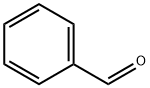
벤즈알데하이드
|
|
벤즈알데하이드 속성
- 녹는점
- ?26 °C (lit.)
- 끓는 점
- 178-179 °C (lit.)
- 밀도
- 1.044 g/cm3 at 20 °C (lit.)
- 증기 밀도
- 3.7 (vs air)
- 증기압
- 4 mm Hg ( 45 °C)
- FEMA
- 2127 | BENZALDEHYDE
- 굴절률
- n
20/D 1.545(lit.)
- 인화점
- 145 °F
- 저장 조건
- Store below +30°C.
- 용해도
- H2O: 가용성100mg/mL
- 산도 계수 (pKa)
- 14.90(at 25℃)
- 색상
- 옅은 노랑
- pH 범위
- 5.9
- 냄새
- 아몬드처럼.
- 수소이온지수(pH)
- 5.9 (1g/l, H2O)
- 폭발한계
- 1.4-8.5%(V)
- ?? ??
- 과일 같은
- 수용성
- 19.5 ºC에서 <0.01g/100mL
- 어는점
- -56℃
- 감도
- Air Sensitive
- JECFA Number
- 22
- Merck
- 14,1058
- BRN
- 471223
- Dielectric constant
- 17.8(20℃)
- 안정성
- 안정적인. 타기 쉬운. 강산화제, 강산, 환원제, 증기와 호환되지 않습니다. 공기, 빛, 습기에 민감합니다.
- InChIKey
- HUMNYLRZRPPJDN-UHFFFAOYSA-N
- LogP
- 1.4 at 25℃
- CAS 데이터베이스
- 100-52-7(CAS DataBase Reference)
안전
- 위험 및 안전 성명
- 위험 및 사전주의 사항 (GHS)
| 위험품 표기 | Xn | ||
|---|---|---|---|
| 위험 카페고리 넘버 | 22 | ||
| 안전지침서 | 24 | ||
| 유엔번호(UN No.) | UN 1990 9/PG 3 | ||
| WGK 독일 | 1 | ||
| RTECS 번호 | CU4375000 | ||
| F 고인화성물질 | 8 | ||
| 자연 발화 온도 | 374 °F | ||
| TSCA | Yes | ||
| HS 번호 | 2912 21 00 | ||
| 위험 등급 | 9 | ||
| 포장분류 | III | ||
| 유해 물질 데이터 | 100-52-7(Hazardous Substances Data) | ||
| 독성 | LD50 in rats, guinea pigs (mg/kg): 1300, 1000 orally (Jenner) | ||
| 기존화학 물질 | KE-02713 |
벤즈알데하이드 C화학적 특성, 용도, 생산
물성
산화하기 쉽고, 공기 속에서 쉽게 벤조산이 되므로, 오랫동안 보존할 때는 주의를 요한다. 산화에 의하여 생긴 벤조산은 벤즈알데하이드에 녹지 않으므로 표면에 막 모양의 물질이 되어 위로 뜬다. 또 수산화알칼리의 작용으로 벤조산과 벤질알코올로, 환원시키면 벤질알코올이 된다.개요
벤즈알데하이드(Benzaldehyde, C7H6O)는 가장 간단한 방향족 알데하이드다. 고편도유의 성분 중 하나이다.요약 방향족 알데하이드 중의 가장 간단한 것으로 특수한 향내가 나는 무색의 액체이다. 공기 중에서 쉽게 산화하여 벤조산이 되므로 보존에 주의해야 하며, 비교적 값싼 향료로서 많이 사용되고 있다.용도
비교적 값싼 향료로서 비누·화장품 등에 다량으로 사용되며, 또한 고편도유의 대용으로도 사용된다.순도시험
(1) 비중 : 이 품목의 비중은 1.041~1.046이어야 한다.
(2) 굴절률 : 이 품목의 굴절률 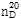 은 1.544~1.547이어야 한다.
은 1.544~1.547이어야 한다.
(3) 염소화합물 : 이 품목은 향료시험법 중 할로겐시험법의 동망법에 따라 시험할 때, 이에 적합하여야 한다.
확인시험
(1) 이 품목 1mL에 아황산수소나트륨시액 3mL를 가하여 흔들어 섞으면 곧 발열하여 결정덩어리가 되고 이에 물 5mL를 가하면 녹는다.
(2) 이 품목 3방울에 페놀 0.1g 및 황산 2mL를 가하여 흔들어 섞으면 암적색이 되고 일부는 수지화한다. 이 액 2방울을 취하여 물 5mL를 가하고 수산화나트륨시액으로 알칼리성이 되게 하면 자색을 나타낸다.
정량법
이 품목은 약 0.8g을 정밀히 달아 향료시험법 중 알데히드류 및 케톤류함량측정법의 히드록실아민법 제2법에 따라 정량한다. 다만, 방치시간은 10분으로 한다.
