| Identification | More | [Name]
Acrylic acid | [CAS]
79-10-7 | [Synonyms]
2-PROPENOIC ACID
Acroleic acid
ACRYLIC ACID
AKOS BBS-00003787
ETHYLENECARBOXYLIC ACID
PROPENOIC ACID
RARECHEM AL BO 0141
2-Propensαure
acideacrylique
acideacrylique(french)
acidoacrilio
Acrylate
acrylicacid,[waste]
acrylicacid,glacial
acrylicacid,inhibited
Acrylsαure
ai3-15717
caswellno.009a
CH2=CHCOOH
Glacial acrylic acid | [EINECS(EC#)]
201-177-9 | [Molecular Formula]
C3H4O2 | [MDL Number]
MFCD00004367 | [Molecular Weight]
72.06 | [MOL File]
79-10-7.mol |
| Chemical Properties | Back Directory | [Appearance]
Acrylic acid is a colorless, flammable, and corrosive liquid or solid (below 13 C) with an irritating, rancid, odor. Sinks and mixes with water; irritating vapor is produced. | [Melting point ]
13 °C(lit.)
| [Boiling point ]
139 °C(lit.)
| [density ]
1.051 g/mL at 25 °C(lit.)
| [vapor density ]
2.5 (vs air)
| [vapor pressure ]
4 mm Hg ( 20 °C)
| [refractive index ]
n20/D 1.421
| [Fp ]
130 °F
| [storage temp. ]
15-25°C
| [solubility ]
1000g/l | [form ]
Liquid | [pka]
4.25(at 25℃) | [color ]
Clear | [Odor]
Acrid odor | [PH]
2.1 (72.06g/l, H2O, 20℃) | [PH Range]
1 - 2 | [Stability:]
Stability Unstable-may contain p-methoxyphenol as an inhibitor. Prone to hazardous polymerization. Combustible. Incompatible with strong oxidizing agents, strong bases, amines. Contact with oxidizers may cause fire. Light and air sensitive. Hygroscopic. | [explosive limit]
3.9-19.8%(V) | [Water Solubility ]
MISCIBLE | [Sensitive ]
Air Sensitive | [λmax]
231nm(lit.) | [Merck ]
14,130 | [BRN ]
635743 | [Henry's Law Constant]
2.7×101 mol/(m3Pa) at 25℃, Duchowicz et al. (2020) | [Exposure limits]
TLV-TWA 10 ppm (30 mg/m3) (ACGIH). | [Cosmetics Ingredients Functions]
NAIL CONDITIONING | [InChI]
1S/C3H4O2/c1-2-3(4)5/h2H,1H2,(H,4,5) | [Contact allergens]
Acrylates are esters from acrylic acid. Occupational
contact allergies from acrylates have frequently been
reported and mainly concern workers exposed to the
glues based on acrylic acid, as well as dental workers
and beauticians. | [InChIKey]
NIXOWILDQLNWCW-UHFFFAOYSA-N | [SMILES]
OC(=O)C=C | [LogP]
0.46 at 25℃ | [CAS DataBase Reference]
79-10-7(CAS DataBase Reference) | [IARC]
3 (Vol. 19, Sup 7, 71) 1999 | [NIST Chemistry Reference]
2-Propenoic acid(79-10-7) | [EPA Substance Registry System]
79-10-7(EPA Substance) | [ECETOC JACC REPORT]
Acrylic acid (79-10-7) |
| Hazard Information | Back Directory | [Chemical Properties]
colourless liquid with an acrid odour | [Uses]
An antibacterial agent. | [Uses]
Usually used in preparing monodispersed poly (N-isopropylacryamide) (PNIPAM)/AAc microgels.And also used primarily as an intermediate in the production of acrylates. | [Uses]
In the manufacture of plastics. | [General Description]
Acrylic acid is a colorless liquid with a distinctive acrid odor. Flash point 130°F. Boiling point 286°F. Freezing point 53°F. Corrosive to metals and tissue. Prolonged exposure to fire or heat can cause polymerization. If polymerization takes place in a closed container, violent rupture may occur. The inhibitor (usually hydroquinone) greatly reduces the tendency to polymerize. | [Reactivity Profile]
ACRYLIC ACID may polymerize violently especially when the frozen acid is partially thawed (freezing point 12°C or 53°F). Frozen acid should be melted at room temperature and the process should be well stirred. Do not use heat during the melting process [Kirk-Othmer, 3rd ed., Vol. 1, 1978, p. 330]. Corrodes iron and steel and polymerization may occur on contact with iron salts. The uninhibited acid polymerizes exothermically at ambient temperature and explodes if confined. The inhibitor (usually hydroquinone) greatly reduces the tendency to polymerize. Explosive polymerization can also occur with strong bases, amines, ammonia, oleum, chlorosulfonic acid, and peroxides. Mixing with 2-aminoethanol, 28% ammonium hydroxide, ethylenediamine or ethyleneimine in a closed container causes an increase in temperature and pressure. Can react violently with oxidizing reagents and strong bases [Bretherick, 5th ed., 1995, p. 419]. | [Air & Water Reactions]
