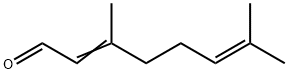
Citral
- CAS No.
- 5392-40-5
- Chemical Name:
- Citral
- Synonyms
- NERAL;GERANIAL;cis-Citral;2,6-Octadienal, 3,7-dimethyl-;3,7-DIMETHYL-2,6-OCTADIENAL;CITRATHAL;FEMA 2303;geranialdehyde;CITRAL NATURAL;(2E)-3,7-Dimethyl-2,6-octadienal
- CBNumber:
- CB8364074
- Molecular Formula:
- C10H16O
- Molecular Weight:
- 152.23
- MDL Number:
- MFCD00006997
- MOL File:
- 5392-40-5.mol
- MSDS File:
- SDS
- TDS File:
- TDS
| Product description | Number | Pack Size | Price |
| Citral natural, ≥96%, FCC, FG | W230316 | 1 each | $62.3 |
| Citral natural, ≥96%, FCC, FG | W230316 | 1kg | $223 |
| Citral mixture of cis and trans, ≥96%, FG | W230308 | 1 each | $58 |
| Citral natural, ≥96%, FCC, FG | W230316 | 9kg | $1330 |
| Citral mixture of cis and trans, ≥96%, FG | W230308 | 1kg | $111 |
| More product size | |||
| Melting point | <-10°C |
|---|---|
| Boiling point | 229 °C (lit.) |
| Density | 0.888 g/mL at 25 °C (lit.) |
| vapor density | 5 (vs air) |
| vapor pressure | 0.2 mm Hg ( 200 °C) |
| FEMA | 2303 | CITRAL |
| refractive index |
n |
| FLAVIS Number | 05.020 | Citral |
| Flash point | 215 °F |
| storage temp. | 2-8°C |
| solubility | 0.42g/l |
| form | Liquid |
| color | colorless to light yellow |
| Odor | at 100.00 %. sweet lemon citral |
| Odor Type | citrus |
| biological source | synthetic |
| explosive limit | 4.3-9.9%(V) |
| Viscosity | 2.42mm2/s |
| Water Solubility | PRACTICALLY INSOLUBLE |
| Merck | 14,2322 |
| JECFA Number | 1225 |
| BRN | 1721871 |
| Henry's Law Constant | 2.9×10-1 mol/(m3Pa) at 25℃, Wu et al. (2022) |
| Exposure limits | ACGIH: TWA 5 ppm (Skin) |
| Stability | Stable. but readily isomerizes. Incompatible with alkalies, strong oxidizing agents, strong acids. Combustible. Air and light sensitive. |
| Major Application | flavors and fragrances |
| Cosmetics Ingredients Functions |
FLAVOURING FRAGRANCE PERFUMING |
| InChI | 1S/C10H16O/c1-9(2)5-4-6-10(3)7-8-11/h5,7-8H,4,6H2,1-3H3 |
| InChIKey | WTEVQBCEXWBHNA-UHFFFAOYSA-N |
| SMILES | [H]C(=O)C=C(C)CC\C=C(\C)C |
| LogP | 2.76 at 25℃ |
| Toxicology and Carcinogenesis | Toxicology and Carcinogenesis Studies of Citral (Microencapsulated) (CASRN 5392-40-5) in F344/N Rats and B6C3F1 Mice (Feed Studies) |
| CAS DataBase Reference | 5392-40-5(CAS DataBase Reference) |
| Substances Added to Food (formerly EAFUS) | CITRAL |
| FDA 21 CFR | 182.60 |
| EWG's Food Scores | 4-7 |
| FDA UNII | T7EU0O9VPP |
| NIST Chemistry Reference | Citral(5392-40-5) |
| EPA Substance Registry System | Citral (5392-40-5) |
| UNSPSC Code | 85151701 |
| NACRES | NA.24 |
SAFETY
Risk and Safety Statements
| Symbol(GHS) |  GHS07 |
|||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Signal word | Warning | |||||||||
| Hazard statements | H315-H317-H319 | |||||||||
| Precautionary statements | P261-P264-P272-P280-P302+P352-P305+P351+P338 | |||||||||
