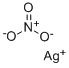질산 은
|
|
질산 은 속성
- 녹는점
- 212 °C (dec.) (lit.)
- 끓는 점
- 444°C
- 밀도
- 4.35 g/mL at 25 °C(lit.)
- 증기 밀도
- 5.8 (vs air)
- 증기압
- 17.535 mm of Hg (@ 20°C)
- 인화점
- 40 °C
- 저장 조건
- 2-8°C
- 용해도
- H2O: 용해성
- 물리적 상태
- 고체
- 물리적 상태
- 단단한 모양
- 색상
- 하얀색
- 냄새
- 냄새 없는
- pH 범위
- 7 - 9
- 수소이온지수(pH)
- 5.4-6.4 (100g/l, H2O, 20℃)
- 수용성
- 219g/100mL(20℃)
- 감도
- Light Sensitive
- Merck
- 14,8518
- 노출 한도
- ACGIH: TWA 0.01 mg/m3
NIOSH: IDLH 10 mg/m3; TWA 0.01 mg/m3
- InChIKey
- SQGYOTSLMSWVJD-UHFFFAOYSA-N
- CAS 데이터베이스
- 7761-88-8(CAS DataBase Reference)
안전
- 위험 및 안전 성명
- 위험 및 사전주의 사항 (GHS)
| 위험품 표기 | C,O,N,Xi | ||
|---|---|---|---|
| 위험 카페고리 넘버 | 34-50/53-8-36/38-51/53-52/53-35-10-40-20/22-22 | ||
| 안전지침서 | 26-45-60-61-36/37/39-27-57-37 | ||
| 유엔번호(UN No.) | UN 1493 5.1/PG 2 | ||
| WGK 독일 | 3 | ||
| RTECS 번호 | VW4725000 | ||
| F 고인화성물질 | 8 | ||
| TSCA | Yes | ||
| 위험 등급 | 5.1 | ||
| 포장분류 | II | ||
| HS 번호 | 28432100 | ||
| 유해 물질 데이터 | 7761-88-8(Hazardous Substances Data) | ||
| 독성 | LD50 oral in rat: 1173mg/kg | ||
| 기존화학 물질 | KE-31281 | ||
| 유해화학물질 필터링 | 97-1-92 | ||
| 함량 및 규제정보 | 물질구분: 유독물질; 혼합물(제품)함량정보: 무기은 염류 및 이를 25% 이상 함유한 혼합물. 다만, 염화 은(Silver chloride) 및 뇌산 은(Silver fulminate)은 제외 |
질산 은 C화학적 특성, 용도, 생산
화학적 성질
Silver nitrate, AgN03, is colorless,transparent,tabular,rhombic crystals that become gray or grayish-black on exposure to light in the presence of organic matter.It is odorless with a bitter,caustic,metallic taste. It is caustic,and a strong oxidizing agent that is soluble in cold water, more soluble in hot water, glycerol,and hot alcohol,slightly soluble in ether,and decomposes at boiling point Used in photographic film, silver plating,silvering mirrors,and as an antiseptic.물리적 성질
Colorless, transparent, large rhombohedral crystals, or white small crystals; bitter, caustic metallic taste; odorless; pure compound is not sensitive to light but trace organics promote photo reduction, turning the salt to grayish black on exposure to light; density 4.35 g/cm3; melts at 212°C; decomposes at 440°C; very soluble in water, soluble in ethanol and acetone.용도
The basis of nearly all photographic silver halides with the exception of the daguerreotype process, silver nitrate is a heavy white crystal made by dissolving elemental silver in nitric acid followed by evaporation. It is soluble in water, ether, and glycerin. Silver nitrate is not sensitive to light, but when combined with an organic material, a halogen, or a halide it will reduce back to a metallic state when exposed to light.Indications
Silver nitrate, 0.1% to 0.5%, is an excellent germicide and astringent. Its germicidal action is due to precipitation of bacterial protein by liberated silver ions. It may cause pain if applied in concentrations >0.5%.Silver nitrate is another cauterizing agent and coagulates cellular protein and removes granulation tissue. This should be applied everyday for approximately 5 days." "Silver nitrate (AgNO3), in solid form or in solutions stronger than 5%, is used for its caustic action; 5% to 10% solutions may be applied to fissures or excessive granulation tissue. Silver nitrate sticks consist of a head of toughened silver nitrate (>94.5%) prepared by fusing the silver salt with sodium chloride. They are dipped in water and applied as needed.
