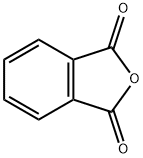
무수프탈산 C화학적 특성, 용도, 생산
용도
프탈산을 가열하여 탈수시키면 프탈산무수물을 얻을 수 있으나, 공업적으로는 오산화바나듐 V2O5을 촉매로 써서 나프탈렌 또는 오쏘자일렌을 기체상에서 산화시켜 제조한다.
용도
페놀프탈레인, 플루오레세인, 에오신, 프탈이미드, 안트라닐산, 기타 색소, 염색 중간체, 향료 등의 합성 원료, 최근 합성 수지 공업에서 알키드 수지, 에스테르계 가소제 등의 제조에 다량 이용되며, 살충제 제조에도 이용된다. 칼륨염으로 변화한 후 이성질화하여 테레프탈산으로 한다.
용도
플라스틱의 가소제로서 쓰이고, 또한 알키드수지·색소(페놀프탈레인)·염료 등의 제조원료로 사용된다.
용도
무수프탈산은 가소제, 불포화폴리에스테르수지, 페인트 등의 주원료로 사용되고 있습니다.
개요
Phthalic anhydride is the organic compound with the formula C6H4(CO)2O. It is the anhydride of phthalic acid. This colourless solid is an important industrial chemical, especially for the large-scale production of plasticizers for plastics.

Phthalic anhydride is an important chemical intermediate in the plastics industry from which are derived numerous phthalate esters that function as plasticizers in synthetic resins. Phthalic anhydride itself is used as a monomer for synthetic resins such as glyptal, the alkyd resins, and the polyester resins.
Phthalic anhydride is also used as a precursor of anthraquinone, phthalein, rhodamine, phthalocyanine, fluorescein, and xanthene dyes.
Phthalic anhydride is used in the synthesis of primary amines, the agricultural fungicide phaltan, and thalidomide. Other reactions with phthalic anhydride yield phenolphthalein, benzoic acid, phthalylsulfathiazole (an intestinal antimicrobial agent), and orthophthalic acid.
화학적 성질
Phthalic Anhydride is moderately flammable, white solid (flake) or a clear, colorless, mobile liquid (molten) Characteristic, acrid, choking odor. It is very slightly soluble in H2O, soluble in alcohol, and slightly soluble in ether.
물리적 성질
Colorless to pale cream crystals with a characteristic, choking odor. Moisture sensitive. Odor
threshold concentration is 53 ppb (quoted, Amoore and Hautala, 1983).
정의
ChEBI: Phthalic anhydride is the cyclic dicarboxylic anhydride that is the anhydride of phthalic acid. It has a role as an allergen. It is a cyclic dicarboxylic anhydride and a member of 2-benzofurans.
제조 방법
The most important modifying component used in the
manufacture of linear unsaturated polyesters is phthalic anhydride. The
anhydride is generally obtained by the oxidation of o-xylene:
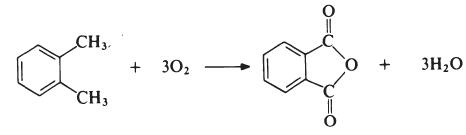
The reaction is carried out in the vapour phase by passing a mixture of
o-xylene and air over a catalyst such as vanadium pentoxide supported on
silica and promoted with titanium dioxide at about 400??C. The exit gases are
cooled and the phthalic anhydride is collected and purified by distillation
under reduced pressure.
일반 설명
A colorless to white lustrous solid in the form of needles with a mild distinctive odor. Moderately toxic by inhalation or ingestion and a skin irritant. Melting point 64°F Flash point 305°F. Forms a corrosive solution when mixed with water. Used in the manufacture of materials such as artificial resins.
공기와 물의 반응
Reacts, usually slowly with water to form phthalic acid and heat [Merck 11th ed. 1989]. The phthalic acid is somewhat soluble in water.
반응 프로필
Phthalic anhydride reacts exothermically with water. The reactions are sometimes slow, but can become violent when local heating accelerates their rate. Acids accelerate the reaction with water. Incompatible with acids, strong oxidizing agents, alcohols, amines, and bases. Undergoes exothmeric nitration with fuming nitric acid-sulfuric acid and may give mixtures of the potentially explosive phthaloyl nitrates or nitrites or their nitro derivatives [Chem. & Ind. 20:790. 1972]. Phthalic anhydride reacts violently with CuO at elevated temperatures [Park, Chang-Man, Richard J. Sheehan. hthalic Acids and Other Benzenepolycarboxylic Acids Kirk-Othmer Encyclopedia of Chemical Technology. John Wiley & Sons, Inc. 2005]. Mixtures of Phthalic anhydride and anhydrous CO2 explode violently if heated [eaflet No. 5, Inst. of Chem., London, 1940].
건강위험
Solid irritates skin and eyes, causing coughing and sneezing. Liquid causes severe thermal burns.
화재위험
Combustible material: may burn but does not ignite readily. Substance will react with water (some violently) releasing flammable, toxic or corrosive gases and runoff. When heated, vapors may form explosive mixtures with air: indoors, outdoors and sewers explosion hazards. Most vapors are heavier than air. They will spread along ground and collect in low or confined areas (sewers, basements, tanks). Vapors may travel to source of ignition and flash back. Contact with metals may evolve flammable hydrogen gas. Containers may explode when heated or if contaminated with water.
