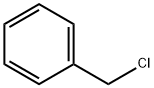
염화벤질
|
|
염화벤질 속성
- 녹는점
- -39 °C
- 끓는 점
- 179 °C
- 밀도
- 1.1 g/mL at 25 °C(lit.)
- 증기 밀도
- 4.36 (vs air)
- 증기압
- 10.3 mm Hg ( 60 °C)
- 굴절률
- n
20/D 1.538(lit.)
- 인화점
- 165 °F
- 저장 조건
- Store below +30°C.
- 용해도
- 용해성0.46g/L @ 30°C (물과 접촉 시 분해)
- 물리적 상태
- 액체
- 색상
- 무색투명~미황색
- 냄새
- 자극적이고 자극적입니다.
- 폭발한계
- 1.1-14%(V)
- 수용성
- 0.3g/L(20℃)
- Merck
- 14,1129
- BRN
- 471308
- Henry's Law Constant
- (x 10-4 atm?m3/mol): 3.57 at 20.00 °C (inert gas stripping, Hovorka and Dohnal, 1997)
- 노출 한도
- TLV-TWA 1 ppm (~5mg/m3) (ACGIH, MSHA, and OSHA); IDLH 10 ppm (NIOSH); carcinogenicity: Animal Limited Evidence, Human Inadequate Evidence (IARC).
- Dielectric constant
- 7.0(13℃)
- 안정성
- Unstable - inhibitors such as propylene oxide or trimethylamine are usually added to prevent polymerization. Combustible. Incompatible with strong oxidizing agents, water, acids, most common metals, dimethyl sulfoxide. Above flash point vapour-air mixtures are explosive within the limits noted above. Contact with water produces toxic fumes.
- InChIKey
- KCXMKQUNVWSEMD-UHFFFAOYSA-N
- LogP
- 2.3 at 20℃ and pH7
- CAS 데이터베이스
- 100-44-7(CAS DataBase Reference)
- IARC
- 2A (Vol. 29, Sup 7, 71) 1999
안전
- 위험 및 안전 성명
- 위험 및 사전주의 사항 (GHS)
| 위험품 표기 | T,T+ | ||
|---|---|---|---|
| 위험 카페고리 넘버 | 45-22-23-37/38-41-48/22-43-26-46 | ||
| 안전지침서 | 53-45-36/37/39-28-26-36/37 | ||
| 유엔번호(UN No.) | UN 1738 6.1/PG 2 | ||
| OEL | Ceiling: 1 ppm (5 mg/m3) [15-minute] | ||
| WGK 독일 | 3 | ||
| RTECS 번호 | XS8925000 | ||
| F 고인화성물질 | 8-19 | ||
| 자연 발화 온도 | 585 °C | ||
| TSCA | Yes | ||
| 위험 등급 | 6.1 | ||
| 포장분류 | II | ||
| HS 번호 | 29039990 | ||
| 유해 물질 데이터 | 100-44-7(Hazardous Substances Data) | ||
| 독성 | LD50 orally in Rabbit: 440 mg/kg | ||
| IDLA | 10 ppm | ||
| 기존화학 물질 | KE-05729 | ||
| 중점관리물질 필터링 | 별표1-48 | ||
| 사고대비 물질 필터링 | 21 |
염화벤질 C화학적 특성, 용도, 생산
물성
액체(무색 ~ 노란색)제품 소개
녹는점/어는점:-39℃,초기끓는점/끓는점범위:179℃,인화점:67℃.화학적 성질
Benzyl chloride is a colorless to s lightly yellow liquid with a strong, unpleasant, irritating odor. The Odor Threshold is 0.05 ppm.The stabilized form of benzyl chloride contains a fixed amount of a sodium carbonate solution or propylene oxide.