제조 방법
Silver nitrate is prepared by dissolving silver metal in dilute nitric acid. The solution is evaporated and residue is heated to dull red heat with concentrated nitric acid to decompose impurities such as copper nitrate. Residue then is dissolved in water, filtered, and recrystallized to obtain pure silver nitrate.일반 설명
A colorless or white crystalline solid becoming black on exposure to light or organic material.공기와 물의 반응
Water soluble.반응 프로필
Silver nitrate is noncombustible but, as an oxidizing agent, can accelerate the burning of combustible materials. If large quantities are involved in a fire or the combustible material is finely divided, an explosion may result. Prolonged exposure to fire or heat may result in an explosion. Toxic oxides of nitrogen are produced in fires. Light sensitive. Mixtures with alkyl esters may explode owing to the formation of alkyl nitrates. Mixtures with phosphorus, tin(II) chloride, or other reducing agents may react explosively [Bretherick 1979 p. 108-109]. Reacts with acetylene in the presence of ammonia to form silver acetylide, a powerful detonator when dry [Bretherick 1979 p. 198]. Reaction with ethyl alcohol (or other alcohols) may produce silver fulminate, which can explode when disturbed [Bretherick 1979 p. 200]. An intimate mixture of Silver nitrate and magnesium may ignite spontaneously on contact with a drop of water [Bretherick 1979 p. 200]. An explosion occurred when purified phosphine was passed rapidly into a concentrated solution of Silver nitrate [Mellor 3:471 1946-47]. When a mixture of 28% ammonium hydroxide and Silver nitrate solution was treated with a small amount of sodium hydroxide. Black precipitate, silver nitride exploded on stirring [MCA Case History 1554 1968].위험도
Strong irritant to skin and tissue.건강위험
Concentrated solutions will produce irritation, ulceration, and discoloration of the skin; also causes severe irritation of the eyes. Ingestion will produce violent abdominal pain and other gastroenteric symptoms.화재위험
Behavior in Fire: Increases flammability of combustibles.Pharmaceutical Applications
Silver nitrate (AgNO3), after salicylic acid, is widely used for the treatment of warts. AgNO3 is a highly water-soluble salt, which readily precipitates as AgCl, black in colour, when in contact with the skin. Warts are caused by a human papillomavirus, and mostly hands, feet and the anogenital areas are affected. The treatment is based on the destruction of the local tissue, and the silver salt is applied via a caustic pen to the affected area. Silver nitrate is highly corrosive and is known to destroy these types of tissue growth. Care has to be taken when this treatment option is used, as the resulting AgCl stains any skin or fabric which it has been in contact with.Safety Profile
A human poison. Experimental poison by ingestion, intravenous, subcutaneous, and intraperitoneal routes. Experimental reproductive effects. Human mutation data reported. A severe eye irritant. A powerful caustic and irritant to skin, eyes, and mucous membranes. Swallowing can cause severe gastroenteritis that may be fatal. Questionable carcinogen with experimental tumorigenic data. A powerful oxidizer. Incompatible with acetylene, acetylides, alkalies, aluminum, antimony salts, arsenic, arsenites, bromides, carbon, carbonates, chlorides, ClF3, chlorosulfuric acid, copper, creosote, ethanol, ferrous salts, hypophosphites, iodides, Mg powder with H20, morphme salts, NH3 with KOH to yield black Ag3N, oils, PH3, phosphates, phosphonium iodide, phosphorus, plastics, sulfur, tannic acid, tartrates, thiocyanates, vegetable decoctions and extracts, zinc with NH3 with KOH. When heated to decomposition it emits toxic fumes of NOx. See also SILVER COMPOUNDS and NITRATES잠재적 노출
Silver nitrate is used in photography, silver plating; as an antiseptic; in chemical reactions; and mirror manufacturing; as starting material in production of other silver compounds.운송 방법
UN1493 Silver nitrate, Hazard Class: 5.1; Labels: 5.1-Oxidizer.Purification Methods
Purify it by recrystallisation from hot water (solubility of AgNO3 in water is 992g/100mL at 100o and 122g/100mL at 0o). It has also been purified by crystallisation from hot conductivity water by slow addition of freshly distilled EtOH. CAUTION: avoid using EtOH for washing the precipitate; and avoid concentrating the filtrate to obtain further crops of AgNO3 owing to the risk of EXPLOSION (as has been reported to us) caused by the presence of silver fulminate. When using EtOH in the purification, the apparatus should be enveloped in a strong protective shield. [Tully, News Ed (Am Chem Soc) 19 3092 1941; Garin & Henderson J Chem Educ 47 741 1970, Bretherick, Handbook of Reactive Chemical Hazards 4th edn, Butterworths, London, 1985, pp 13-14.] Before being used as a standard in volumetric analysis, analytical reagent grade AgNO3 should be finely powdered, dried at 120o for 2hours, then cooled in a desiccator. Recovery of silver residues as AgNO3 [use protective shield during the whole of this procedure] can be achieved by washing with hot water and adding 16M HNO3 to dissolve the solid. Filter this through glass wool and concentrate the filtrate on a steam bath until precipitation commences. Cool the solution in an ice-bath and filter the precipitated AgNO3. Dry it at 120o for 2hours, then cool it in a desiccator in a vacuum. Store it over P2O5 in a vacuum in the dark. AVOID contact with hands due to formation of black stains.비 호환성
A strong oxidizer. Reacts violently with combustible and reducing materials. Reacts with acetylene forming a shock-sensitive explosive. Reacts with alkalis, antimony salts; ammonia, arsenites, bromides, carbonates, chlorides, iodides, hydrogen peroxide; thiocyanates, ferrous salts; oils, hypophosphites, morphine salts; creosote, phosphates, tannic acid; tartarates, halides, vegetable extracts, and others. Attacks some forms of plastics, rubber, and coatings.질산 은 준비 용품 및 원자재
원자재
준비 용품
2-TERT-BUTYLPYRIDINE-4-CARBONITRILE
프탈라미산
Docetaxel
2-tert-Butylpyridine-4-carboxylic acid ,97%
Oxaliplatin
Acetylpyrazine
Faropenem sodium hemipentahydrate
1-(BROMOMETHYL)ISOQUINOLINE HYDROBROMIDE
2-(Bromomethyl)benzoic acid
UV-photoxidation degradation film containing FeDBC photosensitize
1-ETHYL-3-METHYLIMIDAZOLIUM DICYANAMIDE
에틸렌디아민테트라아세트산, 테트라나트륨 염
2-Methyl-2-pentenoic acid
은(I) 산화물
3-FLUOROQUINOLINE-4-CARBOXYLIC ACID
칼륨 은 시안화물
황산은
Prostaglandin E1
1,3-Adamantanediol
시안화은
tert-부틸나이트라이트
5-Phenylthiophene-2-carboxylic acid