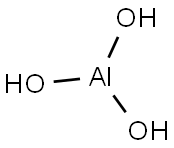
수산화알루미늄분말
|
|
수산화알루미늄분말 속성
- 녹는점
- 300℃
- 끓는 점
- 2980℃[at 101 325 Pa]
- 밀도
- 2.42 g/cm3 at 20 °C
- 증기압
- <0.1 hPa (20 °C)
- 굴절률
- Average refractive index: 1.57-1.59
- 저장 조건
- Store at +5°C to +30°C.
- 용해도
- 0.0015g/L
- 물리적 상태
- 콜로이드 현탁액
- Specific Gravity
- 2.42
- 색상
- 하얀색
- 냄새
- 냄새 없는
- pH 범위
- >7
- 수소이온지수(pH)
- 8-9 (100g/l, H2O, 20℃)(slurry)
- 수용성
- 불용성
- Crystal Structure
- Monoclinic
- Merck
- 14,342
- Solubility Product Constant (Ksp)
- pKsp: 32.89
- 노출 한도
- ACGIH: TWA 1 mg/m3
- Dielectric constant
- 2.2(Ambient)
- 안정성
- 안정적인. 강한 염기와 호환되지 않습니다.
- LogP
- -1.380 (est)
- CAS 데이터베이스
- 21645-51-2(CAS DataBase Reference)
안전
- 위험 및 안전 성명
- 위험 및 사전주의 사항 (GHS)
| 위험품 표기 | Xi | ||
|---|---|---|---|
| 위험 카페고리 넘버 | 36 | ||
| 안전지침서 | 26-36 | ||
| WGK 독일 | - | ||
| RTECS 번호 | BD0940000 | ||
| TSCA | Yes | ||
| HS 번호 | 28183000 | ||
| 유해 물질 데이터 | 21645-51-2(Hazardous Substances Data) | ||
| 독성 | TDLo oral in child: 79gm/kg/2Y-I | ||
| 기존화학 물질 | KE-00980 |
수산화알루미늄분말 C화학적 특성, 용도, 생산
물성
수산화알루미늄 일반제품(수분 약 9~10%함유)을 약 130℃ 이내의 열풍으로 건조시킨 제품으로서, 화학내열성, 투명성, 특히 난연성등의 품질이 특성이 우수한 기초 무기화학 제품이다.존재
수산화 알루미늄(aluminium hydroxide, Al(OH)3, ATH)은 알루미늄의 양쪽성 수산화물이다. 자연 상태에서는 깁사이트 광물과 다음과 같은 세 개의 드문 다형체 형태로 존재한다: bayerite(바이어라이트), doyleite, nordstrandite.화학 반응
보크사이트를 원료로 제조한 수산화알루미늄은 화학식으로 Al(OH)3 또는 Al2O3 3H3O로 표시합니다. 산,알칼리에 쉽게 반응하여 용해하고 열적으로 200℃까지 안정하며 더 높은 온도에서 결정수가 탈수하면 많은양의 열을 흡수하므로 난연재로 널리 사용됩니다. 자유수분 함량과 평균입도에 따라 여러가지 형태의 제품을 다양하게 생산하므로 다양한 중화학공업 분야에 적용할 수 있습니다. 알루민산나트륨 수용액에 이산화탄소를 이용하게 되면 결정이 침전된다.NaAlO2+2H2O+CO2→Al(OH)3+NaHCO3
용도
수산화알루미늄은 습식, 건조, 건조미분, 초미분으로 나뉘어 각각의 용도별로 적용한다. 습식 일반은 황산알루미늄 제조용; 건조 일반은 유리, 인조대리석, 타일유약용; 건조미분은 충전제, 난연제, 건축자재원료, 고압변압기 재료용; 초미분은 난연성과 백색도가 높아 주로 전선용 난연제 사용.독성
다른 알루미늄 화합물과 마찬가지로 수산화알루미늄 혼합물은 수렴제이며 메스꺼움, 구토 및 변비를 일으킬 수 있다. 많은 용량을 복용할 경우 장폐색이 일어날 수 있다. 과도한 용량을 섭취할 때 또는 저인산염 식이요법을 하는 환자가 일반적인 용량을 섭취할 때 인산염 고갈 현상이 일어날 수 있으며 칼슘의 흡수가 증가하고 소변으로 인한 칼슘 배출량이 늘어나서 젊은 사람들은 신장구룻병, 노인 환자들은 골연화증이 생길 위험이 커진다. 분진은 점막 자극을 일으킬 수 있다. 알루미늄 하이드레이트를 포함한 고운 분진을 흡입했을 때 사람의 경우 폐기능이 저하되고 가쁜 얕은 호흡을 일으켰으며, 알루미늄이 포함된 분진을 장기간 흡입할 경우 비결절성 폐섬유증, 기종, 기흉, 그리고 드물게 뇌병증이 발생했다.화학적 성질
Aluminum hydroxide, Al(OH)3, also known as aluminum trihydroxide, aluminum trihydrate, aluminum hydrate, hydrated alumina, and hydrated aluminum oxide, is a white to whitish-yellow water-insoluble powder with a specific gravity of 2.42. Aluminum hydroxide is used as a baseforpigments, as a water repellent in textile coatings, and as an antacid in medicine. Aluminum hydroxide is soluble in hydrochloric or sulfuric acids or in sodiumhydroxide.용도
aluminum hydroxide is an inorganic compound used to make a product less transparent. It is also used by formulators as a humectant, and to soften, smooth, and protect the skin. In addition it helps control product viscosity. often found in facial masks and make-up preparations.생산 방법
Aluminum hydroxide adjuvant is prepared by the precipitation of a soluble aluminum salt by an alkali hydroxide, or the precipitation of an alkali aluminate by acid.Pharmaceutical Applications
Aluminum hydroxide adjuvant is used in parenteral human and veterinary vaccines.It activates Th2 immune responses, including IgG and IgE antibody responses. It is also used for the isolation of certain serum components such as blood clotting factors.Safety Profile
Poison by intraperitoneal route. Human systemic effects by ingestion: fever, osteomalacia, and gastrointestinal effects. When coprecipitated with bismuth hydroxide and reduced by H2, it is violently flammable in air. Incompatible with chlorinated rubber.Safety
