Ferric chloride
- CAS No.
- 7705-08-0
- Chemical Name:
- Ferric chloride
- Synonyms
- FeCl3;IRON(III) CHLORIDE;IRON CHLORIDE;Ferric chloride anhydrous;FERRIC CHLORIDE SOLUTION;DB03;FERRIC CHLORID;Iron perchloride;IRON TRICHLORIDE;Iron(Ⅲ) chloride
- CBNumber:
- CB5444364
- Molecular Formula:
- Cl3Fe
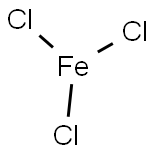
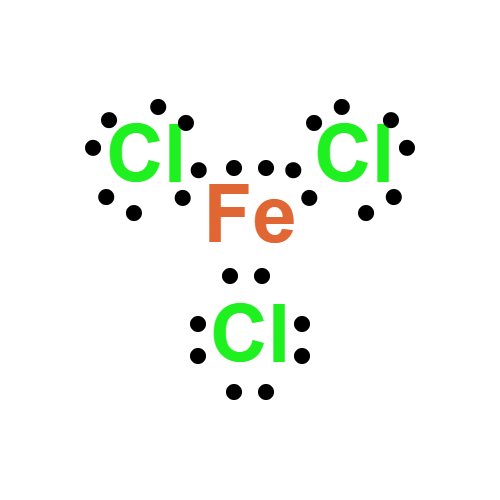
Lewis structure
- Molecular Weight:
- 162.2
- MDL Number:
- MFCD00011005
- MOL File:
- 7705-08-0.mol
- MSDS File:
- SDS
| Melting point | 304 °C(lit.) |
|---|---|
| Boiling point | 316 °C |
| Density | 2,804 g/cm3 |
| vapor density | 5.61 (vs air) |
| vapor pressure | 1 mm Hg ( 194 °C) |
| refractive index | n20/D1.414 |
| Flash point | 316°C |
| storage temp. | Store below +30°C. |
| solubility | H2O: soluble |
| form | powder |
| color | Yellow |
| Specific Gravity | 2.804 |
| PH | 1 (200g/l, H2O, 20℃) |
| Water Solubility | 920 g/L (20 ºC) |
| Sensitive | Hygroscopic |
| Merck | 14,4019 |
| Exposure limits |
ACGIH: TWA 1 mg/m3 NIOSH: TWA 1 mg/m3 |
| Stability | Stable. Very sensitive to moisture. Incompatible with strong oxidizing agents; forms explosive mixtures with sodium, potassium. Hygroscopic. |
| CAS DataBase Reference | 7705-08-0(CAS DataBase Reference) |
| FDA 21 CFR | 184.1297; 175.105; 176.170; 310.545 |
| Substances Added to Food (formerly EAFUS) | FERRIC CHLORIDE |
| SCOGS (Select Committee on GRAS Substances) | Ferric chloride (packaging) |
| EWG's Food Scores | 1-4 |
| FDA UNII | U38V3ZVV3V |
| NIST Chemistry Reference | Ferric chloride(7705-08-0) |
| EPA Substance Registry System | Ferric chloride (7705-08-0) |
SAFETY
Risk and Safety Statements
| Symbol(GHS) |     GHS02,GHS05,GHS07,GHS08 |
|||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Signal word | Danger | |||||||||
| Hazard statements | H225-H290-H302-H318-H335-H351 | |||||||||
| Precautionary statements | P201-P210-P233-P261-P280-P305+P351+P338+P310 | |||||||||
| Hazard Codes | C,Xn,Xi | |||||||||
| Risk Statements | 41-38-22-34-37/38-10-36 | |||||||||
| Safety Statements | 26-39-45-36/37/39 | |||||||||
| RIDADR | UN 2582 8/PG 3 | |||||||||
| WGK Germany | 1 | |||||||||
| RTECS | LJ9100000 | |||||||||
| TSCA | Yes | |||||||||
| HS Code | 2827 39 20 | |||||||||
| HazardClass | 8 | |||||||||
| PackingGroup | III | |||||||||
| Toxicity | LD50 orally in Rabbit: 316 mg/kg | |||||||||
| NFPA 704 |
|
Ferric chloride price More Price(41)
| Manufacturer | Product number | Product description | CAS number | Packaging | Price | Updated | Buy |
|---|---|---|---|---|---|---|---|
| Sigma-Aldrich | 8.03945 | Iron(III) chloride anhydrous for synthesis | 7705-08-0 | 500g | $52.2 | 2024-03-01 | Buy |
| Sigma-Aldrich | 8.03945 | Iron(III) chloride anhydrous for synthesis | 7705-08-0 | 1kg | $69.3 | 2024-03-01 | Buy |
| Sigma-Aldrich | 80353 | TDA Reagent suitable for microbiology | 7705-08-0 | 50mL | $358 | 2024-03-01 | Buy |
