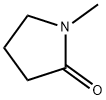
N-메틸-2-피롤리디논
|
|
N-메틸-2-피롤리디논 속성
- 녹는점
- -24 °C (lit.)
- 끓는 점
- 202 °C (lit.) 81-82 °C/10 mmHg (lit.)
- 밀도
- 1.028 g/mL at 25 °C (lit.)
- 증기 밀도
- 3.4 (vs air)
- 증기압
- 0.29 mm Hg ( 20 °C)
- 굴절률
- n
20/D 1.479
- 인화점
- 187 °F
- 저장 조건
- Store at +5°C to +30°C.
- 용해도
- 에탄올: 혼화성0.1ML/mL, 투명, 무색 (10%, v/v)
- 물리적 상태
- 액체
- 산도 계수 (pKa)
- -0.41±0.20(Predicted)
- 색상
- 20(APHA)
- 냄새
- 약간의 아민 냄새
- pH 범위
- 7.7 - 8.0
- 수소이온지수(pH)
- 8.5-10.0 (100g/l, H2O, 20℃)
- 폭발한계
- 1.3-9.5%(V)
- 수용성
- 20°C에서 >=10g/100mL
- 감도
- Hygroscopic
- 최대 파장(λmax)
- 283nm(MeOH)(lit.)
- Merck
- 14,6117
- BRN
- 106420
- Dielectric constant
- 32.200000000000003
- 안정성
- 안정적이지만 빛에 노출되면 분해됩니다. 타기 쉬운. 강산화제, 강산, 환원제, 염기와 호환되지 않습니다.
- InChIKey
- SECXISVLQFMRJM-UHFFFAOYSA-N
- LogP
- -0.46 at 25℃
- CAS 데이터베이스
- 872-50-4(CAS DataBase Reference)
안전
- 위험 및 안전 성명
- 위험 및 사전주의 사항 (GHS)
| 위험품 표기 | T,Xi | ||
|---|---|---|---|
| 위험 카페고리 넘버 | 45-65-36/38-36/37/38-61-10-46 | ||
| 안전지침서 | 41-45-53-62-26 | ||
| WGK 독일 | 1 | ||
| RTECS 번호 | UY5790000 | ||
| F 고인화성물질 | 3-8-10 | ||
| 자연 발화 온도 | 518 °F | ||
| TSCA | Y | ||
| HS 번호 | 2933199090 | ||
| 유해 물질 데이터 | 872-50-4(Hazardous Substances Data) | ||
| 독성 | LD50 orally in Rabbit: 3598 mg/kg LD50 dermal Rabbit 8000 mg/kg | ||
| 기존화학 물질 | KE-25324 | ||
| 유해화학물질 필터링 | 2014-1-700 | ||
| 중점관리물질 필터링 | 별표1-93 | ||
| 함량 및 규제정보 | 물질구분: 유독물질; 혼합물(제품)함량정보: 1-메틸-2-피롤리디논 및 이를 0.3% 이상 함유한 혼합물 |
N-메틸-2-피롤리디논 C화학적 특성, 용도, 생산
개요
N-Methyl-2-pyrrolidone is an aprotic solvent with a wide range of applications: petrochemical processing, surface coating, dyes and pigments, industrial and domestic cleaning compounds, and agricultural and pharmaceutical formulations. It is mainly an irritant, but has also caused several cases of contact dermatitis in a small electrotechnical company.화학적 성질
N-Methyl-2-pyrrolidone is a colourless or light yellow liquid with an amine odour. It can undergo a number of chemical reactions even though it is accepted as a stable solvent. It is resistant to hydrolysis under neutral conditions, but strong acid or base treatment results in ring opening to 4-methyl aminobutyric acid. N-Methyl-2-pyrrolidone can be reduced to 1-methyl pyrrolidine with borohydride. Treatment with chlorinating agents results in amide formation,an intermediate which can undergo further substitution, while treatment with amyl nitrate yields the nitrate. Olefins can be added to the 3 position by treatment first with oxalic esters, then with appropriate aldehyes (Hort and Anderson 1982).