Flammable. Soluble in water. The presence of water, due to different solubilities of the acid and inhibitor (partitioning one from the other), may initiate polymerization. | [Health Hazard]
May burn skin or eyes upon short contact. INHALATION: eye and nasal irritation and lacrimation. INGESTION: may cause severe damage to the gastrointestinal tract. | [Potential Exposure]
Acrylic acid is chiefly used in manufacture of plastics, acrylates, polyacrylic acids, polymer, and resins; as a monomer in the manufacture of acrylic resins and plastic products, leather treatment, and paper coatings. Also, it is used as a tackifier and flocculant. | [First aid]
If this chemical gets into the eyes, remove any contact lenses at once and irrigate immediately for at least 15 minutes, occasionally lifting upper and lower lids. Seek medical attention immediately. If this chemical contacts the skin, remove contaminated clothing and wash immediately with soap and water. Seek medical attention immediately. If this chemical has been inhaled, remove from exposure, begin rescue breathing (using universal precautions, including resuscitation mask) if breathing has stopped and CPR if heart action has stopped. Transfer promptly to a medical facility. When this chemical has been swallowed, get medical attention. If victim is conscious, administer water or milk. Do not induce vomiting. | [Shipping]
UN2218 Acrylic acid, stabilized, Hazard class: 8; Labels: 8-Corrosive material, 3-Flammable liquid | [Incompatibilities]
May form explosive mixture with air. Light, heat, and peroxides can cause polymerization. Use MEHQ (monomethyl ether of hydroquinone) as an inhibitor. Incompatible with oxidizers (chlorates, nitrates, peroxides, permanganates, perchlorates, chlorine, bromine, fluorine, etc.); contact may cause fires or explosions. Keep away from alkaline materials, strong bases, strong acids, oxoacids, epoxides. Incompatible with sulfuric acid, caustics, ammonia, amines, isocyanates, alkylene oxides; epichlorohydrin, toluene diamine, oleum, pyridine, methyl pyridine, n-methyl pyrrolidone, 2-methyl-6-ethyl aniline, aniline, ethylene diamine, ethyleneimine, and 2aminoethanol. Severely corrodes carbon steel and iron; attacks other metals. May accumulate static electrical charges and may cause ignition of its vapors. | [Description]
Acrylic acid (IUPAC: prop-2-enoic acid) is an organic compound with the formula CH2=CHCO2H. It is the simplest unsaturated carboxylic acid, consisting of a vinyl group connected directly to a carboxylic acid terminus. This colorless liquid has a characteristic acrid or tart smell. It is miscible with water, alcohols, ethers, and chloroform. More than one billion kilograms are produced annually. | [Waste Disposal]
Consult with environmental regulatory agencies for guidance on acceptable disposal practices. Generators of waste containing this contaminant (≥100 kg/mo) must conform with EPA regulations governing storage, transportation, treatment, and waste disposal. Incineration. 100 500 ppm potassium permanganate will degrade acrylic acid to a hydroxy acid which can be disposed of at a sewage treatment. | [Definition]
An unsaturated liquid carboxylic acid with a pungent odor.
The acid and its esters are used to make
ACRYLIC RESINS. | [Preparation]
There are three routes to acrylic acid which have commercial significance;
they are based on propylene, acetylene and ethylene respectively. At the
present time, most acrylic acid is produced via the propylene route.