물리적 성질
Colorless to pale yellowish-brown liquid with a pungent, aromatic, irritating odor. Odor threshold concentration is 47 ppbv (Leonardos et al., 1969). Katz and Talbert (1930) reported an experimental detection odor threshold concentration of 210 μg/m3 (41 ppbv). The solubility of benzyl chloride in water is 0.33 g/L at 4°C, 0.49 g/L at 20°C, and 0.55 g/L at 30°C. It is freely soluble in chloroform, acetone, acetic acid esters, diethyl ether, and ethyl alcohol.용도
Benzyl chloride is used in the manufacture of benzyl Compounds, dyes, artificial resins, tanning agents, phar maceuticals, plasticizers, synthetic tannins, perfumes, lubricants, and quaternary ammonium compounds. It is also an intermediate in the preparation of phenylacetic acid (precursor to phamaceuticals).생산 방법
Benzyl chloride can be synthesized by chloromethylation of benzene in the presence of a catalyst (ZnCl2) or by treatment of benzyl alcohol with SO2Cl2. Commercially it is produced by chlorination of boiling toluene in the presence of light.주요 응용
Benzyl chloride is used mainly to produce plasticizers (e.g., benzyl butyl phthalate), benzyl alcohol, and phenylacetic acid via benzyl cyanide (used in the production of synthetic penicillin). On a smaller scale, it is used to produce quaternary ammonium salts (for disinfectants and phase-transfer catalysts), benzyl esters (benzyl benzoate and benzyl acetate for the flavors and perfumes industry), dyes of the triphenylmethane series, dibenzyl disulfide (antioxidant for lubricants), benzylphenol, and benzylamines.정의
ChEBI: Benzyl chloride is a member of the class of benzyl chlorides that is toluene substituted on the alpha-carbon with chlorine.제조 방법
Benzyl chloride is the starting material for a large number of industrial syntheses. The first preparation of it involved not the chlorination of toluene, however, but the reaction of benzyl alcohol with hydrochloric acid (S. Cannizzaro, 1853). Today, most side-chain chlorination of toluene to produce benzyl chloride. Dibenzyl ether is formed as a byproduct in the alkaline hydrolysis of benzyl chloride to benzyl alcohol. This ether can be re-converted to benzyl chloride by cleavage with hydrogen chloride at a temperature below 100°C.화학 반응
Benzyl chloride can be oxidized to benzoic acid or benzaldehyde, or substituted to give the halogenated, sulfonated or nitrated product.With NH3 it yields mono-, di- or tribenzyl amine. With alcohols in base the benzylalkyl ether is formed. With phenols either the phenolic or nuclear hydrogens can react to give benzylaryl ether or benzylated phenols. Reaction with NaCN gives benzyl cyanide (phenylacetonitrile); with aliphatic primary amines the product is the N-alkylbenzylamine, and with aromatic primary amines N-benzylaniline is formed. Benzyl chloride is converted to butyl benzyl phthalate plasticizer and other chemicals.일반 설명
Benzyl chloride appears as a colorless liquid with an irritating odor. Toxic by inhalation and skin absorption. Flash point 153°F. Slightly soluble in water. Corrosive to metals and tissue. A lachrymator. Density 9.2 lb /gal.공기와 물의 반응
A lachrymator. Slightly soluble in water.반응 프로필
Halogenated aliphatic compounds, such as Benzyl chloride, are moderately or very reactive. Reactivity generally decreases with increased degree of substitution of halogen for hydrogen atoms. Materials in this group are incompatible with strong oxidizing and reducing agents. Also, they are incompatible with many amines, nitrides, azo/diazo compounds, alkali metals, and epoxides.위험도
Highly toxic, intense eye and skin irritant. A lachrymator. Upper respiratory tract irritant. Prob- able carcinogen.건강위험
Benzyl chloride is a corrosive liquid. Con tact with the eyes can cause corneal injury.Exposure to its vapors can produce intenseirritation of the eyes, nose, and throat. Highconcentrations may cause lung edema anddepression of the central nervous system.Flury and Zernik (1931) stated that exposureto 16 ppm for 1 minute was intolerable tohumans. The LC50 values for a 2-hour expo sure in mice and rats are 80 and 150 ppm,respectively. The subcutaneous LD50 valuein rats is 1000 mg/kg (NIOSH 1986).Benzyl chloride tested positive to thehistidine reversion–Ames test for mutagenic ity. Subcutaneous administration of this com pound in laboratory animals caused tumors atthe site of application.
?? ??
