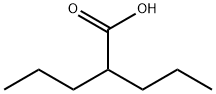
2-Propylpentanoic acid
- CAS No.
- 99-66-1
- Chemical Name:
- 2-Propylpentanoic acid
- Synonyms
- VALPROIC ACID;valproate;VPA;DIVALPROEX;Valproic;depakote;Depakene;Depakine;Mylproin;2-PropyL
- CBNumber:
- CB7149528
- Molecular Formula:
- C8H16O2
- Molecular Weight:
- 144.21
- MDL Number:
- MFCD00002672
- MOL File:
- 99-66-1.mol
| Melting point | -21.25°C (estimate) |
|---|---|
| Boiling point | 220 °C (lit.) |
| Density | 0.9 g/mL at 25 °C (lit.) |
| vapor pressure | 0.01 hPa (20 °C) |
| refractive index |
n |
| Flash point | 232 °F |
| storage temp. | Store below +30°C. |
| solubility | H2O: slightly soluble |
| form | Liquid |
| pka | 4.6(at 25℃) |
| color | Clear colorless to pale yellow |
| explosive limit | 1%(V) |
| Odor Threshold | 0.0033ppm |
| Water Solubility | slightly soluble |
| Merck | 14,9913 |
| BRN | 1750447 |
| BCS Class | 1,2 |
| LogP | 1.59 at 22.1℃ and pH5 |
| CAS DataBase Reference | 99-66-1(CAS DataBase Reference) |
| EWG's Food Scores | 2 |
| NCI Dictionary of Cancer Terms | Depakene; valproic acid |
| FDA UNII | 614OI1Z5WI |
| NCI Drug Dictionary | Depakote |
| ATC code | N03AG01 |
| Proposition 65 List | Valproate (Valproic acid) |
| NIST Chemistry Reference | Valproic Acid(99-66-1) |
| EPA Substance Registry System | Valproic acid (99-66-1) |
SAFETY
Risk and Safety Statements
| Symbol(GHS) |   GHS07,GHS08 |
|||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Signal word | Danger | |||||||||
| Hazard statements | H302-H315-H319-H360D | |||||||||
| Precautionary statements | P201-P301+P312+P330-P302+P352-P305+P351+P338-P308+P313 | |||||||||
| Hazard Codes | Xn,T,F | |||||||||
| Risk Statements | 22-36/37/38-39/23/24/25-23/24/25-11-34-61 | |||||||||
| Safety Statements | 26-45-36/37-16-7-36/37/39-53 | |||||||||
| RIDADR | UN 1230 3/PG 2 | |||||||||
| WGK Germany | 3 | |||||||||
| RTECS | YV7875000 | |||||||||
| HazardClass | 8 | |||||||||
| PackingGroup | III | |||||||||
| HS Code | 29159080 | |||||||||
| Toxicity | LD50 orally in rats: 670 mg/kg (Jenner) | |||||||||
| NFPA 704 |
|
2-Propylpentanoic acid price More Price(51)
| Manufacturer | Product number | Product description | CAS number | Packaging | Price | Updated | Buy |
|---|---|---|---|---|---|---|---|
| Sigma-Aldrich | V-006 | Valproic acid solution 1.0?mg/mL in methanol, ampule of 1?mL, certified reference material, Cerilliant? | 99-66-1 | 1mL | $110 | 2024-03-01 | Buy |
| Sigma-Aldrich | P6273 | 2-Propylpentanoic acid | 99-66-1 | 100ml | $207 | 2024-03-01 | Buy |
| Sigma-Aldrich | 8.14439 | 2-Propylvaleric acid for synthesis | 99-66-1 | 25mL | $76.7 | 2024-03-01 | Buy |
| Sigma-Aldrich | 1708707 | Valproic acid United States Pharmacopeia (USP) Reference Standard | 99-66-1 | 500mg | $436 | 2024-03-01 | Buy |
| TCI Chemical | P0823 | 2-Propylvaleric Acid >99.0%(GC)(T) | 99-66-1 | 25mL | $48 | 2024-03-01 | Buy |
2-Propylpentanoic acid Chemical Properties,Uses,Production
Description
Valproate is a first- generation antiepileptic drug (AED) known with the proprietary brand names of Epilim® (Sanofi, Paris) and Episenta® (Desitin, Hamburg) in the UK and Depakote® (Sanofi, Paris) in the USA.
