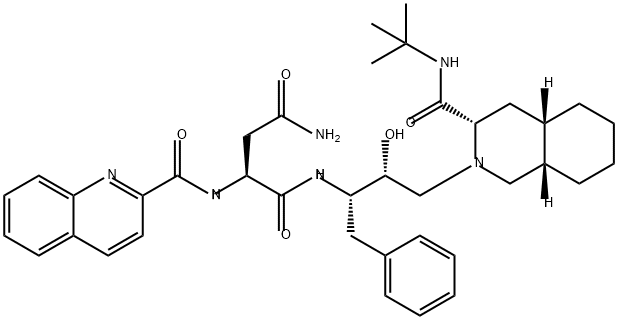
Saquinavir
- CAS No.
- 127779-20-8
- Chemical Name:
- Saquinavir
- Synonyms
- FLUNIXIN;FLUNIXINE;Sch 52852;SAQUINAVIR;SAGUINAVIR;Ro 31-8959/000;Saquinavir USP/EP/BP;SAQUINAVIR(RO 31-8959);Ro-31-8959/003:Invirase;Saquinavir Solution, 100ppm
- CBNumber:
- CB2161048
- Molecular Formula:
- C38H50N6O5
- Molecular Weight:
- 670.84
- MDL Number:
- MFCD00866925
- MOL File:
- 127779-20-8.mol
- MSDS File:
- SDS
| alpha | D20 -55.9° (c = 0.5 in methanol) |
|---|---|
| Boiling point | 1015.0±65.0 °C(Predicted) |
| Density | 1.211±0.06 g/cm3(Predicted) |
| storage temp. | Sealed in dry,Store in freezer, under -20°C |
| solubility | DMSO : 100 mg/mL (149.07 mM; Need ultrasonic) |
| form | Powder |
| pka | 11.05±0.46(Predicted) |
| Water Solubility | 35.8mg/L(25 ºC) |
| BCS Class | 4 |
| FDA 21 CFR | 556.286 |
| CAS DataBase Reference | 127779-20-8(CAS DataBase Reference) |
| FDA UNII | L3JE09KZ2F |
| ATC code | J05AE01 |
SAFETY
Risk and Safety Statements
| Symbol(GHS) |  GHS05 |
|---|---|
| Signal word | Danger |
| Hazard statements | H314 |
| Precautionary statements | P501-P264-P280-P303+P361+P353-P301+P330+P331-P363-P304+P340+P310-P305+P351+P338+P310-P405 |
Saquinavir price
| Manufacturer | Product number | Product description | CAS number | Packaging | Price | Updated | Buy |
|---|---|---|---|---|---|---|---|
| Usbiological | 289619 | Saquinavir | 127779-20-8 | 250mg | $319 | 2021-12-16 | Buy |
| ApexBio Technology | A3790 | Saquinavir | 127779-20-8 | 50mg | $359 | 2021-12-16 | Buy |
| ApexBio Technology | A3790 | Saquinavir | 127779-20-8 | 100mg | $520 | 2021-12-16 | Buy |
| American Custom Chemicals Corporation | INB0000705 | SAGUINAVIR 95.00% | 127779-20-8 | 25MG | $547.05 | 2021-12-16 | Buy |
| ApexBio Technology | A3790 | Saquinavir | 127779-20-8 | 10mg | $82 | 2021-12-16 | Buy |
Saquinavir Chemical Properties,Uses,Production
Description
Saquinavir mesylate, the first HIV protease inhibitor to reach the market, was launched in the U.S.A.. It is indicated for use in combination with approved nucleoside analogs for the treatment of advanced HIV infection. Saquinavir, a transition state analog of Phe-Pro, is a very potent and competitive inhibitor of HIV-1 and HIV-2 proteases with high specificity. Saquinavir inhibits the last stage in the replication process of HIV and prevents virion maturation in both acute and chronically infected cells. Combination of saquinavir with the nucleoside analogs such as zidovudine (AZT) or/and zalcitabine which inhibit the enzyme reverse transcriptase and target at an earlier stage in the HIV replication process, shows a greater than additive effect in increase in CD4 cell counts and reduction in viral load, with the combination delaying the onset of resistance to either drug alone. Saquinavir is well tolerated alone and in combination with A n .
Originator
Roche (Switzerland)
Uses
Antiviral (HIV protease inhibitor).
Definition
ChEBI: An aspartic acid derivative obtained by formal condensation of the primary amino group of (2S,3R)-4-[(3S,4aS,8aS)-3-(tert-butylcarbamoyl)octahydroisoquinolin- (1H)-yl]-3-hydroxy-1-phenylbutan-2-ylamine with the carboxy group of N2(-quinolin-2-ylcarbonyl)-L-asparagine. An inhibitor of HIV-1 protease.