| PPE | Eyeshields, Faceshields, Gloves, type ABEK (EN14387) respirator filter | |||||||||
| Hazard Codes | Xi | |||||||||
| Risk Statements | 38-43 | |||||||||
| Safety Statements | 24/25-37 | |||||||||
| RIDADR | 1760 | |||||||||
| WGK Germany | 1 | |||||||||
| RTECS | RG5075000 | |||||||||
| Autoignition Temperature | 225 °C | |||||||||
| TSCA | TSCA listed | |||||||||
| HS Code | 2912 19 00 | |||||||||
| HazardClass | 8 | |||||||||
| PackingGroup | III | |||||||||
| Storage Class | 10 - Combustible liquids | |||||||||
| Hazard Classifications | Eye Irrit. 2 Skin Irrit. 2 Skin Sens. 1 |
|||||||||
| Hazardous Substances Data | 5392-40-5(Hazardous Substances Data) | |||||||||
| Toxicity | LD50 orally in rats: 4.96 g/kg (Opdyke) | |||||||||
| NFPA 704 |
|
Citral price More Price(42)
| Manufacturer | Product number | Product description | CAS number | Packaging | Price | Updated | Buy |
|---|---|---|---|---|---|---|---|
| Sigma-Aldrich | W230316 | Citral natural, ≥96%, FCC, FG | 5392-40-5 | 1 each | $62.3 | 2026-03-19 | Buy |
| Sigma-Aldrich | W230316 | Citral natural, ≥96%, FCC, FG | 5392-40-5 | 1kg | $223 | 2026-03-19 | Buy |
| Sigma-Aldrich | W230308 | Citral mixture of cis and trans, ≥96%, FG | 5392-40-5 | 1 each | $58 | 2026-03-19 | Buy |
| Sigma-Aldrich | W230316 | Citral natural, ≥96%, FCC, FG | 5392-40-5 | 9kg | $1330 | 2026-03-19 | Buy |
| Sigma-Aldrich | W230308 | Citral mixture of cis and trans, ≥96%, FG | 5392-40-5 | 1kg | $111 | 2026-03-19 | Buy |
Citral Chemical Properties,Uses,Production
Overview
Citral (C10H16O), also called 3,7-dimethyl-2,6-octadienal, a pale yellow liquid, with a strong lemon odour, that occurs in the essential oils of plants. It is insoluble in water but soluble in ethanol (ethyl alcohol), diethyl ether, and mineral oil. It is used in perfumes and flavourings and in the manufacture of other chemicals. Chemically, citral is a mixture of two aldehydes that have the same molecular formula but different structures.
Content analysis
Accurately weigh about 1g of the sample, and then perform the determination by the hydroxylamine method (OT-7, method one) used in aldehyde and ketone determination. The equivalent factor (e) in the calculation is 76.12.
Toxicity
ADI 0~0.5mg/kg (FAO/WHO, 1994-). LD50 4960 mg/kg (rat, oral); MNL 500 mg/kg.
Usage limits
FEMA (mg/kg): soft drinks 9.2; cold drinks 23; candy 41; baked goods 43; chewing gums 170
Chemical Properties
Colorless or slightly yellow liquid; strong lemon flavor; no optical rotation; boiling point 228 °C; flash point 92 °C;
There are cis and trans two isomers. With sodium bisulfite treatment, cis isomer solubility is minimal, while the trans isomer solubility is very large, so the two isomers can be separated.
Cis citral: relative density (d20) 0.8898, refractive index (nD20) 1.4891, boiling point 118~119℃ (2666Pa).
Trans citral: relative density (d20) 0.8888, refractive index (nD20) 1.4891, boiling point 117~118℃ (2666Pa).