Generic formulation
MHRA/ CHM advice to minimize risk when switching patients with epilepsy between different manufacturers’ products (including generic products):
- The need for continued supply of a particular manufacturer’s product should be based on clinical judgment and consultation with the patient and/ or carer, taking into account factors such as seizure frequency and treatment history.
Indications
Epilepsy
Monotherapy and adjunctive therapy of focal and generalized seizures.
Recommendations summarized from NICE (2012)
- Seizure types: first line (generalized tonic- clonic seizures, tonic/ atonic seizures, absence seizures, myoclonic seizures, focal seizures), adjunctive (generalized tonic- clonic seizures, absence seizures, myoclonic seizures, focal seizures).
- Epilepsy types: first line (absence syndromes, juvenile myoclonic epilepsy, epilepsy with generalized tonic- clonic seizures only, idiopathic generalized epilepsy, benign epilepsy with centrotemporal spikes, Panayiotopoulos syndrome, late- onset childhood occipital epilepsy, Dravet syndrome, Lennox– Gastaut syndrome), adjunctive (absence syndromes, juvenile myoclonic epilepsy, epilepsy with generalized tonic- clonic seizures only, idiopathic generalized epilepsy, benign epilepsy with centrotemporal spikes, panayiotopoulos syndrome, late- onset childhood occipital epilepsy).
Psychiatry
Treatment of acute mania associated with bipolar disorder.
Neurology
Migraine prophylaxis (unlicensed).
Dose titration
Epilepsy
600 mg daily divided into 1 or 2 doses, then increased by 150– 300 mg every 3 days; usual maintenance 1000– 2000 mg (or 20–30 mg/ kg) daily divided into 1 or 2 doses (max 2500 mg daily).
Mania
750 mg daily divided into 1 or 2 doses, adjusted according to response; usual maintenance 000– 2000 mg daily divided into 1 or 2 doses (doses greater than 45 mg/ kg daily require careful monitoring).
Plasma levels monitoring
Although plasma levels can be measured, and a therapeutic range has been postulated (40– 100 mg/ L), plasma valproate concentrations are not a useful index of efficacy. Therefore, routine monitoring is unhelpful.
Cautions
- Patients with systemic lupus erythematosus.
- Patients with a personal or family history of severe hepatic dysfunction (contraindication).
- Patients with known metabolic disorders (contraindication).
- • Patients with suspected metabolic disorders (contraindication).
- • Patients with porphyria (contraindication).
Adverse effects
Valproate can be associated with adverse effects at the level the nervous system and other systems.
Interactions
With AEDs
- AEDs with enzyme inducing effect (including carbamazepine, phenobarbital, phenytoin) decrease valproate plasma concentrations.
- Valproate reduces the metabolism of lamotrigine and increases the lamotrigine mean half- life by nearly two fold. This interaction may lead to increased lamotrigine toxicity, in particular serious skin rashes.
- Valproate increases phenobarbital and primidone plasma concentrations with exacerbation of its adverse effects (sedation may occur).
- Valproate may potentiate toxic effects of carbamazepine.
- Valproate decreases phenytoin total plasma concentration, but displaces phenytoin from its plasma protein binding sites and reduces its hepatic catabolism, thereby increasing phenytoin free form with possible overdose symptoms.
- Concomitant administration of valproate and topiramate has been associated with encephalopathy and/ or hyperammonaemia. In patients taking these two AEDs, careful monitoring of signs and symptoms is advised (especially in patients with pre- existing encephalopathy).
