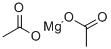마그네슘 아세트산
|
|
마그네슘 아세트산 속성
- 녹는점
- 72-75 °C(lit.)
- 밀도
- 1.5000
- 굴절률
- n
20/D 1.358
- 용해도
- H2O: 1 M at 20 °C, 투명, 무색
- 산도 계수 (pKa)
- 4.756[at 20 ℃]
- 물리적 상태
- 액체
- 색상
- white orthorhombic/monoclinic crystals, crystalline
- 수소이온지수(pH)
- 7.95(1 mM solution);8.23(10 mM solution);8.41(100 mM solution);8.78(1000 mM solution)
- 수용성
- 물과 섞이거나 혼합하기 어렵지 않습니다.
- 최대 파장(λmax)
- λ: 260 nm Amax: 0.04
λ: 280 nm Amax: 0.02
- Merck
- 13,5676
- BRN
- 3692530
- 안정성
- 안정적인. 강한 산화제와 호환되지 않습니다. 습기를 피하십시오.
- InChIKey
- UEGPKNKPLBYCNK-UHFFFAOYSA-L
- LogP
- -1.38
- CAS 데이터베이스
- 142-72-3(CAS DataBase Reference)
안전
- 위험 및 안전 성명
- 위험 및 사전주의 사항 (GHS)
| 그림문자(GHS): |
 
|
||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 신호 어: | Warning | ||||||||||||||||||||||||||||
| 유해·위험 문구: |
|
||||||||||||||||||||||||||||
| 예방조치문구: |
|
마그네슘 아세트산 C화학적 특성, 용도, 생산
개요
Anhydrous Magnesium Acetate has the chemical formula Mg(CH3COO)2 and in its hydrous form, Magnesium Acetate Tetrahydrate, it has the chemical formula Mg(CH3COO)2.4H2O. In this compound the magnesium metal has an oxidation state of 2+.
Magnesium acetate is the magnesium salt of acetic acid.It is deliquescent and upon heating, it decomposes to form magnesium oxide.Magnesium acetate is commonly used as a source of magnesium or as a chemical reagent.
화학적 성질
white deliquescent crystals물리적 성질
Magnesium acetate appears as white hygroscopic crystals. It smells like acetic acid and is soluble in water. When it is in an aqueous solution its pH can be considered to be neutral.Characteristics
In 1881 Charles Clamond invented the Clamond basket, one of the first effective gas mantles. The reagents used in this invention included magnesium acetate, magnesium hydroxide and water.Magnesium acetate is commonly used as a source of magnesium or for the acetate ion in chemistry experiments. One example of this is when magnesium acetate and magnesium nitrate were both used to perform molecular dynamics simulations and surface tension measurements. In the experiment the authors found that the acetate had a stronger affinity for the surface compared to the nitrate ion and that the Mg2+ strongly repelled away from the air/liquid interference. They also found that the Mg2+ had a stronger tendency to bind with the acetate ion compared to the nitrate.
One of the more prevalent uses of magnesium acetate is in the mixture called calcium magnesium acetate (CMA). It is a mixture of calcium acetate and magnesium acetate. CMA is thought of as an environmentally friendly alternative deicer to NaCl and CaCl2. CMA also acts as a powerful SO2, NOx, and toxic particulate emission control agent in coal combustion processes to reduce acid rain, and as an effective catalyst for the facilitation of coal combustion.
용도
Dye fixative in textile printing, deodorant, disinfectant, and antiseptic.Magnesium acetate is used in chemistry and molecular biology as a reagent and a source of magnesium. Magnesium has a variety of biological roles in enzymology, cell membrane and wall structural integrity, muscle cell physiology, and nucleic acid structure.Magnesium is an essential co-factor in many enzymes, including deoxyribonuclease (DNase), the restriction enzymes EcoR I and EcoR V, and Ribonuclease H. Magnesium also stabilizes polymeric nucleic acids such as transfer RNA and ribozymes.
Magnesium acetate has been widely used in the crystallization of proteins. A protocol for the separation of MB and BB isoenzymes of creatine kinase and the LD1 isoenzyme of lactate dehydrogenase that involves elution with magnesium acetate has been published.
정의
ChEBI: The magnesium salt of acetic acid.Safety
Magnesium Acetate is a relatively safe compound to handle and has been given a health hazard rating of zero. However, it should always be handled with gloves and safety goggles. If it is gets in the eyes, the skin, ingested, or inhaled it will cause irritation in the respective areas: eyes, skin, gastrointestinal system, and lungs.저장
Due to the fact that it is very hygroscopic, it must be stored away from water. It is also incompatible with strong oxidizers and should not be mixed with them.Purification Methods
Crystallise it from anhydrous acetic acid, then dry it under vacuum for 24hours at 100o. [Nencollas J Chem Soc 744 1956, Beilstein 2 IV 113.]마그네슘 아세트산 준비 용품 및 원자재
원자재
준비 용품
마그네슘 아세트산 공급 업체
글로벌( 275)공급 업체
| 공급자 | 전화 | 이메일 | 국가 | 제품 수 | 이점 |
|---|---|---|---|---|---|
| Hebei Mojin Biotechnology Co., Ltd | +86 13288715578 +8613288715578 |
sales@hbmojin.com | China | 12446 | 58 |
| Hebei Chuanghai Biotechnology Co,.LTD | +86-13131129325 |
sales1@chuanghaibio.com | China | 5882 | 58 |
| Hebei Jingbo New Material Technology Co., Ltd | +8619931165850 |
hbjbtech@163.com | China | 1000 | 58 |
| Hebei Saisier Technology Co., LTD | +86-18400010335 +86-18034520335 |
admin@hbsaisier.cn | China | 1009 | 58 |
| Hebei Zhuanglai Chemical Trading Co.,Ltd | +8613343047651 |
admin@zlchemi.com | China | 3002 | 58 |
| Hebei Longbang Technology Co., LTD | +86-18032476855 +86-18032476855 |
admin@hblongbang.com | China | 942 | 58 |
| Hebei Shengyang Water Conservancy Engineering Co., Ltd. | +8615373025980 |
clara@hbshengyang.com | China | 874 | 58 |
| HebeiShuoshengImportandExportco.,Ltd | +86-18532138899 +86-18532138899 |
L18532138899@163.com | China | 931 | 58 |
| career henan chemical co | +86-0371-86658258 +8613203830695 |
sales@coreychem.com | China | 29888 | 58 |
| Hubei Jusheng Technology Co.,Ltd. | 18871490254 |
linda@hubeijusheng.com | CHINA | 28180 | 58 |