Soluble in non-volatile oils, volatile oils, propylene glycol and ethanol; insoluble in glycerol and water; unstable in alkaline and strong acids
Natural products present in lemon grass oil (70% to 80%), litsea cubeba oil (about 70%), lemon oil, white lemon oil, citrus leaf oil and so on.
Application
Citral is an artificial flavor permitted to use in China, which can be used to prepare strawberries, apples, apricots, sweet orange, lemon and other fruit-based flavors. According to normal production needs, the citrals amount used in chewing gums is 1.70mg/kg; baked goods 43mg/kg; candy 41mg/kg; cold drinks 23mg/kg; soft drinks 9.2mg/kg.
It is also widely used in dishwashing detergents and the flavoring agents of soap and toilet water. Citral can be used as the raw material to synthesize ionone, methyl ionone and dihydro damascene. As organic raw material, it can also be reduced to generate citronellol, nerol alcohol and geraniol, and be converted into lemonile. In the pharmaceutical industry, it can be used for the manufacture of vitamin A and E, and also as the raw material of chlorophyll.
Production method
Citral natural exists in the litsea cubeba oil (about 80%), lemon grass oil (80%), clove basil oil (65%), sour lemon oil (35%) and lemon oil. In industry, citral can be derived from natural essential oils, or be prepared by chemical.
Synthesis based on methyl heptenone as raw material
Ethoxyacetylene magnesium bromide and methyl heptenone performed condensation reaction to form 3,7-dimethyl-1-ethoxy-3-hydroxy-6-octene-1-yne, which was then partly hydrogenated in the presence of catalysis to generate enol ether. And the enol ether was then hydrolyzed with phosphoric acid and dehydrated to obtain citral, with a yield of 68% calculated by methyl heptenone. In addition, acetylene and methyl heptenone could perform condensation reaction to form dehydrogenation linalool, which was then rearranged in the presence of silicon sulfone catalysis at 140~150 °C in inert solvent to get citral.
Derived from litsea cubeba oil (which is the main method to product citral in China)
Add 30 kg of cubeba oil containing about 75% of citral into a mixture under fully stirring, which was prepared with 18 kg of sodium bicarbonate, 38 kg of sodium sulfite and about 165 kg of water, and then continually stir for 5 to 6 h at room temperature. After standing overnight for stratification, the lower citral precipitated in the form of adduct. And the adduct was then washed with a small amount of toluene to remove oil and dried. And then add 10% sodium hydroxide solution to decompose citral at room temperature, and extract it with benzene. The extract was first distilled at atmospheric pressure (80-82°C) to recover benzene and then distilled under reduced pressure to collect fractions of 110-111°C (1.47kPa) to obtain pure product of 98% citral in an amount of about 15 to 16 kg.
Chemical Properties
mobile light yellow liquid with a lemon-like smell
Chemical Properties
Citral has a strong, lemon-like odor and a characteristic bittersweet taste. Commercially, the product is a mixture of two geometric isomers—α-citral and β-citral, each exhibiting cis- and trans-isomers because of the position of the double bond.
Chemical Properties
Citral occurs as (2Z)- and (2E)-isomers (citral a and b, respectively)
analogous to the corresponding alcohols, geraniol and nerol: geranial
(citral a), bp2.7 kPa 118–119 °C, d20 0.8888, n20
D 1.4898; neral (citral b),
bp2.7 kPa 120 °C, d20 0.8869, n20
D 1.4869.
Natural citral is nearly always a mixture of the two isomers. It occurs in lemongrass
oil (up to 85%), in Litsea cubeba oil (up to 75%), and in small amounts inmany
other essential oils. The citrals are colorless to slightly yellowish liquids, with an
odor reminiscent of lemon.
Since citral is an α,β-unsaturated aldehyde with an additional double bond, it
is highly reactive and may undergo reactions such as cyclization and polymerization.