- Mefloquine and chloroquine increase valproate metabolism and may lower the seizure threshold (therefore, epileptic seizures may occur in cases of combined therapy).
- Decreases in blood levels of valproate have been reported when it is coadministered with carbapenem antibiotics (such as imipenem, panipenem, meropenem), resulting in a 60– 00% decrease in valproate levels within 2 days, sometimes associated with convulsions.
- Colestyramine may decrease the absorption of valproate.
- Rifampicin may decrease valproate blood levels, resulting in a lack of therapeutic effect.
- In case of concomitant use of valproate and highly protein bound agents (e.g. aspirin), free valproate plasma levels may be increased.
- Valproic acid plasma levels may be increased (as a result of reduced hepatic metabolism) in case of concomitant use with cimetidine or erythromycin.
- Valproate may potentiate the effect of other psychotropics such as antipsychotics (especially olanzapine), MAO inhibitors, antidepressants, and benzodiazepines.
- Valproate may raise zidovudine plasma concentration, possibly leading to increased zidovudine toxicity.
- The anticoagulant effect of warfarin and other coumarin anticoagulants may be increased following displacement from plasma protein binding sites by valproate.
There are no specific foods that must be excluded from diet when taking valproate. Alcohol intake is not recommended during treatment with valproate.
Special populations
Hepatic impairment
Avoid if possible: hepatotoxicity and hepatic failure may occasionally occur (usually in first 6 months). Avoid in active liver disease.
Renal impairment
In patients with renal insufficiency, it may be necessary to decrease dosage of valproate. As monitoring of plasma concentrations may be misleading, dosage should be adjusted according to clinical monitoring.
Pregnancy
- Valproate is associated with the highest risk of major and minor congenital malformations (in particular neural tube defects) and neurodevelopmental effects among AEDs.
- Therefore, valproate should not be used during pregnancy or in women of child- bearing age unless there is no safer alternative and only after a careful discussion of the risks.
- If valproate is to be used during pregnancy, the lowest effective dose should be prescribed in divided doses or as modified- release tablets to avoid peaks in plasma valproate concentrations (doses greater than 000 mg daily are associated with an increased risk of teratogenicity). The dose should be monitored carefully during pregnancy and after birth, and adjustments made on a clinical basis.
- Avoid use in the treatment of epilepsy and bipolar disorder unless there is no safer alternative and only after a careful discussion of the risks (effective contraception advised in women of child- bearing potential).
- Neonatal bleeding (related to hypofibrinaemia) and hepatotoxicity have been reported, and specialist prenatal monitoring should be instigated when valproate has been taken in pregnancy.
- Valproate is excreted in human milk with a concentration ranging from 1 to 10% of maternal serum levels. Haematological disorders have been shown in breastfed newborns/ infants of treated women. A decision must be made whether to discontinue breastfeeding or to discontinue/ abstain from valproate therapy, taking into account the benefit of breastfeeding for the child and the benefit of therapy for the woman.
Behavioural and cognitive effects in patients with epilepsy
The incidence of adverse psychiatric effects associated with valproate in patients with epilepsy is overall negligible (apart from reports of depression, irritability, and other behavioural symptoms in the context of encephalopathy). Cognitive difficulties have occasionally been reported in patients with epilepsy treated with valproate, especially affecting attention and memory functions.
Psychiatric use
Valproate is an effective mood stabilizer, licensed for the treatment of acute mania in patients with bipolar disorder. Although it has no formal indication, it is also considered a first- line agent for maintenance treatment in bipolar disorder. There is evidence suggesting efficacy of valproate in the treatment of hostility among patients with acute alcohol- associated hallucinosis or schizophrenia, and in impulsive/ aggressive behaviours, either in isolation or in the context of comorbid bipolar disorder or personality disorder. Available data show a limited efficacy of valproate in depressive disorders, schizophrenia, pathological gambling, as well as benzodiazepine/ cannabis/ cocaine dependence and acute alcohol withdrawal.