(i) Propylene route. This route involves the two-stage oxidation of propylene:
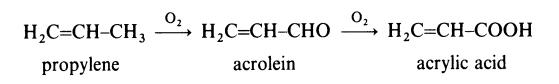
A mixture of propylene, air and steam is fed into a reactor containing a
catalyst at about 320??C to give acrolein. This intermediate is not isolated but
is passed directly to a second reactor, also containing a catalyst, at about
280??C. The effluent is cooled by contact with cold aqueous acrylic acid.
Acrylic acid is extracted from the solution with a solvent and then separated
by distillation. Because of the ready availability of low cost propylene, this
route has become the preferred route for the production of acrylic acid.
(ii) Acetylene route. This route involves the reaction of acetylene, carbon
monoxide and water:
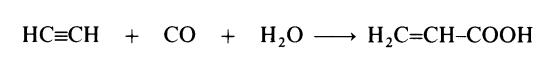
In one process, the reaction is conducted in solution in tetrahydrofuran at
about 200??C and 6-20 MPa (60--200 atmospheres). Nickel bromide is used as
catalyst. The solution of acrylic acid in tetrahydrofuran, after separation of
the unconverted acetylene and carbon monoxide in a degassing column,
passes to a distillation tower where tetrahydrofuran is taken overhead and
acrylic acid is the bottom product. The reaction between acetylene, carbon
monoxide and water may also be carried out by using nickel carbonyl as the
source of carbon monoxide. In this case, milder reaction conditions are
possible. Owing to the high cost of acetylene, this route is now of little
commercial importance.
(iii) Ethylene route. This route consists of the following sequence:
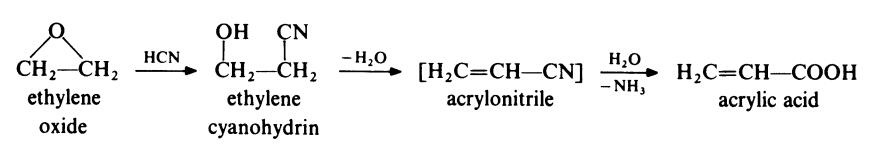
The addition of
hydrogen cyanide to ethylene oxide takes place at 55-60??C in the presence of
a basic catalyst such as diethylamine. The reaction is exothermic and is
carried out in solution to facilitate control; the solvent is conveniently
ethylene cyanohydrin. The reaction mixture is neutralized and ethylene
cyanohydrin is separated by distillation. The second stage of the synthesis
involves the dehydration and hydrolysis of ethylene cyanohydrin; these
reactions are carried out in one step by heating the cyanohydrin with
aqueous sulphuric acid at about 175??C. (It is possible, of course, that the intermediate in this conversion may be acrylonitrile, as shown, or P-hydroxy�propionic acid or both.) At one time this was the standard route for the
preparation of acrylic acid but it has been largely displaced by the more
economical propylene route. | [Production Methods]
Acrylic acid is produced from propene which is a by product of ethylene and gasoline production. CH2=CHCH3 + 1.5 O2→ CH2=CHCO2H + H2O Because acrylic acid and its esters have long been valued commercially, many other methods have been developed but most have been abandoned for economic or environmental reasons. An early method was the hydrocarboxylation of acetylene ("Reppe chemistry") : HCCH + CO + H2O → CH2=CHCO2H This method requires nickel carbonyl and high pressures of carbon monoxide. It was once manufactured by the hydrolysis of acrylonitrile which is derived from propene by ammoxidation, but was abandoned because the method cogenerates ammonium derivatives. Other now abandoned precursors to acrylic acid include ethenone and ethylene cyanohydrin. | [Fire Hazard]
Combustible liquid; flash point (closed cup)
54°C (130°F), (open cup) 68°C (155°F);
vapor pressure 31 torr at 25°C (77°F); vapor
density 2.5 (air=1); autoignition temperature 360°C (680°F). Vapors of acrylic acid
form explosive mixtures with air within the
range 2.9–8.0% by volume in air. Fireextinguishing agent: water spray, “alcohol”
foam, dry chemical, or CO2; use a water
spray to flush and dilute the spill and to disperse the vapors.