용도
N-Methyl-2-pyrrolidone is a polar solvent that is used in organic chemistry and polymer chemistry. Large scale applications include the recovery and purification of acetylenes, olefins, and diolefins, gas purification, and aromatics extraction from feedstocks.N-Methyl-2-pyrrolidone is a versatile industrial solvent. NMP is currently approved for use only in veterinary pharmaceuticals. The determination of the disposition and metabolism of NMP in the rat will contribute toward understanding the toxicology of this exogenous chemical which man may likely be exposed to in increasing amounts.생산 방법
N-Methyl-2-pyrrolidone is manufactured by the reaction of buytrolactone with methylamine (Hawley 1977). Other processes include preparation by hydrogenation of solutions of maleic or succinic acids with methylamine (Hort and Anderson 1982). Manufacturers of this chemical include Lachat Chemical, Inc, Mequon, Wisconsin and GAF Corporation, Covert City, California.정의
ChEBI: N-methylpyrrolidin-2-one is a member of the class of pyrrolidine-2-ones that is pyrrolidin-2-one in which the hydrogen attached to the nitrogen is replaced by a methyl group. It has a role as a polar aprotic solvent. It is a N-alkylpyrrolidine, a lactam and a member of pyrrolidin-2-ones.일반 설명
N-Methyl-2-Pyrrolidone (NMP) is a powerful, aprotic solvent with high solvency, and low volatility. This colorless, high boiling, high flash point and low vapor pressure liquid carries a mild amine-like odor. NMP has high chemical and thermal stability and is completely miscible with water at all temperatures. NMP can serve as a co-solvent with water, alcohols, glycol ethers, ketones, and aromatic/chlorinated hydrocarbons. NMP is both recyclable by distillation and readily biodegradable. NMP is not found on the Hazardous Air Pollutants (HAPs) list of the 1990 Clean Air Act Amendments.공기와 물의 반응
Soluble in water.반응 프로필
This amine is a very mild chemical base. N-Methyl-2-pyrrolidone does tend to neutralize acids to form salts plus water. The amount of heat that is evolved per mole of amine in a neutralization is largely independent of the strength of the amine as a base. Amines may be incompatible with isocyanates, halogenated organics, peroxides, phenols (acidic), epoxides, anhydrides, and acid halides. Flammable gaseous hydrogen is generated by amines in combination with strong reducing agents, such as hydrides.위험도
Severe skin and eye irritant. Explosive lim-its 2.2–12.2%.건강위험
Inhalation of hot vapors can irritate nose and throat. Ingestion causes irritation of mouth and stomach. Contact with eyes causes irritation. Repeated and prolonged skin contact produces a mild, transient irritation.화재위험
Special Hazards of Combustion Products: Toxic oxides of nitrogen may be formed in fire.공업 용도
1) N-Methyl-2-pyrrolidone is used as a general dipolar aprotic solvent, stable and unreactive;2) for extraction of aromatic hydrocarbons from lubricating oils;
3) for carbon dioxide removal in ammonia generators;
4) as a solvent for polymerization reactions and polymers;
5) as a paint stripper;
6) for pesticide formulations (USEPA 1985).
Other non-industrial uses of N-Methyl-2-pyrrolidone are based on its properties as a dissociating solvent suitable for electrochemical and physical chemical studies (Langan and Salman 1987). Pharmaceutical applications make use of the properties of N-Methyl-2-pyrrolidone as a penetration enhancer for a more rapid transfer of substances through the skin (Kydoniieus 1987; Barry and Bennett 1987; Akhter and Barry 1987). N-Methyl-2-pyrrolidone has been approved as a solvent for slimicide application to food packaging materials (USDA 1986).
색상 색인 번호
N-Methyl-2-pyrrolidone is an aprotic solvent with a wide range of applications: petrochemical processing, surface coating, dyes and pigments, industrial and domestic cleaning compounds, and agricultural and pharmaceutical formulations. It is mainly an irritant, but it can cause severe contact dermatitis due to prolonged contact.Safety Profile
Poison by intravenous route. Moderately toxic by ingestion and intraperitoneal routes. Mildly toxic by skin contact. An experimental teratogen. Experimental reproductive effects. Mutation data reported. Combustible when exposed to heat, open flame, or powerful oxidizers. To fight fire, use foam, CO2, dry chemical. When heated to decomposition it emits toxic fumes of NOx.Carcinogenicity
