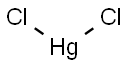염화수은(II) C화학적 특성, 용도, 생산
화학적 성질
Mercuric chloride,HgC12, is white crystals that are soluble in water and alcohol that melt at 276℃ and boil at 302℃. Highly toxic and corrosive, it is used in the manufacture of mercury compounds, in organic synthesis, as a reagent and catalyst, as a fungicide, insecticide, and wood preservative, and for many other purposes.
용도
Preserving (kyanizing) wood and anatomical specimens; also embalming; disinfecting; browning and etching steel and iron; intensifier in photography; white reserve in fabric printing; tanning leather; electroplating aluminum; depolarizer for dry batteries; freeing gold from lead; magic photograms; mordant for rabbit and beaver furs; staining wood and vegetable ivory pink; manufacture of ink for mercurography; treating seed potatoes; manufacture of other mercury Compounds. As an important reagent In animal chemistry.
정의
ChEBI: A mercury coordination entity made up of linear triatomic molecules in which a mercury atom is bonded to two chlorines. Water-soluble, it is highly toxic. Once used in a wide variety of applications, including preserving wood and anatomical specimens, emba
ming and disinfecting, as an intensifier in photography, as a mordant for rabbit and beaver furs, and freeing gold from lead, its use has markedly declined as less toxic alternatives have been developed.
일반 설명
An odorless white crystalline solid. Density 5.4 g / cm3. Melting point 277°C. Slightly volatile at ordinary temperatures. Can be sublimed unchanged. Corrosive to the mucous membranes. Toxic by inhalation (dusts, etc.), ingestion, and skin absorption. Used in photography, disinfectants, wood preservatives, fungicides.
공기와 물의 반응
Slightly soluble in water.
반응 프로필
Mercury chloride is decomposed by sunlight. Incompatible with formates sulfites, phpophosphites, phosphates, sulfides, albumin, gelatin, alkalis, alkaloid salts, ammonia, lime water, antimony, arsenic, bromides, borax, carbonates, reduced iron, iron, copper, lead and silver salts, infusions of cinchona, oak bark or senna, tannic acids and vegetable astringents. .
위험도
Toxic by ingestion, inhalation, and skin
absorption; a poison.
건강위험
Mercury chloride is classified as extremely toxic. All forms of mercury are poisonous if absorbed. Probable oral lethal dose is 5-50 mg/kg; between 7 drops and 1 teaspoonful for a 150 lb. person. Mercury chloride is one of the most toxic salts of mercury. Material attacks the gastrointestinal tract and renal systems.
화재위험
Material may explode on heating, with friction, or contact with alkali metals, sulfides, acetylene, ammonia, and oxalic acid. Upon decomposition highly toxic chloride and mercury fumes are emitted. Avoid formates, sulfites, hypophosphites, phosphates, sulfides, albumin, gelatin, alkalies, alkaloid salts, ammonia, lime water, antimony, arsenic, bromides, borax, carbonates, reduced iron, copper, iron, lead, silver salts, infusions of cinchona, columbo, oak bark or senna, and tannic acid. Mercury chloride may explode with friction or application of heat. Mixtures of Mercury chloride and sodium or potassium are shock sensitive and will explode on impact. Avoid contact with acids or acid fumes.
Safety Profile
A human poison by
ingestion. Poison experimentally by
ingestion, skin contact, and subcutaneous
routes. Human systemic effects by ingestion:
respiratory obstruction, nausea or vomiting,
urine volume decrease or anuria. Human
reproductive effects by ingestion: terminates
pregnancy. Experimental teratogenic and
reproductive effects. Human mutation data
reported. Questionable carcinogen. A severe
eye and skin irritant. Reaction with sodmm
aci-nitromethanide + acids forms the
explosive mercury fulminate. Reacts
violently with K, Na. When heated to
decomposition it emits toxic fumes of Hg.
잠재적 노출
Mercuric chloride is used as dip for
bulbs and tubers; for earthworm control; as repellent to
ants, roaches, etc.; in preserving wood and anatomical
specimens; embalming, browning, etching steel and iron;
as a catalyst for organic synthesis; disinfectant, antiseptic,
tanning; textile printing aid; manufacture of dyes; in
agricultural chemicals; dry batteries; pharmaceuticals, and
photographic chemicals
환경귀착
Mercury adsorbed from mercuric chloride and 2-methoxyethylmercury
chloride (Aretan) solutions by three contrasting
soils showed a dependence on soil–solution ratio and initial
mercury (Hg) concentration in soil solution. Changing the
soil–solution ratio from 1:10 to 1:100 but keeping the initial
concentration constant resulted in an increase in initial
concentration but, on the other hand, resulted in decrease in
Hg adsorption. Upon manipulation of the pH of the surface
soils, adsorption of mercuric chloride at 100 mg Hg l
-1
concentration increased from ~ 70 to over 95 mg Hg kg
-1
when the pH was raised from 5.0 to 8.0. Precipitation of Hg
may also have contributed to this trend. Removal of organic
matter from soil resulted in large reductions of Hg adsorbed, as
much as 95% from the mercuric chloride solutions. Mercuric
compounds found in the atmosphere are likely to be transformed
by chemical or physical processes. Theoretical calculations
on the photodissociation of mercuric compounds have
indicated that mercuric chloride and mercuric cyanide are
stable, while mercuric hydroxide may dissociate in the gas
phase. Exchange reactions between water and mercury
compounds are likely to occur in the atmosphere. These
exchange reactions eventually result in the release of elemental
mercury into the gaseous phase.
운송 방법
UN1624 Mercuric chloride, Hazard Class: 6.1;
Labels: 6.1-Poisonous materials.
Purification Methods
It is soluble in EtOH and is extracted into Et2O from an aqueous solution. It is very POISONOUS and 0.2-0.4g is fatal. The antidote is immediate administration of white of egg as an emetic.
비 호환성
A strong reducing agent; keep away
from oxidizers. Mercuric chloride may explode with friction or application on heat. Mixtures of mercuric chloride
and sodium or potassium are shock sensitive and will
explode on impact. Avoid contact with acids or acid fumes.
Also avoid the presence of formats, sulfites, hypophosphites, phosphates, sulfide; albumin, gelatin, alkalies,
alkaloid salts; ammonia, lime water; antimony, arsenic,
bromides, borax, carbonates, reduced iron, copper; iron,
lead, silver salts; infusions of cinchona; columbo, oak bark
or senna; and tannic acid
염화수은(II) 준비 용품 및 원자재
원자재
준비 용품