Acrylic acid may readily polymerize at
ambient temperature. Polymerization may
be inhibited with 200 ppm of hydroquinone
monomethyl ether (Aldrich 2006). In the
presence of a catalyst or at an elevated temperature, the polymerization rate may accelerate, causing an explosion. The reactions of
acrylic acid with amines, imines, and oleum
are exothermic but not violent. Acrylic acid
should be stored below its melting point with
a trace quantity of polymerization inhibitor.
Its reactions with strong oxidizing substances
can be violent. | [Flammability and Explosibility]
Flammable | [Safety]
Acrylic acid is severely irritating and corrosive to the skin and the respiratory tract. Eye contact can result in severe and irreversible injury. Low exposure will cause minimal or no health effects, while high exposure could result in pulmonary edema. The LD50 is 340 mg/kg (rat, oral). | [Environmental Fate]
Acrylic acid is corrosive, and its toxicity occurs at the site of
contact. | [Purification Methods]
It can be purified by steam distillation, or vacuum distillation through a column packed with copper gauze to inhibit polymerisation. (This treatment also removes inhibitors such as methylene blue that may be present.) Azeotropic distillation of the water with *benzene converts aqueous acrylic acid to the anhydrous material. [Beilstein 2 H 397, 2 I 186, 2 II 383, 2 III 1215, 2 IV 1455.] | [Substituents]
As a substituent acrylic acid can be found as an acyl group or a carboxyalkyl group depending on the removal of the group from the molecule. More specifically these are :
The acryloyl group, with the removal of the -OH from carbon-1.
The 2-carboxy ethenyl group, with the removal of a -H from carbon-3. This substituent group is found in chlorophyll.. | [Toxicity evaluation]
Acrylic acid’s large-scale use and production results in its release
into the environment. The most likely route of exposure is
inhalation because acrylic acid has a low vapor pressure. The
miscibility of acrylic acid in water combined with its low vapor
pressure prevent it from accumulating in the soil. Acrylic acid
that is emitted into the atmosphere is degraded photochemically
by reaction with hydroxyl radicals. There is no potential
for long-range atmospheric transport of acrylic acid because it
has an atmospheric lifetime of 1 month. | [Toxics Screening Level]
The current ITSL for acrylic acid (1 μg/m3) was originally established on 4/1/94. The averaging time (AT) assigned at that time was 24 hours. |
| Safety Data | Back Directory | [Symbol(GHS) ]
   
GHS02,GHS05,GHS07,GHS09 | [Signal word ]
Danger | [Hazard statements ]
H226-H302+H312+H332-H314-H335-H410 | [Precautionary statements ]
P210-P273-P280-P303+P361+P353-P304+P340+P310-P305+P351+P338 | [Hazard Codes ]
C,N | [Risk Statements ]
R10:Flammable.
R20/21/22:Harmful by inhalation, in contact with skin and if swallowed .
R35:Causes severe burns.
R50:Very Toxic to aquatic organisms. | [Safety Statements ]
S26:In case of contact with eyes, rinse immediately with plenty of water and seek medical advice .
S36/37/39:Wear suitable protective clothing, gloves and eye/face protection .
S45:In case of accident or if you feel unwell, seek medical advice immediately (show label where possible) .