Aluminum hydroxide adjuvant is intended for use in parenteral vaccines and is generally regarded as nontoxic. It may cause mild irritation, dryness, and dermatitis on skin contact. On eye contact, aluminum hydroxide adjuvant may also cause redness, conjunctivitis, and short-term mild irritation. Ingestion of large amounts may cause gastrointestinal irritation with nausea, vomiting, and constipation. Inhalation of the dried product may cause respiratory irritation and cough. Type I hypersensitivity reactions following parenteral administration have been reported.저장
Aluminum hydroxide adjuvant is stable for at least 2 years when stored at 4–308℃ in well-sealed inert containers. It must not be allowed to freeze as the hydrated colloid structure will be irreversibly damaged.비 호환성
When exposed to phosphate, carbonate, sulfate, or borate anions, the point of zero charge for aluminum hydroxide adjuvant decreases.Regulatory Status
GRAS listed. Accepted for use in human and veterinary parenteral vaccines in Europe and the USA. The limits for use in human vaccines are 0.85 mg aluminum/dose (FDA) and 1.25 mg aluminum/ dose (WHO). There are no established limits for use in veterinary vaccines. Reported in the EPA TSCA Inventory.수산화알루미늄분말 준비 용품 및 원자재
원자재
준비 용품
OH-18 hydrotreating catalyst for reforming feedstocks
hydrofining catalyst CH-17
catalyst for selective hydrogenation of C^{3^} fractions
질산 알루미늄
플루오알루민산나트륨
sodium aluminium acid phosphate for food
methanation catalysts
질산알루미늄
식용 적색 2호
Silicic acid, aluminum potassium sodium salt
식용 적색 102호
브로민화 알루미늄, 무수물
ALUMINUM OXIDE,ACTIVATED,NEUTRAL,FOR COLUMN CHROMATOGRAPHY,63-200ΜM
황색 100 침착염료
Erythrosine aluminium lake
인산 알루미늄
EC 1.1.3.22
황산알루미늄
new red
Potassium fluoroaluminate
모노알루미늄 인산염 용액
코발트 알루미네이트 청색 첨정석
칼리크레인
암모늄명반
Food Blue 1:1
Fast Pink lake
플루오린화 알루미늄
ALBUMIN, BOVINE
reforming catalysts(series)
sodium aluminium phosphate,basic,for food
carboxyl type cation exchange resin
hydrocracking catalyst 3824 for middle oil
알루미늄 클로로수화물
FD와 C 황색 NO. 6 알루미늄 레이크
low temperature CO shift catalysts
CuO-Al inorganic adhesive
Aluminium sulfate hyd랫드 e
트리나트륨 알루미늄 헥사플루오리드
dewatering desiccants
hydrofining catalysts (RN series)
수산화알루미늄분말 공급 업체
글로벌( 582)공급 업체
| 공급자 | 전화 | 이메일 | 국가 | 제품 수 | 이점 |
|---|---|---|---|---|---|
| Hebei Yanxi Chemical Co., Ltd. | +8617531190177 |
peter@yan-xi.com | China | 5993 | 58 |
| Henan Fengda Chemical Co., Ltd | +86-371-86557731 +86-13613820652 |
info@fdachem.com | China | 7845 | 58 |
| Shandong Juchuang Chemical Co., LTD | +86-18885615001 +86-18885615001 |
admin@juchuangchem.com | China | 387 | 58 |
| Henan Tianfu Chemical Co.,Ltd. | +86-0371-55170693 +86-19937530512 |
info@tianfuchem.com | China | 21691 | 55 |
| Shanghai Time Chemicals CO., Ltd. | +86-021-57951555 +8617317452075 |
jack.li@time-chemicals.com | China | 1807 | 55 |
| Hangzhou FandaChem Co.,Ltd. | 008657128800458; +8615858145714 |
fandachem@gmail.com | China | 9348 | 55 |
| Hefei TNJ Chemical Industry Co.,Ltd. | +86-0551-65418679 +86-18949832763 |
info@tnjchem.com | China | 2989 | 55 |
| Yancheng Green Chemicals Co.,Ltd | +undefined-86-25-86655873 |
info@royal-chem.com | China | 114 | 58 |
| career henan chemical co | +86-0371-86658258 |
sales@coreychem.com | China | 29914 | 58 |
| SHANDONG ZHI SHANG CHEMICAL CO.LTD | +86 18953170293 |
sales@sdzschem.com | China | 2931 | 58 |