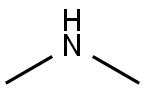
디메틸아민
|
|
디메틸아민 속성
- 녹는점
- −93 °C(lit.)
- 끓는 점
- 7 °C(lit.)
- 밀도
- 0.89 g/mL at 25 °C
- 증기 밀도
- 1.55 (vs air)
- 증기압
- 16.97 psi ( 55 °C)
- 굴절률
- n
20/D 1.37
- 인화점
- 60 °F
- 저장 조건
- Flammables area
- 용해도
- very soluble in water (163 g/100 g water at 40°C); soluble in ethanol, ethyl ether, and many organic solvents
- 물리적 상태
- 액체
- 산도 계수 (pKa)
- 10.68(at 25℃)
- 색상
- 약간 노란색으로 맑음
- 냄새
- 암모니아 냄새
- Odor Threshold
- 0.033ppm
- 폭발한계
- 14.4%
- 수용성
- 물 및 대부분의 유기용매와 섞일 수 있습니다.
- 감도
- Hygroscopic
- Merck
- 14,3228
- BRN
- 605257
- Henry's Law Constant
- 1.75(x 10-5 atm?m3/mol) at 25 °C (Christie and Crisp, 1967)
- 노출 한도
- TLV-TWA 10 ppm (~18 mg/m3) (ACGIH, MSHA, and OSHA); IDLH 2000 ppm (NIOSH).
- Dielectric constant
- 6.3(0℃)
- 안정성
- 안정적인. 일반적으로 최대 약 40% 농도의 물에 용해되어 사용됩니다. 순수한 형태에서는 인화성이 매우 높습니다. 강한 산화제와 호환되지 않습니다.
- LogP
- -0.274 at 25℃
- CAS 데이터베이스
- 124-40-3(CAS DataBase Reference)
안전
- 위험 및 안전 성명
- 위험 및 사전주의 사항 (GHS)
| 위험품 표기 | F+,Xn,C,F,T | ||
|---|---|---|---|
| 위험 카페고리 넘버 | 12-20-37/38-41-34-20/22-11-39/23/24/25-23/24/25-52/53-40-19 | ||
| 안전지침서 | 3-16-26-29-36/37/39-45-39-61 | ||
| 유엔번호(UN No.) | UN 2924 3/PG 2 | ||
| OEB | A | ||
| OEL | TWA: 10 ppm (18 mg/m3) | ||
| WGK 독일 | 2 | ||
| RTECS 번호 | IP8750000 | ||
| F 고인화성물질 | 3 | ||
| 자연 발화 온도 | 753 °F | ||
| TSCA | Yes | ||
| DOT ClassificationII | 2.1 (Flammable gas) | ||
| 위험 등급 | 3 | ||
| 포장분류 | II | ||
| HS 번호 | 29211100 | ||
| 유해 물질 데이터 | 124-40-3(Hazardous Substances Data) | ||
| 독성 | Acute oral LD50 for guinea pigs 340 mg/kg, mice 316 mg/kg, rats 698 mg/kg, rabbits 240 mg/kg (quoted, RTECS, 1985). | ||
| IDLA | 500 ppm | ||
| 기존화학 물질 | KE-11124 |
디메틸아민 C화학적 특성, 용도, 생산
물성
물, 에탄올 및 에테르에 용해되고, 쉽게 연소되며 약 알칼리성이며 물에 용해되는 무기산 염이 생성됩니다.개요
디메틸 아민 (DMA)의 다른 이름 N- 메틸 메탄 아민 (디메틸) 아민은 동식물에 널리 분포되어있다. 디메 폭스 (dimefox) 및 디페 닐 하이드라 민 (diphenhydramine)과 같은 많은 농약 및 의약품 생산을위한 원료입니다. DMA는 20 ℃에서 무색의 가스이며 암모니아 냄새와 유사합니다.용도
디메틸 아민 (DMA)은 주로 고무 가황 촉진제, 가죽 탈모 방지제, 의약품, 살충제, 섬유 산업용 솔벤트, 염료, 폭발물, 추진제 및 디메틸 히드라진, N, N- 디메틸 포름 아미드 및 유기 중간체의 기타 원료에 사용됩니다.개요
Dimethylamine is a colourless flammable gas at room temperature. It has a pungent, fishy, or ammonia-like odour at room temperature and is shipped and marketed in compressed liquid form. It is very soluble in water and soluble in alcohol and ether. It is incompatible with oxidising materials, acrylaldehyde, fluorine, maleic anhydride, chlorine, or mercury. Dimethylamine is a precursor to several industrially important compounds. For instance, it used in the manufacture of several products, for example, for the vulcanisation process of rubber, as detergent soaps, in leather tanning, in the manufacture of pharmaceuticals, and also for cellulose acetate rayon treatment.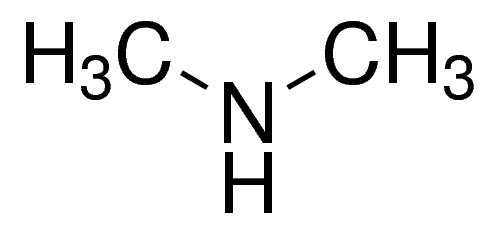
dimethylamine structure
화학적 성질