Geraniol, citronellol, and 3,7-dimethyloctan-l-ol can be obtained from citral
by stepwise hydrogenation. Citral can be converted into a number of addition
compounds; the (Z)- and (E)-isomers can be separated via the hydrogen sulfite
addition compounds.The condensation of citral with active methylene groups is
used on an industrial scale in the synthesis of pseudoionones, which are starting
materials for ionones and vitamins.
Occurrence
Reported found in apricot, clary sage, ginger, grape, grapefruit, lemon, lime, mandarin, orange, raspberry, tamarind, tangerine, tea and tomato.
Uses
Citral is an anti-microbial agent found in plants with antibacterial activity against some food pathogens. It is also a fragrance compound with a distinct lemon scent.
Uses
Citral is a liquid flavoring agent, light yellow in color with a citrus odor. it occurs in lemon and lemongrass oils. it is usually obtained from citral-containing oils by chemical means but may also be pre- pared synthetically. it is soluble in fixed oils, mineral oil, and pro- pylene glycol. it is moderately stable and should be stored in glass, tin, or resin-lined containers. it is used in flavors for lemon with applications in candy, baked goods, and ice cream at 20–40 ppm. it is also termed 2,6-dimethyl-octadian-2-6-al-8.
Uses
citral is a naturally occurring aroma compound used to provide a lemon-type fragrance. Citral is a constituent of lemon oil, lemongrass oil, lime oil, ginger oil, verbena oil, and other plant-derived C essential oils.
Definition
Commercial material is a mixture of α and β isomers.
Preparation
Since citral is used in bulk as a starting material for the synthesis of
vitamin A, it is produced industrially on a large scale. Smaller quantities are also
isolated from essential oils.
1) Isolation from essential oils: Citral is isolated by distillation from lemongrass
oil and from L. cubeba oil. It is the main component of these oils.
2) Synthesis from geraniol: Currently, the most important synthetic procedures
are vapor-phase dehydrogenation and oxidation of geraniol or geraniol–nerol
mixtures. Catalytic dehydrogenation under reduced pressure using copper
catalysts is preferred.
3) Synthesis from dehydrolinalool: Dehydrolinalool is produced on a large scale
from 6-methyl-5-hepten-2-one and acetylene and can be isomerized to citral
in high yield by a number of catalysts. Preferred catalysts include organic
orthovanadates, organic trisilyl oxyvanadates, and vanadium catalysts
with silanols added to the reaction system.
4) Synthesis from isobutene and formaldehyde: 3-Methyl-3-buten-l-ol, obtained
from isobutene and formaldehyde, isomerizes to form 3-methyl-
2-buten-lol. However, it is also converted into 3-methyl-2-butenal
by dehydrogenation and subsequent isomerization. Under
azeotropic conditions in the presence of nitric acid, 3-methyl-2-buten-l-ol
and 3-methyl-2-butenal form an acetal (shown as follows), which
eliminates one molecule of 3-methyl-2-buten-l-ol at higher temperatures.
The intermediate enol ether undergoes Claisen rearrangement followed by
Cope rearrangement to give citral in excellent yield:
Today, this route is performed on a very large industrial scale in a continuous
reactive distillation process.
Aroma threshold values
Detection at 1.0%: characterizing lemon-like, distilled lime peel, intense aldehydic citruslike.
Taste threshold values
Taste characteristics at 5 ppm in 5% sugar and 0.1% CA: characteristic lemon, peely, citrus, green floral juicy with woody and candy notes.
Synthesis Reference(s)
Journal of the American Chemical Society, 101, p. 7131, 1979 DOI: 10.1021/ja00517a088
Tetrahedron Letters, 35, p. 4007, 1994 DOI: 10.1016/S0040-4039(00)76726-8
General Description
A clear yellow colored liquid with a lemon-like odor. Less dense than water and insoluble in water. Toxic by ingestion. Used to make other chemicals.
Air & Water Reactions
Insoluble in water.