Description
Valproic acid and its salts are a new group of antiepileptic drugs that differs from the
known drugs both structurally and in terms of its mechanism of action. It is believed that
it acts on the metabolism of the GABA system. Valproic acid has been shown to elevate
the level of GABA in the brain by means of competitive inhibition of GABA transaminase
and the dehydrogenase of succinic semialdehyde.
This drug not only exhibits anticonvulsant action, but also betters the mental condition
of the patient.
Chemical Properties
Colorless Liquid
Originator
Depakote,Abbott
Uses
Antiepileptic; increases levels of -aminobutyric acid(GABA) in the brain. Anticonvulsant that also has efficacy as a mood stabilizer in bipolar disorder
Uses
For treatment and management of seizure disorders, mania, and prophylactic treatment of migraine headache. In epileptics, valproic acid is used to control absence seizures, tonic-clonic seizures (grand mal), complex partial seizures, and the seizures asso
Uses
2-Propylpentanoic acid has been used as a supplement in mouse embryonic fibroblast - conditioned medium (MEF-CM)?to feed the cells.
Uses
Antiepileptic; Anticonvulsant that also acts as a mood stabilizer for those with bipolar disorder.
Definition
ChEBI: A branched-chain saturated fatty acid that comprises of a propyl substituent on a pentanoic acid stem.
Manufacturing Process
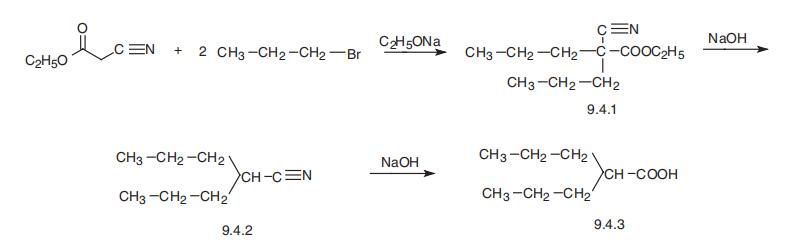
Dipropyl acetic acid or valproic acid may be prepared the next way.
Propylbromide is mixed with cyanacetic acid in the presence of sodium
ethylate, made from absolute ethanol and sodium. By that prepared α,α-
dipropylcyanacetic acid ethyl ester is saponified with equimolecular amounts
of NaOH to give dipropylacetonitril. The desired dipropylacetic acid is produced
by saponification of dipropylacetonitryl with aquatic NaOH. It is colorless
liquid. BP 219°-220°C.
Sodium salt of this acid may be prepared by adding of equivalent of NaOH.
brand name
Depakene (Abbott);Valproine;Vederon.
Therapeutic Function
Anticonvulsant
Biological Functions
Although it is marketed as both valproic acid
(Depakene) and as sodium valproate (Depakote), it is
the valproate ion that is absorbed from the gastrointestinal
tract and is the active form.
As with several other AEDs, it is difficult to ascribe
a single mechanism of action to valproic acid.This compound
has broad anticonvulsant activity, both in experimental
studies and in the therapeutic management of
human epilepsy.Valproic acid has been shown to block
voltage-dependent sodium channels at therapeutically
relevant concentrations. In several experimental studies,
valproate caused an increase in brain GABA; the
mechanism was unclear.There is evidence that valproate may also inhibit T-calcium channels and that this may
be important in its mechanism of action in patients with
absence epilepsy.
General Description
Clear colorless liquid.
General Description
VPA is an established AED with a simple chemical structurebut an unusually broad spectrum of action. It is generallywell tolerated, but its use is limited by two rare but significanttoxic side effects (hepatotoxicity and teratogenicity) thatcan be dose-dependent or idiosyncratic in nature.Thesedrawbacks are apparently shared by its equipotent activemetabolite, (E)-2-propyl-2-pentenoic acid (2-ene-VPA).
VPA is also an important inhibitor of the cytochrome P450isozymes, mainly of CYP2C9 and also of uridine diphosphate(UDP)-glucuronyl transferase and epoxide hydrolase.