A concentration of 16 ppm of benzyl chloride in air is reported to be intolerable to humans within 1 min. The compound is a potent lachrymator, strongly irritating to the eyes, nose, and throat and capable of causing lung edema.화재위험
Benzyl chloride burns but does not ignite readily. Benzyl chloride may ignite combustibles. When heated to decomposition, Benzyl chloride emits toxic and corrosive fumes. Some organic chlorides decompose to yield phosgene. Incompatible with active metals such as copper, aluminum, magnesium, iron, zinc, and tin and keep from strong oxidizing agents. Avoid contact with acids or acid fumes. Keep separate from oxidizing materials. May become unstable at elevated temperatures and pressures; may react with water resulting in some nonviolent release of energy. Polymerizes with evolution of heat and hydrogen chloride when in contact with all common metals except nickel and lead.Toxicology
The acute oral toxicity (LD50) of benzyl chloride in rats is 1231 mg/kg and in mice 1624 mg/kg. The subcutaneous LD50 (in rats) of benzyl chloride in oil solution is 1000 mg/kg. Exposure of rats and mice to benzyl chloride concentrations of 100 –1000 mg/m3 for 2 h caused irritation of the mucous membranes and conjunctivitis, vol. 11. Benzyl chloride is a strong skin-sensitizing agent for guinea pigs . Benzyl chloride acts weakly mutagenic in validated test systems. Subcutaneous injection of weekly doses of 80 mg/kg for 1 year followed by a post-observation period resulted in local sarcomas with lung metastases in rats. The mean induction time was 500 d. After dermal application of benzyl chloride, skin carcinomas were observed in mice.잠재적 노출
Used as an intermediate and as an irritant gas in chemical warfare. In contrast to phenyl halides, benzyl halides are very reactive. Benzyl chloride is used in production of benzal chloride, benzyl alcohol, and benzaldehyde. Industrial usage includes the manufacture of benzyl compounds, cosmetics, dyes, plastics, synthetic tannins, perfumes and resins. It is used in the manufacture of many pharmaceuticals. Suggested uses of benzyl chloride include: the vulcanization of fluororubbers and the benzylation of phenol and its derivatives for the production of possible disinfectants.Carcinogenicity
Benzyl chloride caused genetic mutations and chromosome-damaging effects in a wide variety of in vitro assays; it was not mutagenic in vivo in the mouse micronucleus assay환경귀착
Biological. When incubated with raw sewage and raw sewage acclimated with hydrocarbons, benzyl chloride degraded forming nonchlorinated products (Jacobson and Alexander, 1981).Chemical/Physical. Anticipated products from the reaction of benzyl chloride with ozone or OH radicals in the atmosphere are chloromethyl phenols, benzaldehyde and chlorine radicals (Cupitt, 1980).
Slowly hydrolyzes in water forming HCl and benzyl alcohol. The estimated hydrolysis half-life in water at 25 °C and pH 7 is 15 h (Mabey and Mill, 1978). The hydrolysis rate constant for benzyl chloride at pH 7 and 59.2 °C was determined to be 0.0204/min, resulting in a half-life of 34 min (Ellington et al., 1986).
May polymerize in contact with metals except nickel and lead (NIOSH, 1997).
When heated to decomposition, hydrogen chloride gas may be released (CHRIS, 1984).