S61:Avoid release to the environment. Refer to special instructions safety data sheet . | [OEB]
B | [OEL]
TWA: 2 ppm (6 mg/m3) [skin] | [RIDADR ]
UN 2218 8/PG 2
| [WGK Germany ]
1
| [RTECS ]
AS4375000
| [F ]
8-13 | [Autoignition Temperature]
744 °F | [TSCA ]
Yes | [REACH Registrations]
Active | [HazardClass ]
8 | [PackingGroup ]
II | [HS Code ]
29161110 | [Storage Class]
3 - Flammable liquids | [Hazard Classifications]
Acute Tox. 4 Dermal
Acute Tox. 4 Inhalation
Acute Tox. 4 Oral
Aquatic Acute 1
Aquatic Chronic 2
Eye Dam. 1
Flam. Liq. 3
Skin Corr. 1A
STOT SE 3 | [Safety Profile]
Poison by ingestion, skin contact, and intraperitoneal routes. An experimental teratogen. Other experimental reproductive effects. A severe skin and eye irritant. Questionable carcinogen with experimental carcinogenic and tumorigenic data. Corrosive. Flammable liquid. May undergo exothermic polymerization at room temperature. May become explosive if confined. A fire hazard when exposed to heat or flame. | [Hazardous Substances Data]
79-10-7(Hazardous Substances Data) | [Toxicity]
LD50 orally in rats: 2.59 g/kg (Smyth) |
| Questions And Answer | Back Directory | [Basic Information]
Acrylic acid (AAc, IUPAC: prop-2-enoic acid) is the simplest unsaturated carboxylic acid, consisting of a vinyl group connected directly to a carboxylic acid terminus with the formula CH2=CHCO2H[1][2] which is a colorless liquid above its freezing point of 13°C ( 56°F) with a distinctive acrid odor[3][4][5]. It is corrosive to metals and tissue and prolonged exposure to fire or heat can cause polymerization. If polymerization takes place in a closed container, violent rupture may occur because the polymerizaiton of acrylic acid is exothermic. The inhibitor (usually hydroquinone) can greatly reduce the tendency to polymerize. It is miscible with water, alcohol, ether, benzene, chloroform, and acetone, but incompatible with strong oxidisers, strong bases, strong alkalies and pure nitrogen. It may polymerize (sometimes explosively) when contacting with amines, ammonia, oleum and chlorosulfonic acid, iron salts and peroxides[2][4].
| [Preparation methods]
Acrylic acid can be prepared in different ways, for example as follows:
The easiest way to prepare pure acrylic acid in the laboratory is to exchange the ester of formic acid and readily available methyl acrylate. Sulfuric acid is a good catalyst.
CH2=CHCOOCH3 +HCOOH→CH2=CHCOOH+HCOOCH3
Acrolein can be oxidized in the liquid phase at 20-40℃ with silver or vanadium as the catalyst and methoxybenzene as the solvent which glacial acetic acid is more commonly used. It has been reported that the yield of acrylic acid can reach 65-90% based on the consumption of acrolein.
CH2=CHCHO+ 1/2 O2→CH2=CHCOOH
A mixture of water and 2,3 dibromopropionic acid is treated with zinc powder can obtain an aqueous solution of acrylic acid with a yield of 90%.
CH2BrCHBrCOOH +Zn →CH2=CHCOOH+ZnBr2
Acrylic acid also can be obtained by pyrolyzing sec-butyl acrylate at 500℃, or pyrolyzing ethyl acrylate at 570℃.
CH2=CHCOOCH(CH3)CH2CH3 → CH2=CHCOOH+CH3CH=CHCH3
At 200-300℃, acrylic acid can be obtained by passing carbon dioxide and ethylene through the silica gel impregnated with iron sulfite.
CH2=CH2+CO2→CH2=CHCOOH | [Application]
Acrylic acid is a carboxylic acid, of which the primary use is in the production of acrylic esters[2]. It has been traditionally used as the raw material for acrylic esters – methyl acrylate, ethyl acrylate, butyl acrylate and 2-ethylhexyl acrylate which were originally used to produce solvent-based acrylic resins but environmental concerns about solvent use led to the development of water-based acrylics[7]. Acrylic acid can readily react with a wide variety of organic and inorganic compounds which results in it’s considered as a very useful feedstock to manufacture many low molecular compounds, such as propionic acid, unsaturated fatty acids, heterocyclic compounds, and Diels-Alder addition products. As a vinyl compound and a carboxylic acid, acrylic acid is used widely for polymerisation, including production of polyacrylates. It is also a monomer for polyacrylic and polymethacrylic acids and other acrylic polymers[4].
Acrylic acid and esters are flammable, reactive, volatile liquids based on an alpha-, beta-unsaturated carboxyl structure. Incorporation of varying percentages of acrylate monomers permits the production of many formulations for latex and solution copolymers, copolymer plastics and cross-linkable polymer systems. Their performance characteristics—which impart varying degrees of tackiness, durability, hardness, and glass transition temperatures—promote consumption in many end-use applications. Major markets for the esters include surface coatings, textiles, adhesives, and plastics[8][9].