Air & Water Reactions
Insoluble in water.
Reactivity Profile
2-Propylpentanoic acid is a carboxylic acid. Carboxylic acids donate hydrogen ions if a base is present to accept them. They react in this way with all bases, both organic (for example, the amines) and inorganic. Their reactions with bases, called "neutralizations", are accompanied by the evolution of substantial amounts of heat. Neutralization between an acid and a base produces water plus a salt. Carboxylic acids with six or fewer carbon atoms are freely or moderately soluble in water; those with more than six carbons are slightly soluble in water. Soluble carboxylic acid dissociate to an extent in water to yield hydrogen ions. The pH of solutions of carboxylic acids is therefore less than 7.0. Many insoluble carboxylic acids react rapidly with aqueous solutions containing a chemical base and dissolve as the neutralization generates a soluble salt. Carboxylic acids in aqueous solution and liquid or molten carboxylic acids can react with active metals to form gaseous hydrogen and a metal salt. Such reactions occur in principle for solid carboxylic acids as well, but are slow if the solid acid remains dry. Even "insoluble" carboxylic acids may absorb enough water from the air and dissolve sufficiently in 2-Propylpentanoic acid to corrode or dissolve iron, steel, and aluminum parts and containers. Carboxylic acids, like other acids, react with cyanide salts to generate gaseous hydrogen cyanide. The reaction is slower for dry, solid carboxylic acids. Insoluble carboxylic acids react with solutions of cyanides to cause the release of gaseous hydrogen cyanide. Flammable and/or toxic gases and heat are generated by the reaction of carboxylic acids with diazo compounds, dithiocarbamates, isocyanates, mercaptans, nitrides, and sulfides. Carboxylic acids, especially in aqueous solution, also react with sulfites, nitrites, thiosulfates (to give H2S and SO3), dithionites (SO2), to generate flammable and/or toxic gases and heat. Their reaction with carbonates and bicarbonates generates a harmless gas (carbon dioxide) but still heat. Like other organic compounds, carboxylic acids can be oxidized by strong oxidizing agents and reduced by strong reducing agents. These reactions generate heat. A wide variety of products is possible. Like other acids, carboxylic acids may initiate polymerization reactions; like other acids, they often catalyze (increase the rate of) chemical reactions. 2-Propylpentanoic acid is incompatible with bases, oxidizing agents and reducing agents. 2-Propylpentanoic acid is corrosive. .
Fire Hazard
2-Propylpentanoic acid is combustible.
Biochem/physiol Actions
Anticonvulsant that also has efficacy as a mood stabilizer in bipolar disorder
Mechanism of action
Although its mechanism of action is not clearly established, valproate appears to increase the inhibitory effect of GABA,
possibly by activation of glutamic acid decarboxylase or inhibition of GABA-transaminase). The high drug
concentrations required, however, cast doubt on the clinical relevance of this effect. Furthermore, valproate recently has been
shown to decrease the uptake of GABA into cultured astrocytes; this action may contribute to the AED efficacy. Valproate
is known to produce a blockade of high-frequency repetitive firing by slowing the rate of Na+
recovery from inactivation, a
mechanism consistent with the actions of phenytoin and CBZ. Valproate blocks the low-threshold T-type Ca2+ channel.
Consequently, the overall therapeutic utility of valproate is likely caused by multiple effects.
Valproate is indicated for initial or adjunct treatment of absence seizures or as an adjunct when absence seizures occur in
combination with either tonic-clonic seizures, myoclonic seizures, or both. For patients with unambiguous idiopathic generalized
epilepsy, valproate often is the drug of choice, because it controls absence, myoclonic, and generalized tonic-clonic seizures
well. It also is approved by U.S. FDA for use in complex partial seizures, occurring with or without other seizure types in
adults or children 10 years of age or older. In new patients with typical absence seizures, ethosuximide is preferred to
valproate because of the latter drug's risk of producing hepatotoxicity. In a comparative trial, sodium valproate and
ethosuximide were equally effective when either drug was given alone or in combination with other AEDs in children with typical
absence seizures. In atypical absence seizures (Lennox-Gastaut syndrome), sodium valproate is more effective, whereas in
myoclonic seizures, it is less effective than clonazepam. Valproate is approved by the U.S. FDA for use in bipolar disorder and
against migraine headaches.