운송 방법
UN1738 Benzyl chloride, Hazard class: 6.1; Labels: 6.1—Poisonous materials, 8—Corrosive material.Purification Methods
Dry it with MgSO4 or CaSO4, or reflux it with fresh Ca turnings, then fractionally distil it under reduced pressure, collecting the middle fraction and storing it over CaH2 or P2O5. It has also been purified by passage through a column of alumina. Alternatively it is dried over MgSO4 and distilled in a vacuum. The middle fraction is degassed by several freeze-thaw cycles and then fractionated in an 'isolated fractionating column' (which has been evacuated and sealed off at ~10-6 mm) over a steam bath. The middle fraction is retained. The final samples are distilled in a vacuum from this sample and again retaining the middle fraction. The purity is >99.9% (no other peaks are visible by GLC, and the NMR spectrum is consistent with the structure. [Mohammed & Kosower J Am Chem Soc 93 1709 1971, Beilstein 5 IV 809.] IRRITANT and strongly LACHRYMATORY.비 호환성
May form explosive mixture with air. Contact with water forms hydrogen chloride fumes. Strong oxidizers may cause fire and explosions. Unstabilized benzyl chloride undergoes polymerization with copper, aluminum, iron, zinc, magnesium, tin, and other common metals except lead and nickel, with the liberation of heat and hydrogen chloride gas. May accumulate static electrical charges, and may cause ignition of its vapors. Attacks some plastics and rubber. Thermal decomposition and polymerization reactions are inhibited, to a limited extent, by addition of triethylamine, propylene oxide, or sodium carbonate.폐기물 처리
Incineration @ 816 C for 0.5 second minimum for primary combustion and 1204 C for 12.0 second for secondary combustion. Elemental chlorine formation may be alleviated by injection of steam or methane into the combustion process.염화벤질 준비 용품 및 원자재
원자재
준비 용품
2-Benzyl-2-thiopseudourea hydrochloride
벤질 뷰틸산
염화벤잘코늄
4-(아세트아미도메틸)벤젠술포닐클로라이드
4-BENZYLOXY-2-NITROTOLUENE
에틸5-벤질록시인돌-2-카르복실레이트
Fast Scarlet Base LG
3-Benzyloxyphenol
디벤질 디설파이드
Meclozine
벤질부틸프탈레이트
암모늄O,O-디이소프로필티오포스페이트
염화 벤질디메틸옥타데실암모늄
N-벤질안트라닐산
살리실산벤질
N-Benzylcinchonidinium chloride
에틸벤젤아닐린
염화트리메틸벤질암모늄
S-Benzyl-L-cysteine
비벤질
4-벤질피리딘
염화 벤질다이메틸테트라데실암모늄
pesticide emulsifier BCH
azelaic acid, compound with morpholine
benzyltrimeehyl ammonium chloride
softener BCH
softener DOD
leucotrope O
벤질 3-페닐프로펜산
프로피온산 벤질
Levelling agent S
디벤질에테르
2,3-DIPHENYLPROPIONIC ACID
1-Benzyloxy-2-methoxy-4-propenylbenzene
1-벤질-3-하이드록시-1H-인다졸
4-(페닐메톡시)아조벤젠
sodium dibenzyl amine enzene sulfonate
4-메틸-1-페닐-2-펜타놀
디벤질프탈레이트
6-benzylaminopurine hydrochloride
염화벤질 공급 업체
글로벌( 673)공급 업체
| 공급자 | 전화 | 이메일 | 국가 | 제품 수 | 이점 |
|---|---|---|---|---|---|
| SIMAGCHEM CORP | +86-13806087780 |
sale@simagchem.com | China | 17367 | 58 |
| Shanxi Naipu Import and Export Co.,Ltd | +86-13734021967 +8613734021967 |
kaia@neputrading.com | China | 1011 | 58 |
| Wuhan Biet Co., Ltd. | +8617320528784 |
min@biet.com.cn | China | 41 | 58 |
| Yujiang Chemical (Shandong) Co.,Ltd. | +86-17736087130 +86-18633844644 |
catherine@yjchem.com.cn | China | 983 | 58 |
| SHANGHAI KEAN TECHNOLOGY CO., LTD. | +8613817748580 |
cooperation@kean-chem.com | China | 40068 | 58 |
| Hebei Mojin Biotechnology Co., Ltd | +86 13288715578 +8613288715578 |
sales@hbmojin.com | China | 12446 | 58 |
| Hebei Chuanghai Biotechnology Co,.LTD | +86-13131129325 |
sales1@chuanghaibio.com | China | 5892 | 58 |
| Henan Fengda Chemical Co., Ltd | +86-371-86557731 +86-13613820652 |
info@fdachem.com | China | 20291 | 58 |
| Hangzhou FandaChem Co.,Ltd. | 008657128800458; +8615858145714 |
fandachem@gmail.com | China | 9308 | 55 |
| ATK CHEMICAL COMPANY LIMITED | +undefined-21-51877795 |
ivan@atkchemical.com | China | 32836 | 60 |