| Sigma-Aldrich | 8.03945 | Iron(III) chloride anhydrous for synthesis | 7705-08-0 | 60kg | $991 | 2024-03-01 | Buy |
| Sigma-Aldrich | 12322 | Iron(III) chloride solution purum, 45% FeCl3 basis | 7705-08-0 | 2.5l | $204 | 2024-03-01 | Buy |
Ferric chloride Chemical Properties,Uses,Production
Physical properties
Ferric chloride, solution appears as a colorless to light brown aqueous solution that has a faint hydrochloric acid odor. Highly corrosive to most metals and probably corrosive to tissue. Noncombustible. Used in sewage treatment and water purification.
Ferric chloride is an dark brown hexagonal crystals; hygroscopic; density 2.898g/cm3; melts at 306°C; decomposes at 315°C; highly soluble in water (74.4g/100g water at 0°C); very soluble in alcohol, ether and acetone. The hexahydrate is brownish-yellow crystalline mass; deliquesces; melts at 37°C; vaporizes around 280°C; highly soluble in water (92g/100g water at 20°C); very soluble in organic solvents such as ethanol, ether and acetone.
Uses
Iron(III) chloride occurs naturally as the mineral molysite. The compound is widely used to prepare a number of iron(III) salts. Also, it is applied in sewage and industrial waste treatment processes. It also is used in the manufacture of dyes, pigments and inks; as a chlorinating agent; and as a catalyst in chlorination reactions of aromatics.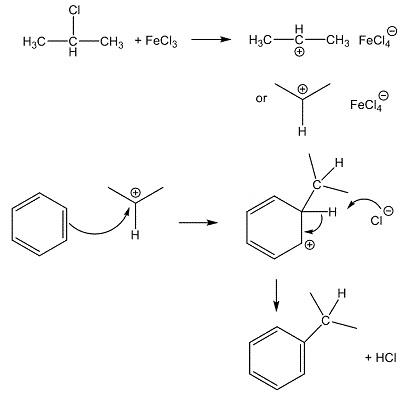
In the laboratory, anhydrous Ferric chloride(FeCl3)can be used as an alternative to anhydrous AlCl3 in Friedel-Crafts reactions, like the alkylation or acylation of aromatic rings. Being a small, highly charged metal, it polarises reagents like halogenoalkanes (alkyl halides), which generates carbocations (carbenium ions) that then attack benzene rings, as in the synthesis here.
Production Methods
Iron(III) chloride forms passing chlorine gas over iron filings at 350°C:
2Fe + 3Cl2 → 2FeCl3
It also forms heating iron(III) oxide with HCl at elevated temperatures:
Fe2O3 + 6HCl → 2FeCl3 + 3H2O
The product may be sublimed in a stream of chlorine to give high purity grade iron(III) chloride.
The anhydrous chloride also may be made by heating the hexahydrate, FeCl3•6H2O, with thionyl chloride:
FeCl3•6H2O + 6SOCl2 → FeCl3 + 12HCl + SO2
Application
The following list represents some of the most common and largest applications of Ferric Chloride:
As a purifying agent in water supply and as a coagulant in municipal and industrial wastewater treatment. In this application, Ferric chloride’s rapid hydrolysis in water makes it an ideal flocculating and precipitating agent. The ferric hydroxide (Fe[OH]3) produced forms flocs (small clumps or tufts) that adsorb suspended particles of various materials (e.g., colloids, clays and bacteria). The clumps, with the adsorbed matter, are then allowed to settle for easy removal. Ferric chloride forms precipitates with hydrogen sulfide (H2S), phosphate (PO4), arsenic as arsenate (AsO4) and hydroxide alkalinity (OH).
