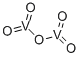오산화 바나듐
|
|
오산화 바나듐 속성
- 녹는점
- 690 °C
- 끓는 점
- 3380 °C(lit.)
- 밀도
- 3.35 g/mL at 25 °C(lit.)
- 증기압
- 8 mm Hg ( 20 °C)
- 인화점
- 1750°C
- 저장 조건
- Poison room
- 용해도
- H2O: 용해성
- 물리적 상태
- turnings
- 색상
- 오렌지 레드
- 색상
- 오렌지색
- Specific Gravity
- 3.357
- 냄새
- 100.00%. 냄새 없는
- 수소이온지수(pH)
- 4 (50g/l, H2O, 20℃)(slurry)
- 수용성
- 1g/125mL
- Merck
- 14,9921
- 노출 한도
- OSHA: Ceiling 0.5 mg/m3; Ceiling 0.1 mg/m3
NIOSH: TWA 1 mg/m3; STEL 3 mg/m3; Ceiling 0.05 mg/m3
- 안정성
- 안정적인. 염소, 염소산염, 산, 알칼리 금속, 인터할로겐과 호환되지 않습니다.
- CAS 데이터베이스
- 1314-62-1(CAS DataBase Reference)
- IARC
- 2B (Vol. 86) 2006
안전
- 위험 및 안전 성명
- 위험 및 사전주의 사항 (GHS)
| 위험품 표기 | Xi,N,T,C,T+ | ||
|---|---|---|---|
| 위험 카페고리 넘버 | 36/38-68-63-51/53-48/23-37-20/22-41-48/20-34-40-26/27/28 | ||
| 안전지침서 | 26-61-45-38-36/37-36-36/37/39-23 | ||
| 유엔번호(UN No.) | UN 3289 6.1/PG 2 | ||
| WGK 독일 | 3 | ||
| RTECS 번호 | YW1355000 | ||
| F 고인화성물질 | 10 | ||
| TSCA | Yes | ||
| 위험 등급 | 8 | ||
| 포장분류 | III | ||
| HS 번호 | 28253010 | ||
| 유해 물질 데이터 | 1314-62-1(Hazardous Substances Data) | ||
| 독성 | LD50 orally in Rabbit: 400 - 500 mg/kg LD50 dermal Rat > 2000 mg/kg | ||
| IDLA | 35 mg V/m3 | ||
| 기존화학 물질 | KE-12750 |
오산화 바나듐 C화학적 특성, 용도, 생산
개요
Vanadium pentoxide is a yellow to red colour solid and is odourless. Vanadium pentoxide dust is the particulate form of a non-combustible, odourless, yellow-orange or dark grey crystalline solid.On decomposition by heating, vanadium pentoxide produces toxic fumes. Vanadium is widely distributed in the Earth’s crust in a wide range of minerals and in fossil fuels. Vanadium pentoxide, the major commercial product of vanadium, is mainly used in the production of alloys with iron and aluminium. It is also used as an oxidation catalyst in the chemical industry and in a variety of minor applications.
화학적 성질
A yellow to rust-brown or orange crystals or powder. Slightly soluble in water and denser than water. Contact may cause severe irritation to skin, eyes, and mucous membranes. May be toxic by ingestion, inhalation and skin absorption.용도
Vanadium(V) oxide is used in Vacuum deposition, catalysts for conversion of toluene to benzonitrile or propylene to acrylonitrile, as a detector material in bolometers and microbolometer arrays for thermal imaging.정의
vanadium pentoxide: A crystalline compound,V2O5, used extensively as a catalyst inindustrial gas-phase oxidationprocesses.반응 프로필
Vanadium pentoxide is acidic in many reactions. Hence, soluble in bases. [Kirk-Othmer]. Can react with ClF3, Li, peroxyformic acid and (Ca+S+H2O). Also reacts with strong acids.위험도
The compound is toxic by ingestion, inhalation, and contact. Inhalation can cause asthma, cough, dyspnea, and bronchial constriction. Ingestion can cause gastrointestinal tract disturbances. Other toxic symptoms are skin pallor, greenish-black tongue, and papular skin rash (Lewis, R.J. (Sr) 1996. Sax’s Dangerous Properties of Industrial Materials, 9thed. New York: Van Nostrand Reinhold).The oral LD50 for V2O5 dust in rats is 10 mg/kg and the inhalation LCLO in rats is 70 mg/m3/2hr.