Polyacrylic acid which produced by acrylic acid can be further modified to produce superabsorbent polymers (SAPs) and other polyacrylic acid homopolymers or copolymers used as detergents, dispersants/antiscalants, anionic polyelectrolytes for water treatment, and rheology modifiers[7][8].
SAPs are cross-linked polyacrylates with the ability to absorb and retain more than 100 times their own weight in liquid. They have experienced very strong growth, primarily in baby diapers (nappies) and incontinence products[7][8].
A new application for SAPs is soaker pads used in food packaging. In 2007, the US Food and Drug Adminstration authorised SAPs in packaging with indirect food contact for poultry, meat, fish, fruit and vegetables[7].
Crude acrylic acid (CAA) is made by the oxidation of propylene. About 55% of the CAA is converted to acrylate esters. The remaining 45% is purified to 98–99.5% purity to glacial acrylic acid (GAA), which, in turn, is converted to polyacrylic acid, which is used to produce superabsorbent polymers (SAPs) and other polyacrylic acid copolymers. In 2016, global glacial acrylic acid consumption was estimated to account for about 45% of total crude acrylic acid consumption, of which 79% was consumed for superabsorbent polymers. Growth in GAA consumption is forecast at about 5% per year during 2016?21. Growth in demand for crude acrylic acid is forecast at 4.5% per year during 2016?21, driven by growth in superabsorbent polymers at 5.5% and acrylate esters at about 4%. SAP growth will be strongest in China and other areas of Asia, but will be much more moderate in the mature regions of North America, Western Europe, and Japan[8].
| [Health & Safety Hazard]
Tests involving acute exposure of rats, mice, and rabbits have demonstrated that acrylic acid has moderate acute toxicity by inhalation or ingestion, and high acute toxicity by dermal exposure. Acrylic acid is a strong irritant to the skin, eyes, and mucous membranes in humans. The liquid may cause blindness if splashed into the eye. Acute (short-term) exposure of rats to acrylic acid by inhalation has been observed to produce nose and eye irritation, lung haemorrhage, and degenerative changes in the liver and kidneys.
Some ill-health effects could happen when people exposed to acrylic acid, while people can be easily exposed to acrylic acid through direct contact with a product containing it or by inhaling it in air contaminated by a nearby plant manufacturing acrylic acid, for example, in the workplace, exposure to acrylic acid occurs primarily via inhalation and dermal contact during its manufacture or use; consumers may be exposed to acrylic acid in polishes, paints, coatings, rug backings, adhesives, plastics, textiles, and paper finishes. In addition, acrylic acid may be released in wastewater and can be also produced naturally by some species of algae. When we do not feel well, we should get medical attention immediately.
Acrylic acid is sensitive to heat and sunlight and also a fire hazard when exposed to heat or flame. The product should be stored in a segregated and approved area away from heat, sources of ignition and the container should be kept in a cool, well-ventilated area, tightly closed and sealed until ready for use. It is also very necessary to keep the product away from incompatibles such as oxidising agents, acids, alkalis, moisture.
| [Reference]
[1]https://www.sigmaaldrich.com/catalog/product/aldrich/147230?lang=zh®ion=CN
[2]https://www.chemwatch.net/211745/acrylic-acid
[3]https://www.arkema.com/en/products/product-finder/product-viewer/Acrylic-acid/
[4]https://www.chemicalbook.com/ChemicalProductProperty_EN_CB7307797.htm
[5]http://www.chemspider.com/Chemical-Structure.6333.html
[6]Manhua Mandy Lin. Selective oxidation of propane to acrylic acid with molecular oxygen[J]. Applied Catalysis A:General 207(2001)1-16.
[7]https://www.icis.com/resources/news/2007/11/01/9074870/acrylic-acid-uses-and-market-data/
[8]https://ihsmarkit.com/products/acrylic-acid-acrylate-esters-chemical-economics-handbook.html;
[9]https://www.arkema.com/en/products/product-finder/product-viewer/Acrylic-acid/
[10]https://www.epa.gov/sites/production/files/2016-09/documents/acrylic-acid.pdf
|
|
|