Reactivity Profile
Citral is an aldehyde. Aldehydes are frequently involved in self-condensation or polymerization reactions. These reactions are exothermic; they are often catalyzed by acid. Aldehydes are readily oxidized to give carboxylic acids. Flammable and/or toxic gases are generated by the combination of aldehydes with azo, diazo compounds, dithiocarbamates, nitrides, and strong reducing agents. Aldehydes can react with air to give first peroxo acids, and ultimately carboxylic acids. These autoxidation reactions are activated by light, catalyzed by salts of transition metals, and are autocatalytic (catalyzed by the products of the reaction). The addition of stabilizers (antioxidants) to shipments of aldehydes retards autoxidation. Citral can react with alkalis and strong acids. Citral can readily isomerize.
Hazard
Questionable carcinogen.
Fire Hazard
Citral is combustible.
Flammability and Explosibility
Non flammable
Contact allergens
Citral is an aldehyde fragrance and flavoring ingredient, a blend of isomers cis (Neral) and trans (geranial). As a fragrance allergen, citral has to be mentioned by name in cosmetics within the EU.
Safety Profile
Moderately toxic by intraperitoneal route. Mildly toxic by ingestion. Experimental reproductive effects. A severe human and experimental skin irritant. Mutation data reported. Combustible liquid. When heated to decomposition it emits acrid smoke and irritating fumes.
Synthesis
Citral is usually isolated from the citral-containing oil by chemical means or by chemical synthesis (from β-pinene, isoprene, etc.).
Citral Preparation Products And Raw materials
Raw materials
1of4
Preparation Products
1of5
| Supplier | Tel | Country | ProdList | Advantage | |
|---|---|---|---|---|---|
| Changsha Hongyuxiang Chemical Co., Ltd | +8613757949618 | info@hoyoshee.com | China | 105 | 58 |
| Hebei Chuanghai Biotechnology Co., Ltd | +8615350571055 | Sibel@chuanghaibio.com | China | 8738 | 58 |
| Hebei Chuanghai Biotechnology Co,.LTD | +86-86-13131129325 +8613131129325 | sales1@chuanghaibio.com | China | 5235 | 58 |
| Hebei Dangtong Import and export Co LTD | +86-86-4001020630 +8619831957301 | admin@hbdangtong.com | China | 1000 | 58 |
| Hebei Chuanghai Biotechnology Co., Ltd | +8615531151365 | mina@chuanghaibio.com | China | 18126 | 58 |
| Shandong Juchuang Chemical Co., LTD | +16837774622 | admin@juchuangchem.com | China | 295 | 58 |
| Hebei Zhuanglai Chemical Trading Co.,Ltd | +8613343047651 | admin@zlchemi.com | China | 3692 | 58 |
| JINING XINHE CHEMICAL CO., LTD | +8615318402391 | sales@xinhepharma.com | China | 809 | 58 |
| HEBEI SHENGSUAN CHEMICAL INDUSTRY CO.,LTD | +86-15350851019; +8615350851019 | admin@86-ss.com | China | 999 | 58 |
| Capot Chemical Co.,Ltd. | +86-(0)57185586718; +8613336195806 | sales@capot.com | China | 29640 | 60 |
View Lastest Price from Citral manufacturers
| Image | Update time | Product | Price | Min. Order | Purity | Supply Ability | Manufacturer | |
|---|---|---|---|---|---|---|---|---|
 |
2026-04-29 | Citral
5392-40-5
|
0.99 | RongNa Biotechnology Co.,Ltd | ||||
 |
2026-04-29 | Citral
5392-40-5
|
US $0.00 / KG | 1KG | 98.0% | 1000kg/month | WUHAN FORTUNA CHEMICAL CO., LTD | |
 |
2026-04-28 | Citral
5392-40-5
|
US $980.00-750.00 / kg | 1kg | 99% | 5000 | HEBEI SHENGSUAN CHEMICAL INDUSTRY CO.,LTD |