As an oxidant in indigo blue dyestuff production.
As an etching medium in producing printed circuit boards (PCBs).
As a catalyst for the reaction of ethylene with chlorine, forming ethylene dichloride (1,2-dichloroethane), an important commodity chemical, which is mainly used for the industrial production of vinyl chloride, the monomer for making PVC.
www.marchpump.com
Description
Ferric chloride (iron(IH)chloride, FeCl3, CAS No. 7705-08-0) may be prepared from iron and chlorine or from ferric oxide and hydrogen chloride. The pure material occurs as hydroscopic, hexagonal, dark crystals. Ferric chloride hexahydrate (iron(III)chloride hexahydrate, FeCl3*6H2O, CAS No. 10025-77-1) is readily formed when ferric chloride is exposed to moisture.
Chemical Properties
Ferric chloride,FeCl3, is a brown crystalline solid and is soluble in water,alcohol,and glycerol. It is also known as anhydrous ferric chloride,ferric trichloride, Flores martis,and iron chloride. Ferric chloride is used as a coagulant for sewage and industrial wastes, as an oxidizing and chlorinating agent,as a disinfectant, in copper etching, and as amordant. In addition, this compound is employed in the ferric chloride test,which is used to assess the relative corrosion resistance of stainless and nickel-base alloys. The ferric chloride test has been shown to be an appropriate measure of the suitability of such alloys for service in paper mill bleach plants and seawater.
Chemical Properties
Ferric Chloride is a black-brown, dark-green, or black crystalline solid.
Uses
Treatment of sewage and industrial wastes; etching agent for engraving, photography, and printed circuitry; condensation catalyst in FriedelCrafts reactions; mordant; oxidizing, chlorinating, and condensing agent; disinfectant; pigment; feed additive; wat
Uses
Ferric Chloride is a nutrient and dietary supplement that serves as a source of iron.
Uses
It is used for sewerage treatment of industrial wastes, chloride hydrometallurgy (Silgarin process for the production of silicon), drinking water production, production of precursor for polyvinyl chloride (PVC), and copper-based metals in printed circuit boards. Iron (III) chloride is used as catalyst for the reaction of ethylene with chlorine, as a leaching agent, and as a drying reagent in certain reactions. It is a mild oxidizing agent and converts copper(I) chloride in to copper(II) chloride. As a moderately strong Lewis acid, it catalyzes chlorination of aromatic compounds and Friedel-Crafts reactions. Along with sodium iodide, it brings about reduction of organic azides to corresponding amines. It is useful in the detection of phenols and phenolic derivatives, gamma-hydroxybutyric acids, and also in Trinder spot test for detecting salicylic acids.
Definition
ChEBI: Iron trichloride is an iron coordination entity. It has a role as a Lewis acid and an astringent.
General Description
Ferric chloride is an orange to brown-black solid. Ferric chloride is slightly soluble in water. Ferric chloride is noncombustible. When wet Ferric chloride is corrosive to aluminum and most metals. Pick up and remove spilled solid before adding water. Ferric chloride is used to treat sewage, industrial waste, to purify water, as an etching agent for engraving circuit boards, and in the manufacture of other chemicals.
Air & Water Reactions
Very hygroscopic. Slightly water soluble, where a 0.1M solution has a pH of 2.0.
Reactivity Profile
Alkali metal hydroxides, acids, anhydrous chlorides of iron, tin, and aluminum, pure oxides of iron and aluminum, and metallic potassium are some of the catalysts that may cause ethylene oxide to rearrange and polymerize, liberating heat, [J. Soc. Chem. Ind. 68:179(1949)]. Explosions occur , although infrequently, from the combination of ethylene oxide and alcohols or mercaptans, [Chem. Eng. News 20:1318(1942)]. Allyl chloride may polymerize violently under conditions involving an acid catalyst, such as sulfuric acid, Ferric chloride, aluminum chloride, Lewis acids, and Ziegler type catalysts (initiators), [Ventrone (1971)].