Pharmacokinetics
Valproate undergoes rapid and complete absorption, which is only slightly slowed by food. It is 90% protein bound, and its clearance is dose-dependent because of an increase in the free fraction of the drug at higher doses. It is metabolized almost entirely by the liver, with 30 to 50% of an orally administered dose being eliminated in the urine as its acyl glucuronide conjugate, 40% from mitochondrial β-oxidation, approximately 15 to 20% by ω-oxidation, and less than 3% is excreted unchanged in urine. Its major active metabolite is (E)-2-ene valproate (trans 2-ene valproate). Its 4-ene metabolite has been proposed to be a reactive metabolite responsible for the hepatotoxicity of valproate. Other metabolites found in the urine include 3-oxo- and 4-hydroxyvalproate. The elimination half-life for valproate ranged from 9 to 16 hours following oral dosing regimens of 250 to 1,000 mg. Patients who are not taking enzyme-inducing AEDs (carbamazepine, phenytoin, and phenobarbital) will clear valproate more rapidly; therefore, monitoring of AED plasma concentrations should be intensified whenever concurrent AEDs are introduced or withdrawn.
Clinical Use
Valproic acid is well absorbed from the gastrointestinal
tract and is highly bound (~90%) to plasma protein,
and most of the compound is therefore retained
within the vascular compartment.Valproate rapidly enters
the brain from the circulation; the subsequent decline
in brain concentration parallels that in plasma, indicating
equilibration between brain and capillary
blood. A large number of metabolites have been identified,
but it is not known whether they play a role in the
anticonvulsant effect of the parent drug. Valproic acid
inhibits the metabolism of several drugs, including phenobarbital,
primidone, carbamazepine, and phenytoin,
leading to an increased blood level of these compounds.
At high doses, valproic acid can inhibit its own metabolism.
It can also displace phenytoin from binding sites
on plasma proteins, with a resultant increase in unbound
phenytoin and increased phenytoin toxicity. In
this instance, the dosage of phenytoin should be adjusted
as required. These examples reinforce the need
to determine serum anticonvulsant levels in epileptic
patients when polytherapy is employed.
Valproic acid has become a major AED against several
seizure types. It is highly effective against absence
seizures and myoclonic seizures. In addition, valproic
acid can be used either alone or in combination with
other drugs for the treatment of generalized tonic–
clonic epilepsy and for partial seizures with complex
symptoms.
Side effects
The most serious adverse effect associated with valproic
acid is fatal hepatic failure. Fatal hepatotoxicity is
most likely to occur in children under age 2 years, especially
in those with severe seizures who are given multiple
anticonvulsant drug therapy. The hepatotoxicity is
not dose related and is considered an idiosyncratic reaction;
it can occur in individuals in other age groups,
and therefore, valproic acid should not be administered
to patients with hepatic disease or significant hepatic
dysfunction or to those who are hypersensitive to it.
Valproic acid administration has been linked to an increased
incidence of neural tube defects in the fetus of
mothers who received valproate during the first
trimester of pregnancy. Patients taking valproate may
develop clotting abnormalities.
Valproic acid causes hair loss in about 5% of patients,
but this effect is reversible. Transient gastrointestinal
effects are common, and some mild behavioral
effects have been reported. Metabolic effects, including
hyperglycemia, hyperglycinuria, and hyperammonemia,
have been reported. An increase in body weight also
has been noted. Valproic acid is not a CNS depressant,
but its administration may lead to increased depression
if it is used in combination with phenobarbital, primidone,
benzodiazepines, or other CNS depressant agents.
