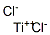티타늄 트리염화물 C화학적 특성, 용도, 생산
화학적 성질
purple crystalline solid
물리적 성질
Red-violet hexagonal crystals; hygroscopic; density 2.64 g/cm
3; decomposes on heating above 425°C; also decomposes in water, evolving heat; soluble in alcohol, acetonitrile and certain amines; insoluble in hydrocarbons and ether.
용도
As powerful reducing agent, Titanous chlorideb can reduces nitrate to ammonia; when boiled with aqueous SO2, sulfur is separated; hence is used as an aqueous solution for estimation of nitro groups, ferric ions, per-salts, etc. Removes stains, etc. (stripper) in laundering.
제조 방법
Titanium trichloride may be prepared by reducing titanium tetrachloride with hydrogen at 600°C. The tetrachloride may alternatively be reduced with aluminum, zinc, magnesium, tin, or by electrolysis.
일반 설명
A dark violet crystalline solid. Denser than water. Contact may burn skin, eyes, and mucous membranes. May be toxic by ingestion, inhalation and skin absorption. Used to make other chemicals.
공기와 물의 반응
Pyrophoric, very reactive with water and moisture in air produces hydrochloric acid, [Merck 11th ed. 1989]. Ignites spontaneously on contact with air; decomposed by water and water vapor forming HCl. [Handling Chemcials Safely 1980. p. 905].
반응 프로필
Acidic salts, such as TITANIUM TRICHLORIDE, are generally soluble in water. The resulting solutions contain moderate concentrations of hydrogen ions and have pH's of less than 7.0. They react as acids to neutralize bases. These neutralizations generate heat, but less or far less than is generated by neutralization of inorganic acids, inorganic oxoacids, and carboxylic acid. They usually do not react as either oxidizing agents or reducing agents but such behavior is not impossible. Many of these compounds catalyze organic reactions. Ethylene can polymerize at low pressure if catalyzed by titanium halides. (Sundaram, K. M, M. M. Shreehan, E. F. Olszewski. thylene. Kirk-Othmer Encyclopedia of Chemical Technology. John Wiley & Sons, Inc. 2001.)
위험도
Fire risk in the presence of oxidizing materials.
Irritant to skin and tissue; open container only
in oxygen-free or inert atmosphere.
건강위험
Fire will produce irritating, corrosive and/or toxic gases. Inhalation of decomposition products may cause severe injury or death. Contact with substance may cause severe burns to skin and eyes. Runoff from fire control may cause pollution.
화재위험
Flammable/combustible material. May ignite on contact with moist air or moisture. May burn rapidly with flare-burning effect. Some react vigorously or explosively on contact with water. Some may decompose explosively when heated or involved in a fire. May re-ignite after fire is extinguished. Runoff may create fire or explosion hazard. Containers may explode when heated.
Safety Profile
A corrosive irritant to
skin, eyes, and mucous membranes. A
severe corrosive because it liberates heat and
hydrochloric acid upon contact with
moisture. If spilled on slun, wipe off with
dry cloth before applying water. May ignite
spontaneously in air. Flammable when
exposed to heat or flame. Reacts violently
with K, HF. Experimental reproductive
effects. When heated to decomposition it
emits toxic fumes of Cl-. See also
TITANIUM COMPOUNDS.
Purification Methods
It is a brown purple powder that is very reactive to H2O and pyrophoric when dry. It should be manipulated in a dry box. It is soluble in CH2Cl2 and tetrahydrofuran, and is used as a M solution in these solvents in the ratio of 2:1, and stored under N2. It is a powerful reducing agent. [Ingraham et al. Inorg Synth VI 52 1960.]
티타늄 트리염화물 준비 용품 및 원자재
원자재
준비 용품