Hazard
Toxic by ingestion, strong irritant to skin and tissue.
Health Hazard
Inhalation of dust may irritate nose and throat. Ingestion causes irritation of mouth and stomach. Dust irritates eyes. Prolonged contact with skin causes irritation and burns.
Fire Hazard
Special Hazards of Combustion Products: Irritating hydrogen chloride fumes may form in fire.
Flammability and Explosibility
Non flammable
Industrial uses
Ferric chloride (FeCl3) is obtained by an iron chlorination method at a temperature of 600–700 °C. Very limited data are available on the use of ferric chloride in the mineral processing industry. Ferric chloride has a depressing effect on barite and can be used in barite–celestite separation. It was also evaluated as a depressant during niobium– zirconium separation. In general, ferric and ferrous compounds are not selective depressants and in many cases are detrimental for flotation of oxidic and industrial minerals as in the case of anionic flotation, fatty acid, iron complexes or oleate iron complexes.
Safety Profile
Poison by ingestion and intravenous routes. Experimental reproductive effects. Corrosive. Probably an eye, skin, and mucous membrane irritant. Mutation data reported. Reacts with water to produce toxic and corrosive fumes. Catalyzes potentially explosive polymerization of ethylene oxide, chlorine + monomers (e.g., styrene). Forms shock sensitive explosive mixtures with some metals (e.g., potassium, sodium). Violent reaction with all$ chloride. When heated to decomposition it emits highly toxic fumes of HCl.
Potential Exposure
Iron chloride is used to treat sewage and industrial waste. It is also used as an etchant for photo engraving and rotogravure; in textiles; photography; as a disinfectant; as a feed additive.
First aid
If this chemical gets into the eyes, remove anycontact lenses at once and irrigate immediately for at least15 min, occasionally lifting upper and lower lids. Seek medical attention immediately. If this chemical contacts theskin, remove contaminated clothing and wash immediatelywith soap and water. Seek medical attention immediately. Ifthis chemical has been inhaled, remove from exposure,begin rescue breathing (using universal precautions, including resuscitation mask) if breathing has stopped and CPR ifheart action has stopped. Transfer promptly to a medicalfacility. When this chemical has been swallowed, give milkand get medical attention. Do not induce vomiting. Thesymptoms of metal fume fever may be delayed for 4-12 hfollowing exposure: it may last less than 36 h.Note to physician: Gastric lavage should be performed followed by saline catharsis and anodyne. Administer deferoxamine, I.V. Watch for late stricture. Serum, plasma, orurinary iron levels may be employed to estimate the amountingested.Note to physician: For severe poisoning do not use BAL[British Anti-Lewisite, dimercaprol, dithiopropanol(C3H8OS2)] as it is contraindicated or ineffective in poisoning from iron.
storage
(1) Color Code—White: Corrosive or ContactHazard; Store separately in a corrosion-resistant location.(2) Color Code—Blue: Health Hazard/Poison: Store in asecure poison location. Prior to working with this chemicalyou should be trained on its proper handling and storage.Iron chloride must be stored in tightly closed containers toavoid contact with sulfuric acid, sodium, potassium, allylchloride, and water, since violent reactions occur and toxicvapors may be produced.
Shipping
UN1773 Ferric chloride, anhydrous, Hazard class: 8; Labels: 8-Corrosive material. UN2582 Ferric chlo ride, solution, Hazard class: 8; Labels: 8-Corrosive material
Purification Methods
Sublime it at 200o in an atmosphere of chlorine. It is an “iron-black” coloured powder with green irridescence. Store it in a weighing bottle inside a desiccator as it absorbs moisture from air to form the yellow hexahydrate (see next entry). [Tarr Inorg Synth III 191 1950, Pray Inorg Synth V 153 1957, Epperson Inorg Synth VII 163 1963.]