Indications
Saquinavir is a potent inhibitor of HIV-1 and HIV-2 protease. Fortovase, a soft gel preparation of saquinavir, has largely replaced saquinavir mesylate capsules (Invirase) because it has improved bioavailability. Saquinavir is usually well tolerated and most frequently produces mild gastrointestinal side effects.
brand name
Fortovase (Roche).
Acquired resistance
Resistance is associated with an amino acid substitution at position 48 in the HIV protease (G48V). An L90M mutation also confers resistance, as it does for most protease inhibitors. Saquinavir-resistant isolates from patients on long-term therapy often show cross-resistance to other protease inhibitors.
General Description
Saquinavir (Invirase) is well tolerated following oral administration.Absorption of saquinavir is poor but is increasedwith a fatty meal. The drug does not distribute intothe CSF, and it is approximately 98% bound to plasma proteins.Saquinavir is extensively metabolized by the firstpasseffect. Bioavailability is 4% from a hard capsule and12% to 15% from a soft capsule. Saquinavir lowers p24antigen levels in HIV-infected patients, elevates CD4+counts, and exerts a synergistic antiviral effect when combinedwith RT inhibitors such as AZT and ddC.Although HIV-1 resistance to saquinavir and other HIVPIs occurs in vivo, it is believed to be less stringent andless frequent than resistance to the RT inhibitors.Nevertheless,cross-resistance between different HIV PIsappears to be common and additive,suggesting thatusing combinations of inhibitors from this class would notconstitute rational prescribing. The drug should be used incombination with at least two other antiretroviral drugs tominimize resistance. Dosage forms are Invirase (hard capsule)and Fortovase (soft capsule).
Pharmaceutical Applications
A peptidomimetic protease inhibitor formulated as the mesylate for oral use.
Pharmacokinetics
Oral absorption: c. 4%
Cmax 1200 mg thrice daily: c. 1–2.2 mg/L
Cmin 1200 mg thrice daily: c. 0.1–0.22 mg/L
Plasma half-life: c. 7–12 h
Volume of distribution: c. 700 L
Plasma protein binding: c. 98%
Absorption and distribution
It is poorly absorbed and penetrates poorly into the CNS. The semen:plasma ratio is 0.04. It is not known if it is distributed into human breast milk.
Metabolism and excretion
It is metabolized via CYP3A4, principally to mono- and dihydroxylated derivatives. Around 88% of the dose is excreted in feces and 1% in urine. Caution should be exercised in severe renal impairment and moderate hepatic impairment; use in decompensated hepatic impairment is contraindicated.
Clinical Use
Treatment of HIV infection (in combination with other antiretroviral drugs)
Side effects
The most frequently reported adverse effects include abdominal discomfort, diarrhea and nausea. Ritonavir-boosted saquinavir is associated with a dyslipidemic profile characteristic of those treated with a boosted protease inhibitor requiring 200 mg of the ritonavir ‘booster’.
Drug interactions
Potentially hazardous interactions with other drugs
Analgesics: increased risk of ventricular arrhythmias
with alfentanil, fentanyl and methadone - avoid.
Anti-arrhythmics: increased risk of ventricular
arrhythmias with amiodarone, disopyramide,
dronedarone, flecainide, lidocaine or propafenone -
avoid.
Antibacterials: increased risk of ventricular
arrhythmias with clarithromycin, dapsone,
erythromycin or moxifloxacin - avoid; increased
risk of ventricular arrhythmias with delamanid;
concentration of rifabutin increased; rifampicin and
rifabutin can reduce saquinavir levels by 80% and
40% respectively (metabolism accelerated); increased
hepatoxicity with rifampicin - avoid; concentration
of both drugs increased with fusidic acid.
Anticoagulants: avoid with apixaban and
rivaroxaban.
Antidepressants: increased risk of ventricular
arrhythmias with trazodone or tricyclics - avoid;
concentration reduced by St John’s wort - avoid.
Antiepileptics: carbamazepine, phenobarbital,
and phenytoin and possibly primidone can reduce
saquinavir levels.
Antifungals: concentration increased by ketoconazole
- avoid.
Antihistamines: increased risk of ventricular
arrhythmias with mizolastine - avoid.
Antimalarials: avoid with piperaquine with
artenimol; use artemether/lumefantrine with
caution; increased risk of ventricular arrhythmias
with quinine - avoid.
Antipsychotics: increased risk of ventricular
arrhythmias with clozapine, haloperidol or
phenothiazines - avoid; possibly increased risk
of ventricular arrhythmias with pimozide and
quetiapine - avoid; possibly inhibits aripiprazole
metabolism - reduce aripiprazole dose; possibly
increases lurasidone concentration - avoid.
Antivirals: tipranavir and efavirenz can reduce
saquinavir levels; increased risk of ventricular
arrhythmias with atazanavir or lopinavir - avoid;
concentration increased by indinavir and ritonavir;
reduced darunavir concentration; concentration
of maraviroc increased, consider reducing dose of
maraviroc.