Dimethylamine reacts readily with acids to produce salts due to the presence of the unshared electron pair on the nitrogen atom. Similarly, dimethylamine reacts with acid anhydrides, halides, and esters, with CO2 or CS2, or with isocyanic or isothiocyanic acid derivatives. It can also react with nitrite, especially under acidic conditions, and possibly nitrogen oxides (Iqbel 1986) to form N-nitrosodimethylamine, a potent carcinogen in various animal species and a suspect human carcinogen (ATSDR 1989; Scanlan 1983; Zeisel et al 1988). N-Nitrosodimethylamine also can be formed upon storage of aqueous dimethylamine solutions or formulations of the dimethylamine salts of the herbicides 2,4D and MCPA (Wigfield and McLenaghan 1987a,b). Dimethylamine also can be nitrosated photochemically in aqueous solutions containing nitrite with the reaction occurring most readily at alkaline pH (Ohta et al 1982).물리적 성질
Clear, colorless liquid or gas with a strong, ammonia-like odor. Odor threshold concentrations of 33 ppbv and 47 ppbv were experimentally determined by (Leonardos et al., 1969) and Nagata and Takeuchi (1990), respectively.용도
Dimethylamine is used in the manufactureof N-methylformamide, N-methylacetamide,and detergent soaps; in tanning; and as anaccelerator in vulcanizing rubber. It is commercially sold as a compressed liquid intubes or as a 33% aqueous solution..정의
ChEBI: A secondary aliphatic amine where both N-substituents are methyl.공기와 물의 반응
Highly flammable. Water soluble.반응 프로필
DIMETHYLAMINE is a base, neutralizing acids in exothermic reactions, and a reducing agent. Dimethylamine is temperature sensitive. Reacts vigorously with mercury and chlorine . Reacts violently with strong oxidizing agents and attacks copper and copper compounds [Handling Chemicals Safely, 1980 p. 123]. Reacts with hypochlorites to give N-chloroamines, some of which are explosives when isolated [Bretherick, 1979 p. 108].위험도
Dimethylamine is an irritant, with a TLV of 10 ppm in air. The four-digit UN identification number is 1032. The NFPA 704 designation is health 3, flammability 4, and reactivity 0. The primary uses are in electroplating and as gasoline stabilizers, pharmaceuticals, missile fuels, pesticides, and rocket propellants.건강위험
Dimethylamine is a strong irritant to the eyes,skin, and mucous membranes. Spill of liquidinto the eyes can cause corneal damage andloss of vision. Skin contact with the liquidcan produce necrosis. At sublethal concentra tions, inhalation of dimethylamine producedrespiratory distress, bronchitis, pneumonitis,and pulmonary edema in test animals. Theacute oral toxicity was moderate, greater thanfor monomethylamine.LC50 value, inhalation (rats): 4540 ppm/6 hLD50 value, oral (mice): 316 mg/kg
Buckley and coworkers (1985) have investigated the inhalation toxicity of dimethylamine in F-344 rats and B6C3F1 mice.Animals exposed to 175 ppm for 6 h/day,5 days/week for 12 months showed significant lesions in the nasal passages. Rats developed more extensive olfactory lesions thandid mice. The study indicated that olfactory sensory cells were highly sensitive todimethylamine. Even at a concentration of10 ppm, the current threshold limit value,the rodents developed minor lesions fromexposure.