Rats were exposed to N-Methyl-2-pyrrolidone vapor at 0, 0.04, or 0.4 mg/L for 6 h/day, 5 days/week for 2 years.Male rats at 0.4 mg/L showed slightly reduced mean body weight. No life-shortening toxic or carcinogenic effects were observed in rats exposed for 2 years to either 0.04 or 0.4mg/L of N-Methyl-2-pyrrolidone. By the dermal route, a group of 32 mice received an initiation dose of 25mg of N-Methyl-2-pyrrolidone followed 2 weeks later by applications of the tumor promoter phorbol myristate acetate, three times a week, for more than 25 weeks. Dimethylcarbamoyl chloride and dimethylbenzanthracene served as positive controls. Although the N-Methyl-2-pyrrolidone group had three skin tumors, this response was not considered significant when compared with that of the positive controls.신진 대사 경로
Rats are administered radio-labeled N-methyl-2- pyrrolidinone (NMP), and the major route of excretion by rats is via the urine. The major metabolite, representing 70-75% of the administered dose, is 4-(methylamino)butenoic acid. This unsaturated intact product may be formed from the elimination of water, and a hydroxyl group may be present on the metabolite prior to acid hydrolysis.신진 대사
Male Sprague-Dawley rats were given a single intraperitoneal injection (45 mg/kg) of radiolabeled 1-methyl-2-pyrrolidone. Plasma levels of radioactivity and compound were monitored for six hours and the results suggested a rapid distribution phase which was followed by a slow elimination phase. The major amount of label was excreted in the urine within 12 hours and accounted for approximately 75% of the labelled dose. Twenty-four hours after dosage, cumulative excretion (urine) was approximately 80% of the dose. Both ring- and methyl-labeled species were used, as well as both [14C]- and [3H]-labeled l-methyl-2-pyrrolidone. The initial labeled ratios were maintained during the first 6 hours after dosage. After 6 hours, the liver and intestines were found to contain the highest accumulations of radioactivity, approximately 2-4% of the dose. Little radioactivity was noted in the bile or respired air. High performance liquid chromatography of urine showed the presence of one major and two minor metabolites. The major metabolite (70-75% of the administered radioactive dose) was analyzed by liquid chromatography-mass spectrometry and gas chromatography-mass spectrometry and was proposed to be a 3- or 5-hydroxy-l-methyl-2-pyrrolidone (Wells 1987).Purification Methods
Dry the pyrrolidone by removing water as the *benzene azeotrope. Fractionally distil at 10 torr through a 100-cm column packed with glass helices. [Adelman J Org Chem 29 1837 1964, McElvain & Vozza J Am Chem Soc 71 896 1949.] The hydrochloride has m 86-88o (from EtOH or Me2CO/EtOH) [Reppe et al. Justus Liebigs Ann Chem 596 1 1955]. [Beilstein 21 II 213, 21 III/IV 3145, 21/6 V 321.]N-메틸-2-피롤리디논 준비 용품 및 원자재
원자재
준비 용품
4-(트리플루오로메틸)피리딘-3-카복실산
1-BENZOFURAN-5-CARBONITRILE
트리메칠시아나이드
3,5-DIMETHOXYPHENYLACETIC ACID
2-(DIPHENYLPHOSPHINO)ETHYLTRIETHOXYSILANE
감마-아미노-N-부티르 산
3-AMINO-2-FLUORO-4-PICOLINE
5-(Trifluoromethyl)pyridine-2-carboxylic acid
1-[6-(Trifluoromethyl)pyridin-2-yl]piperazine
C-(1H-INDOL-5-YL)-METHYLAMINE
Vat Orange 9
ethyl 2-(4-chloroquinazolin-7-yloxy)acetate
THIOPHENE-2-THIOCARBOXAMIDE
5-Cyanoindole
3-Amino-6-chloropyridine-2-carboxylic acid
3-Amino-2-pyridinecarbonitrile
2-Furonitrile
1-(3-AMINOPROPYL)-2-PYRROLIDINONE
2-Chloro-6-trifluoromethylnicotinic acid
6-chloro-1H-indazol-3-amine
4-DIMETHYLAMINOBUTYRIC ACID HYDROCHLORIDE
2-Trifluoromethyl-5-bromopyridine
2-(TRIFLUOROMETHYL)-4-ISOCYANATOPYRIDINE
2-Trifluoromethyl-6-pyridinecarboxylic acid
6-(TRIFLUOROMETHYL)PYRIDINE-3-CARBOXALDEHYDE
2-(TRIFLUOROMETHYL)-PYRIDINE-4-CARBOXALDEHYDE
4-(PYRROLIDIN-1-YLMETHYL)BENZENEBORONIC ACID, PINACOL ESTER 97%1-[4-(4,4,5,5-TETRAMETHYL-1,3,2-DIOXABOROLAN-2-YL)BENZYL]PYRROLIDINE
N-메틸-2-피롤리디논 공급 업체
글로벌( 974)공급 업체
| 공급자 | 전화 | 이메일 | 국가 | 제품 수 | 이점 |
|---|---|---|---|---|---|
| NEM MATERIALS CO.,LTD | +8613781394683 |
chai205214@163.com | China | 4 | 58 |
| Xiamen AmoyChem Co., Ltd | +86-592-6051114 +8618959220845 |
sales@amoychem.com | China | 6387 | 58 |
| Hefei TNJ Chemical Industry Co.,Ltd. | +86-0551-65418679 +86-18949832763 |
info@tnjchem.com | China | 2989 | 55 |
| Tianjin Zhongxin Chemtech Co., Ltd. | +86-022-66880623 +8618622897568 |
sales@tjzxchem.com | China | 559 | 58 |
| SIMAGCHEM CORP | +86-13806087780 |
sale@simagchem.com | China | 17367 | 58 |
| Hebei Guanlang Biotechnology Co., Ltd. | +86-19930503282 |
alice@crovellbio.com | China | 8823 | 58 |
| Changzhou Xuanming Pharmaceutical Technology Co., Ltd. | +8618068519287 |
sales@xuanmingchem.com | China | 822 | 58 |
| Hebei Mojin Biotechnology Co., Ltd | +8613288715578 |
sales@hbmojin.com | China | 12456 | 58 |
| Hebei Yanxi Chemical Co., Ltd. | +8617531190177 |
peter@yan-xi.com | China | 5993 | 58 |
| Nanjing Deda New Material Technology Co., Ltd | +8613223293093 |
bella@njdeda.com | China | 81 | 58 |