorates, chlorine, bromine, fluorine, etc.); contact may cause fires or explosions. Keep away from alkaline materials, strong bases, strong acids, oxoacids, epoxides, and reducing agents. Nitriles may polymerize in the presence of metals and some metal compounds. They are incompatible with acids; mixing nitriles with strong oxidizing acids can lead to extremely violent reactions. Nitriles are generally incompatible with other oxidizing agents such as peroxides and epoxides. The combination of bases and nitriles can produce hydrogen cyanide. Nitriles are hydrolyzed in both aqueous acid and base to give carboxylic acids (or salts of carboxylic acids). These reactions generate heat. Peroxides convert nitriles to amides. Nitriles can react vigorously with reducing agents. Acetonitrile and propionitrile are soluble in water, but nitriles higher than propionitrile have low aqueous solubility. They are also insoluble in aqueous acids. Reacts with moisture, water, and steam, forming toxic fumes.폐기물 처리
Consult with environmental regulatory agencies for guidance on acceptable disposal practices. Generators of waste containing this contaminant (≥100 kg/mo) must conform with EPA regulations governing storage, transportation, treatment, and waste disposal.에틸 사이아노아세테이트 준비 용품 및 원자재
원자재
준비 용품
5-시아노-6-하이드록시-4-메톡시메틸-2-메틸피리딘
에틸2-시아노-3-메틸-2-펜테노에이트
ETHYL 2-AMINO-4,5,6,7-TETRAHYDROBENZO[B]THIOPHENE-3-CARBOXYLATE
트리암테렌
2-(4,6-디아미노-1,3,5-트리아진-2-일)아세트산
알로퓨리놀
2-Amino-4-trifluoromethylbenzonitrile
5-AMINO-4-CYANO-3-METHYL-THIOPHENE-2-CARBOXYLIC ACID ETHYL ESTER
1-ETHYL-1-METHYLSUCCINIC ACID
Disperse Yellow S-3GL
6-AMINO-2-METHYLTHIO-3-METHYLURACIL
1,2,3,5-TETRAHYDRO-8-THIA-5,7-DIAZA-CYCLOPENTA[A]INDENE-4-ONE
2-아미노-4,6-다이메틸-3-피리딘카복사미드
6-Aminouracil
2-클로로-4-메틸퀴놀린-3-탄소니트릴
ETHYL 2-AMINO-4,5-DIMETHYLTHIOPHENE-3-CARBOXYLATE
6-AMINO-2-MERCAPTO-PYRIMIDIN-4-OL
Ethyl α-cyanoacrylate instantaneous adhesive
2-아미노-6-메틸-4,5,6,7-테트라히드로-벤조[B]티오펜-3-카르복실산에틸에스테르
Ethyl 4-amino-2-(ethylthio)-5-pyrimidinecarboxylate
3-아미노-2,2-디메틸-1-프로판올
2-AMINO-5-ISOPROPYL-THIOPHENE-3-CARBOXYLIC ACID ETHYL ESTER
2-아미노-4-에틸-5-메틸-티오펜-3-카르복실산에틸에스테르
ETHYL(Z)-2-CYANO-3-ETHOXY-2-PROPENOATE
히드로염화 아밀로리드
5-AMINO-3-METHYL-1H-PYRAZOLE-4-CARBOXYLIC ACID ETHYL ESTER
6-Amino-1-methyluracil
2-FORMAMIDINO-2-PHENYLDIAZOACETAMIDE염산염
1-Cyclohexyl-1,2,3,4-tetrahydro-2,4-dioxopyrimidine-5-carbonitrile ,97%
6-HYDROXY-2,4,5-TRIAMINOPYRIMIDINE
6-CHLORO-5-CYANO-4-METHOXYMETHYL-3-NITRO-2-PICOLINE
4-Amino-6-hydroxy-2-mercaptopyrimidine monohydrate
ETHYL 5-AMINO-1-PYRIDIN-2-YL-1H-PYRAZOLE-4-CARBOXYLATE
GUANINE SULFATE
다카바진
2-AMINO-5,6-DIHYDRO-4H-CYCLOPENTA[B]THIOPHENE-3-CARBOXYLIC ACID ETHYL ESTER
6-AMINO-1-METHYL-5-NITROSOURACIL
6-AMINO-3-METHYLURACIL
Ethyl 3-ethoxy-3-iminopropionate hydrochloride
5,6-Diamino-1-methyluracil
에틸 사이아노아세테이트 공급 업체
글로벌( 482)공급 업체
| 공급자 | 전화 | 이메일 | 국가 | 제품 수 | 이점 |
|---|---|---|---|---|---|
| Hebei Chuanghai Biotechnology Co,.LTD | +86-13131129325 |
sales1@chuanghaibio.com | China | 5895 | 58 |
| Capot Chemical Co.,Ltd. | 571-85586718 +8613336195806 |
sales@capotchem.com | China | 29798 | 60 |
| Henan Tianfu Chemical Co.,Ltd. | +86-0371-55170693 +86-19937530512 |
info@tianfuchem.com | China | 21666 | 55 |
| Shanghai Time Chemicals CO., Ltd. | +86-021-57951555 +8617317452075 |
jack.li@time-chemicals.com | China | 1807 | 55 |
| ATK CHEMICAL COMPANY LIMITED | +undefined-21-51877795 |
ivan@atkchemical.com | China | 32836 | 60 |
| Hefei TNJ Chemical Industry Co.,Ltd. | +86-0551-65418679 +8618949832763 |
info@tnjchem.com | China | 2989 | 55 |
| career henan chemical co | +86-0371-86658258 +8613203830695 |
sales@coreychem.com | China | 29888 | 58 |
| Hubei Jusheng Technology Co.,Ltd. | 18871490254 |
linda@hubeijusheng.com | CHINA | 28180 | 58 |
| Jinan Finer Chemical Co., Ltd | +86-531-88989536 +86-15508631887 |
sales@finerchem.com | China | 2966 | 58 |
| Hebei Guanlang Biotechnology Co., Ltd. | +86-19930503282 |
alice@crovellbio.com | China | 8820 | 58 |