화재위험
FLAMMABLE. Flashback along vapor trail may occur. May explode if ignited in an enclosed area. Vapors are eye, skin and respiratory irritants.공업 용도
Dimethylamine is used as an accelerator in vulcanizing rubber, as an antiknock agent for fuels, in photography, as a plasticizer, ion exchange agent, as an acid gas absorbent, a flotation agent, a dehairing agent in the tanning of leather and in electroplating (HSDB 1989; Sax and Lewis 1987; Windholz et al 1983). Dimethylamine also serves as the base for a large number of commercial products including detergent soaps, dyes, pharmaceuticals, textile chemicals, surfactants and in the manufacture of unsymmetrical dimethylhydrazine (used in missile fuels), the solvent dimethylacetanilide and in the synthesis of dimethylformamide, one of the most commonly used organic solvents. Usage of dimethylamine in 1972 was estimated at 50% for production of dimethylformamide and dimethylacetamide (used as spinning solvents for acrylic fibers), 15% as an intermediate in the preparation of the surfactant laurel dimethylamine oxide, 15% as an intermediate for rubber chemicals (including thorium accelerators), and 20% for other applications including the production of unsymmetrical dimethylhydrazine in rocket fuels and the dimethylamine salt of 2,4-dichlorophenoxyacetic acid (HSDB 1989). U.S. production and sales of dimethylamine in 1985 was 65.9 million pounds.Safety Profile
Poison by ingestion. Moderately toxic by inhalation and intravenous routes. Mutation data reported. An eye irritant. Corrosive to the eyes, skin, and mucous membranes. A flammable gas. When heated to decomposition it emits toxic fumes of Nx,. Incompatible with acrylddehyde, fluorine, and maleic anhydrideCarcinogenicity
In a 2 year inhalation study in male F344 rats exposed to 175 ppm, no evidence of carcinogenicity was observed, and in addition, despite severe tissue destruction in the anterior nose following a single 6 h exposure, the nasal lesions exhibited very little evidence of progression, even at 2 years of exposure. The authors concluded that this indicated possible regional susceptibility to DMA toxicity or a degree of adaptation by the rat to continued DMA exposure.A detailed evaluation of mucociliary apparatus function and response to alterations of nasal structure was presented by the authors.
저장
Dimethylamine should be stored in a cool, dry, well-ventilated area in tightly sealed containers that are labeled in accordance with OSHA’s Hazard Communication Standard [29 CFR 1910.1200]. Containers of dimethylamine should be protected from physical damage and ignition sources, and should be stored separately from oxidizing materials, acrylaldehyde, fl uorine, maleic anhydride, chlorine, and mercury. Outside or detached storage is preferred. If stored inside, a standard flammable liquids cabinet or room should be used. Ground and bond metal containers and equipment when transferring liquids. Empty containers of dimethylamine should be handled appropriately.Purification Methods