Pharmaceutical Applications
Phthalic anhydride reacted with cellulose acetate forms cellulose acetate phthalate (CAP), a common enteric coating excipient that has also been shown to have antiviral activity. Phthalic anhydride is a degradation product of CAP.
색상 색인 번호
Phthalic anhydride is used in the manufacture of unsaturated
polyesters and as a curing agent for epoxy resins.
When used as a pigment, it can be responsible for sensitization
in ceramic workers. Phthalic anhydride per se is
not responsible for the sensitization to the resin used in
nail varnishes phthalic anhydride/trimellitic anhydride/
glycols copolymer.
Safety Profile
Poison by ingestion.
Experimental teratogenic effects. A
corrosive eye, skin , and mucous membrane
irritant. A common air contaminant.
Combustible when exposed to heat or flame; can react with oxidzing materials.
Moderate explosion hazard in the form of
dust when exposed to flame. The
production of ths material has caused many
industrial explosions. Mixtures with copper
oxide or sodium nitrite explode when
heated. Violent reaction with nitric acid +
sulfuric acid above 80℃. To fight fire, use
CO2, dry chemical. Used in plasticizers,
polyester resins, and alkyd resins, dyes, and
drugs. See also ANHYDRIDES.
잠재적 노출
Phthalic anhydride is used in plasticizers; in the manufacture of phthaleins; benzoic acid; alkyd and polyester resins; synthetic indigo; and phthalic acid;which is used as a plasticizer for vinyl resins. To a lesser extent, it is used in the production of alizarin, dye, anthranilic acid; anthraquinone, diethyl phthalate; dimethyl phthalate; erythrosine, isophthalic acid; methylaniline, phenolphthalein, phthalamide, sulfathalidine, and terephthalic acid. It has also found uses as a pesticide intermediate.
운송 방법
UN2214 Phthalic anhydride with>.05 % maleic anhydride, Hazard class: 8; Labels: 8-Corrosive material.
Purification Methods
Distil the anhydride under reduced pressure. Purify it from the acid by extracting with hot CHCl3, filtering and evaporating. The residue is crystallised from CHCl3, CCl4 or *benzene, or sublimed. Fractionally crystallise it from its melt. Dry it under vacuum at 100o. [Saltiel J Am Chem Soc 108 2674 1986, Beilstein 17/11 V 253.]
비 호환성
Dust forms an explosive mixture with air. Phthalic anhydride reacts exothermically with water. The reactions are sometimes slow, but can become violent when local heating accelerates their rate. Acids accelerate the reaction with water. Incompatible with acids, strong oxidizing agents, alcohols, amines, and bases. Converted to phthalic acid in hot water. Incompatible with oxidizers (chlorates, nitrates, peroxides, permanganates, perchlorates, chlorine, bromine, fluorine, etc.); contact may cause fires or explosions. Keep away from alkaline materials, strong bases, strong acids, oxoacids, epoxides. caustics, ammonia, amines, water. Reacts violently with copper oxide or sodium nitrite 1 heat.
폐기물 처리
Use a licensed professional waste disposal service to dispose of this material. Dissolve or mix the material with a combustible solvent and burn in a chemical incinerator equipped with an afterburner and scrubber. All federal, state, and local environmental regulations must be observed. Consult with environmental regulatory agencies for guidance on acceptable disposal practices. Generators of waste containing this contaminant (≥100 kg/mo) must conform with EPA regulations governing storage, transportation, treatment, and waste disposal.
무수프탈산 준비 용품 및 원자재
원자재
준비 용품
프탈산디알릴모노머
수소화 칼륨 프탈산
1,2,5-OXADIAZOLE-3-CARBOXYLIC ACID
N-(Hydroxymethyl)phthalimide
N-PHTHALOYL-DL-GLUTAMIC ANHYDRIDE
C.I. 솔벤트 레드 49
Alkyd resin insulating paint
2',7'-dibromo-3',6'-dihydroxyspiro[isobenzofuran-1(3H),9'-[9H]xanthene]-3-one
DI-ISO-DECYL PHTHALATE
2-BENZOYLBENZOYL CHLORIDE
N-PHENYLPHTHALIMIDE
2-펜에틸벤조익 산
1-CHLORO-4-METHOXYPHTHALAZINE
2-Chloroanthraquinone
5-(2-Carboxybenzoyl)-2-chlorobenzenesulfonyl chloride
프탈산 디페닐
디펜틸 프탈산염
Amino resin varnish
2,3,4,5-사불화 벤조익산
디노닐 프탈산염
2-벤조일안식향산
2,3-DIHYDRO-2-PHENYL-1H-ISOINDOL-1-OXO-ISOINDOLINE
프탈산디이소노닐에스테르
2-Methyl anthraquinone
디이소뷰틸 프탈산
디시클로헥실 프탈레이트
Indobufen
디 이소옥틸 프탈레이트
2-에틸안트라퀴논
디옥틸 프탈레이트
시스-사이클로옥텐
폴펫
플루오레세인
디카프릴 프탈산
N-(2-Bromoethyl)phthalimide
2-(MORPHOLINE-4-CARBONYL)-BENZOIC ACID
3-벤질아이덴나프탈라이드
3-METHYL-1,2,5-OXADIAZOLE
C.I. 용제 오렌지 60
Didecyl phthalate