Anxiolytics and hypnotics: midazolam concentration
possibly increased (prolonged sedation) - avoid with
oral midazolam.
Beta-blockers: increased risk of ventricular
arrhythmias with sotalol - avoid.
Ciclosporin: concentration of both drugs increased.
Cytotoxics: possibly increases concentration of
axitinib, ibrutinib and panobinostat, reduce dose
of axitinib, ibrutinib and panobinostat; possibly
increases bosutinib, cabazitaxel, ceritinib and
docetaxel concentration - avoid or consider reducing
dose; possibly increases concentration of crizotinib
and everolimus - avoid; avoid with lapatinib,
olaparib and pazopanib; reduce dose of ruxolitinib.
Dapoxetine: increased risk of toxicity - avoid.
Domperidone: possibly increases risk of ventricular
arrhythmias - avoid.
Ergot alkaloids: risk of ergotism - avoid.
Guanfacine: concentration possibly increased - halve
guanfacine dose.
Lipid-lowering drugs: increased risk of myopathy
with rosuvastatin and simvastatin - avoid; possibly
increased myopathy with atorvastatin; avoid with
lomitapide.
Naloxegol: possibly increases naloxegol concentration
- avoid.
Orlistat: absorption possibly reduced by orlistat.
Pentamidine: increased risk of ventricular
arrhythmias - avoid.
Ranolazine: possibly increases ranolazine
concentration - avoid.
Metabolism
Saquinavir is absorbed to a limited extent (about 30%)
after oral doses of the mesilate and undergoes extensive
first-pass hepatic metabolism via cytochrome P450
isoenzyme, CYP3A4 to form a range of mono- and
di-hydroxylated inactive compounds.
It is excreted mainly in the faeces.
Saquinavir Preparation Products And Raw materials
Raw materials
1of2
Preparation Products
| Supplier | Tel | Country | ProdList | Advantage | |
|---|---|---|---|---|---|
| CONIER CHEM AND PHARMA LIMITED | +8618523575427 | sales@conier.com | China | 49390 | 58 |
| career henan chemical co | +86-0371-86658258 15093356674; | factory@coreychem.com | China | 29826 | 58 |
| Shaanxi Dideu Medichem Co. Ltd | 18192627656 | 1012@dideu.com | China | 3422 | 58 |
| HANGZHOU CLAP TECHNOLOGY CO.,LTD | 86-571-88216897,88216896 13588875226 | sales@hzclap.com | CHINA | 6313 | 58 |
| Dideu Industries Group Limited | +86-29-89586680 +86-15129568250 | 1026@dideu.com | China | 29220 | 58 |
| AFINE CHEMICALS LIMITED | 0571-85134551 | info@afinechem.com | CHINA | 15377 | 58 |
| Baoji Guokang Bio-Technology Co., Ltd. | 0917-3909592 13892490616 | gksales1@gk-bio.com | China | 9339 | 58 |
| XI'AN TIANGUANGYUAN BIOTECH CO., LTD. | +86-029-86333380 18829239519 | sales06@tgybio.com | China | 959 | 58 |
| Nextpeptide Inc | +86-0571-81612335 +8613336028439 | sales@nextpeptide.com | China | 19915 | 58 |
| InvivoChem | +1-708-310-1919 +1-13798911105 | sales@invivochem.cn | United States | 6393 | 58 |
Related articles
- Uses and toxicity of Saquinavir
- Saquinavir, a synthetic peptidomimetic analog that inhibits HIV protease, was developed using computer-led rational design tec....
- Apr 1,2022
View Lastest Price from Saquinavir manufacturers
| Image | Update time | Product | Price | Min. Order | Purity | Supply Ability | Manufacturer | |
|---|---|---|---|---|---|---|---|---|
 |
2021-07-16 | Saquinavir USP/EP/BP
127779-20-8
|
US $1.10 / g | 1g | 99.9% | 100 Tons Min | Dideu Industries Group Limited | |
 |
2021-07-13 | Saquinavir
127779-20-8
|
US $15.00-10.00 / KG | 1KG | 99%+ HPLC | Monthly supply of 1 ton | Zhuozhou Wenxi import and Export Co., Ltd | |
 |
2021-07-10 | Saquinavir
127779-20-8
|
US $15.00-10.00 / KG | 1KG | 99%+ HPLC | Monthly supply of 1 ton | Zhuozhou Wenxi import and Export Co., Ltd |
-

- Saquinavir USP/EP/BP
127779-20-8
- US $1.10 / g
- 99.9%
- Dideu Industries Group Limited
-

- Saquinavir
127779-20-8
- US $15.00-10.00 / KG
- 99%+ HPLC
- Zhuozhou Wenxi import and Export Co., Ltd
-

- Saquinavir
127779-20-8
- US $15.00-10.00 / KG
- 99%+ HPLC
- Zhuozhou Wenxi import and Export Co., Ltd