Dry dimethylamine by passage through a KOH-filled tower, or by standing with sodium pellets at 0o during 18hours. [Beilstein 4 IV 128.]주의 사항
During handling of dimethylamine, workers should use proper fume hoods, personal protective clothing and equipment, avoid skin contact, and use gloves, sleeves, and encapsulating suits. Dimethylamine is extremely flammable and may be ignited by heat, sparks, or open flames. Liquid dimethylamine will attack some forms of plastic, rubber, and coatings and is flammable. The vapors of dimethylamine are an explosion and poison hazard. Containers of dimethylamine may explode in the heat of a fi re and require proper disposal. Workers should use dimethylamine with adequate ventilation and containers must be kept properly closed.디메틸아민 준비 용품 및 원자재
원자재
준비 용품
(2,4-디클로로페녹시)아세트산 다이메틸아민
6-디메틸아미노퓨린
Methyl cyclopentenolone
디페녹소론
Topotecan
디메틸아미노-1-프로판올
올린산 칼륨
Ethyl 3-(N,N-dimethylamino)acrylate
클로르프로마진 수화염화물
디메틸시클로헥실아민
camazepam
헥사데실트리메틸암모늄 염화물
비소디메틸디티오카르바메이트
Insecticide double agent
3-클로로프로필(디메틸)아민
3-DIMETHYLAMINOPROPIONIC ACID
2-브로모-N,N-디메틸벤질아민
디알릴 디메틸 암모늄 클로라이드
Dye-fixing agent,no formaldehyde
디에틸3,5-디-tert-부틸-4-히드록시벤질포스페이트
N,N-디메틸-N-2-프로펜일-2-프로펜-1-아미늄 염화, 중합체 WITH 2-프로펜아미드
디메틸아미노프로피온니트릴
N,N,N',N'-TETRAMETHYL-2-BUTENE-1,4-DIAMINE
2-Hydrazinyl-N,N-dimethylacetamide
3-(CHLOROMETHYL)-N,N-DIMETHYLBENZENESULFONAMIDE
독세핀
N,N-디메틸디티오카르밤산
디메틸(2-페녹시에틸)아민
디펜히드라민
염산 메트포르민
Light stabilizer 2002
그라민
5-클로로-1-메틸이미다졸
N,N-다이메틸아크릴아마이드
4-AMINO-N,N-DIMETHYLBENZYLAMINE
2,4,6-트리스(디메틸아미노메틸)페놀
Dimethyldioctadecylammonium bromide
3-(DIMETHYLAMINO)PROPYL CHLORIDE HYDROCHLORIDE
알트레타민
2,2'-티오비스에틸아민
디메틸아민 공급 업체
글로벌( 537)공급 업체
| 공급자 | 전화 | 이메일 | 국가 | 제품 수 | 이점 |
|---|---|---|---|---|---|
| Hebei Dangtong Import and export Co LTD | +86-13910575315 +86-13910575315 |
admin@hbdangtong.com | China | 1005 | 58 |
| Yujiang Chemical (Shandong) Co.,Ltd. | +86-17736087130 +86-18633844644 |
catherine@yjchem.com.cn | China | 983 | 58 |
| Capot Chemical Co.,Ltd. | 571-85586718 +8613336195806 |
sales@capotchem.com | China | 29798 | 60 |
| Henan Tianfu Chemical Co.,Ltd. | +86-0371-55170693 +86-19937530512 |
info@tianfuchem.com | China | 21667 | 55 |
| Anhui Royal Chemical Co., Ltd. | +86-25-86655873 +8613962173137 |
marketing@royal-chem.com | China | 189 | 55 |
| career henan chemical co | +86-0371-86658258 +8613203830695 |
sales@coreychem.com | China | 29888 | 58 |
| Hubei Jusheng Technology Co.,Ltd. | 18871490254 |
linda@hubeijusheng.com | CHINA | 28180 | 58 |
| Hubei xin bonus chemical co. LTD | 86-13657291602 |
linda@hubeijusheng.com | CHINA | 22968 | 58 |
| Standardpharm Co. Ltd. | 86-714-3992388 |
overseasales1@yongstandards.com | United States | 14335 | 58 |
| Chongqing Chemdad Co., Ltd | +86-023-6139-8061 +86-86-13650506873 |
sales@chemdad.com | China | 39916 | 58 |
디메틸아민 관련 검색:
트라이메틸아민(트리메틸아민) 메틸아민 디메틸아미노아세트알데히드에틸아세탈 과플루오로트리부틸아민 2'-플루오로아세트아닐라이드 N,N-디메틸카바모일클로라이드 디에틸카바모일클로라이드 펜아미노설프 디메틸아미노프로피온니트릴 2-디메틸아미노에탄올 디메틸포름아미드디에틸아세탈 N,N-다이메틸도데실아민 N-산화물 N,N-디메틸아닐린
4'-Chloroacetoacetanilide
3-CHLORODIPHENYLAMINE
4-Chloro-N-methylaniline
4'-CHLOROACETANILIDE
N-Methyl-N-(trimethylsilyl)trifluoroacetamide