Structure and conformation
The crystalline solid has a semicovalent layer structure with hexagonal packing of chloride ions, each iron atom being surrounded octahedrally by six chlorines in a BiI3 type structure. The dimers in the vapour phase have a structure similar to that of Al2Cl6 with the iron atoms surrounded by chlorines in a roughly tetrahedral fashion. The magnetic properties of iron(III) chloride in its different environments have been investigated extensively. The magnetic moment at 290°K is 5-73 B.M. and is independent of the field strength. In aqueous hydrochloric acid the room temperature moment is 5-94 B.M. and the hexahydrate has a similar moment (5-95 B.M.).
Incompatibilities
Aqueous solutions are a strong acid. Violent reaction with bases, allyl chloride; sulfuric acid; water. Shock- and friction-sensitive explosive material forms with potassium, sodium and other active metals. Attacks metals when wet.
Waste Disposal
Neutralize with lime or soda ash and bury in an approved landfill.
Ferric chloride Preparation Products And Raw materials
Raw materials
1of2
Preparation Products
1of8
| Supplier | Tel | Country | ProdList | Advantage | |
|---|---|---|---|---|---|
| Hebei Guanlang Biotechnology Co,.LTD | +8619930503252 | daisy@crovellbio.com | China | 5964 | 58 |
| Henan Tianfu Chemical Co.,Ltd. | +86-0371-55170693 +86-19937530512 | info@tianfuchem.com | China | 21691 | 55 |
| Shanghai Time Chemicals CO., Ltd. | +86-021-57951555 +8617317452075 | jack.li@time-chemicals.com | China | 1807 | 55 |
| Hefei TNJ Chemical Industry Co.,Ltd. | +86-0551-65418679 +86-18949832763 | info@tnjchem.com | China | 2989 | 55 |
| Tianjin Zhongxin Chemtech Co., Ltd. | +86-022-66880623 +8618622897568 | sales@tjzxchem.com | China | 559 | 58 |
| career henan chemical co | +86-0371-86658258 | sales@coreychem.com | China | 29914 | 58 |
| City Chemical LLC | 2039322489 | sales@citychemical.com | United States | 168 | 58 |
| Hubei Jusheng Technology Co.,Ltd. | 18871490254 | linda@hubeijusheng.com | CHINA | 28180 | 58 |
| Hebei Guanlang Biotechnology Co., Ltd. | +86-19930503282 | alice@crovellbio.com | China | 8823 | 58 |
| Hubei xin bonus chemical co. LTD | 86-13657291602 | linda@hubeijusheng.com | CHINA | 22968 | 58 |
Related articles
- What is the charge of the metal ion in FeCl3?
- Ferric chloride is named as iron chloride. It is a chemical compound with a chemical formula consisting of FeCl3. The charge ....
- Feb 19,2024
- Application of Ferric trichloride
- Ferric trichloride is an important chemical raw material, because it has trivalent iron ions, so ferric trichloride has good o....
- Jan 13,2022
View Lastest Price from Ferric chloride manufacturers
| Image | Update time | Product | Price | Min. Order | Purity | Supply Ability | Manufacturer | |
|---|---|---|---|---|---|---|---|---|
 |
2024-02-22 | Ferric chloride
7705-08-0
|
US $1.00 / kg | 1kg | 99% | 100000kg/month | Hubei Langyou International Trading Co., Ltd | |
 |
2023-10-10 | Ferric chloride
7705-08-0
|
US $96.00 / KG | 25KG | 99.99% | 200ton | Hebei Mojin Biotechnology Co., Ltd | |
 |
2023-09-11 | Ferric chloride
7705-08-0
|
US $0.00-0.00 / kg | 1kg | 0.99 | 100tons | Hebei Yanxi Chemical Co., Ltd. |
-

- Ferric chloride
7705-08-0
- US $1.00 / kg
- 99%
- Hubei Langyou International Trading Co., Ltd
-

- Ferric chloride
7705-08-0
- US $96.00 / KG
- 99.99%
- Hebei Mojin Biotechnology Co., Ltd
-

- Ferric chloride
7705-08-0
- US $0.00-0.00 / kg
- 0.99
- Hebei Yanxi Chemical Co., Ltd.