Synthesis
Valproic acid, 2-propylvaleric acid (9.4.3), is synthesized by the alkylation of cyanoacetic ester with two moles of propylbromide, to give dipropylcyanoacetic ester (9.4.1). Hydrolysis and decarboxylation of the carbethyoxy group gives dipropylacetonitrile (9.4.2), which is hydrolyzed into valproic acid (9.4.3) [12¨C15].
Solubility in organics
soluble in most organic solvents, including methanol, chloroform, and ether, solubility in water: 1.27 mg/mL.
References
[1] phiel c j, zhang f, huang e y, et al. histone deacetylase is a direct target of valproic acid, a potent anticonvulsant, mood stabilizer, and teratogen. journal of biological chemistry, 2001, 276(39): 36734-36741.
[2] chateauvieux s, morceau f, dicato m, et al. molecular and therapeutic potential and toxicity of valproic acid. biomed research international, 2010, 2010.
2-Propylpentanoic acid Preparation Products And Raw materials
Raw materials
Preparation Products
| Supplier | Tel | Country | ProdList | Advantage | |
|---|---|---|---|---|---|
| Hebei Mojin Biotechnology Co., Ltd | +86 13288715578 +8613288715578 | sales@hbmojin.com | China | 12446 | 58 |
| Hebei Yanxi Chemical Co., Ltd. | +8617531190177 | peter@yan-xi.com | China | 5873 | 58 |
| Hebei Chuanghai Biotechnology Co,.LTD | +86-13131129325 | sales1@chuanghaibio.com | China | 5882 | 58 |
| Hangzhou Hyper Chemicals Limited | +86-0086-57187702781 +8613675893055 | info@hyper-chem.com | China | 295 | 58 |
| Henan Tianfu Chemical Co.,Ltd. | +86-0371-55170693 +86-19937530512 | info@tianfuchem.com | China | 21667 | 55 |
| Hefei TNJ Chemical Industry Co.,Ltd. | +86-0551-65418679 +8618949832763 | info@tnjchem.com | China | 2989 | 55 |
| career henan chemical co | +86-0371-86658258 +8613203830695 | sales@coreychem.com | China | 29888 | 58 |
| SHANDONG ZHI SHANG CHEMICAL CO.LTD | +86 18953170293 | sales@sdzschem.com | China | 2931 | 58 |
| Hebei Guanlang Biotechnology Co., Ltd. | +86-19930503282 | alice@crovellbio.com | China | 8820 | 58 |
| Chongqing Chemdad Co., Ltd | +86-023-6139-8061 +86-86-13650506873 | sales@chemdad.com | China | 39916 | 58 |
View Lastest Price from 2-Propylpentanoic acid manufacturers
| Image | Update time | Product | Price | Min. Order | Purity | Supply Ability | Manufacturer | |
|---|---|---|---|---|---|---|---|---|
 |
2024-09-23 | 2-Propylpentanoic acid
99-66-1
|
US $10.00 / KG | 1KG | 99.% | 10 ton | Hebei Chuanghai Biotechnology Co,.LTD | |
 |
2024-08-26 | 2-Propylpentanoic acid
99-66-1
|
US $0.00-0.00 / KG | 1KG | 99% | 500KG | Hangzhou Hyper Chemicals Limited | |
 |
2024-08-15 | valproic acid
99-66-1
|
US $0.00 / kg | 1kg | 0.99 | 20tons | Hebei Yanxi Chemical Co., Ltd. |
-

- 2-Propylpentanoic acid
99-66-1
- US $10.00 / KG
- 99.%
- Hebei Chuanghai Biotechnology Co,.LTD
-

- 2-Propylpentanoic acid
99-66-1
- US $0.00-0.00 / KG
- 99%
- Hangzhou Hyper Chemicals Limited
-

- valproic acid
99-66-1
- US $0.00 / kg
- 0.99
- Hebei Yanxi Chemical Co., Ltd.