0.5N 염산 1mL = 53.06mg C7H6O
화학적 성질
Benzaldehyde is a colorless to yellow, oily liquid with an odor of bitter almonds. Benzaldehyde is commercially available in two grades: (i) pure benzaldehyde and (ii) and double-distilled benzaldehyde. The latter has applications in the pharmaceutical, perfume, and fl avor industries. Benzaldehyde may contain trace amounts of chlorine, water, benzoic acid, benzyl chloride, benzyl alcohol, and/or nitrobenzene. Benzaldehyde is ignited relatively easily on contact with hot surfaces. This has been attributed to the property of very low auto-ignition temperature. Benzaldehyde also undergoes autoxidation in air and is liable to self-heat. It is used as a food flavoring and in the manufacture of dyes and antibiotics, and can be readily manufactured by the chlorination of methylbenzene and the subsequent hydrolysis of (dichloromethyl) benzene: C6H5CH3 + Cl2 →C6H5CHCl2C6H5CHCl2 + 2H2O →C6H5CH(OH)2+ 2HCl C6H5CH(OH)2 →C6H5CHO + H2O.출처
Benzaldehyde exists in nature, occurring in combined and uncombined forms in many plants. It is also the main constituent of the essential oils obtained by pressing the kernels of peaches, cherries, apricots, and other fruits. Benzaldehyde is released into the environment in emissions from combustion processes, such as gasoline and diesel engines, incinerators, and wood burning. It is formed in the atmosphere through photochemical oxidation of toluene and other aromatic hydrocarbons.용도
Benzaldehyde is a flavoring agent which is liquid and colorless, and has an almond-like odor. it has a hot (burning) taste. it is oxidized to benzoic acid when exposed to air and deteriorates under light. it is miscible in volatile oils, fixed oils, ether, and alcohol; it is spar- ingly soluble in water. it is obtained by chemical synthesis and by natural occurrence in oils of bitter almond, peach, and apricot kernel. it is also termed benzoic aldehyde.정의
ChEBI: Benzaldehyde is an arenecarbaldehyde that consists of benzene bearing a single formyl substituent; the simplest aromatic aldehyde and parent of the class of benzaldehydes. It has a role as a flavouring agent, a fragrance, an odorant receptor agonist, a plant metabolite, an EC 3.5.5.1 (nitrilase) inhibitor and an EC 3.1.1.3 (triacylglycerol lipase) inhibitor.제조 방법
Benzaldehyde is prepared by hydrolysis of benzal chloride, for example, in acidic media in the presence of a catalyst such as ferric chloride or in alkaline media with aqueous sodium carbonate. Part of the commercially available benzaldehyde originates from a technical process for phenol. In this process, benzaldehyde is a by-product in the oxidation, in air, of toluene to benzoic acid.일반 설명
Benzaldehyde appears as a clear colorless to yellow liquid with a bitter almond odor. Flash point near 145 °F. More denser than water and insoluble in water. Hence sinks in water. Vapors are heavier than air. The primary hazard is to the environment. Immediate steps should be taken to limit spread to the environment. Easily penetrates the soil to contaminate groundwater and nearby waterways. Used in flavoring and perfume making.공기와 물의 반응
Oxidizes in air to form benzoic acid, which is moderately toxic by ingestion. Insoluble in water.반응 프로필
A nontoxic, combustible liquid, reacts with oxidizing reagents. Benzaldehyde must be blanketed with an inert gas at all times since Benzaldehyde is oxidized readily by air to benzoic acid [Kirk-Othmer, 3rd ed., Vol. 3, 1978, p. 736]. In contact with strong acids or bases Benzaldehyde will undergo an exothermic condensation reaction [Sax, 9th ed., 1996, p. 327]. A violent reaction was observed on contact with peroxyacids (peroxyformic acid) [DiAns, J. et al., Ber., 1915, 48, p. 1136]. An explosion occurred when pyrrolidine, Benzaldehyde, and propionic acid were heated to form porphyrins.위험도
Highly toxic.건강위험
Benzaldehyde exhibited low to moderate toxicityin test animals, the poisoning effectdepending on dosage. Ingestion of 50–60 mLmay be fatal to humans. Oral intake of a largedose can cause tremor, gastrointestinal pain,and kidney damage. Animal experimentsindicated that ingestion of this compoundby guinea pigs caused tremor, bleeding fromsmall intestine, and an increase in urine volume;in rats, ingestion resulted in somnolenceand coma.LD50 value, oral (guinea pigs): 1000 mg/kg
LD50 value, oral (rats): 1300 mg/kg
A 500-mg amount for a 24-hour periodresulted in moderate skin irritation in rabbits.Because of its low toxicity, high boilingpoint, and low vapor pressure, the healthhazard to humans from exposure to benzaldehydeis very low.
화재위험
HIGHLY FLAMMABLE: Will be easily ignited by heat, sparks or flames. Vapors may form explosive mixtures with air. Vapors may travel to source of ignition and flash back. Most vapors are heavier than air. They will spread along ground and collect in low or confined areas (sewers, basements, tanks). Vapor explosion hazard indoors, outdoors or in sewers. Runoff to sewer may create fire or explosion hazard. Containers may explode when heated. Many liquids are lighter than water.화학 반응
Reactivity with Water: No reaction; Reactivity with Common Materials: No reactions; Stability During Transport: Stable; Neutralizing Agents for Acids and Caustics: Not pertinent; Polymerization: Not pertinent; Inhibitor of Polymerization: Not pertinent.Safety Profile
Poison by ingestion and intraperitoneal routes. Moderately toxic by subcutaneous route. An allergen. Acts as a feeble local anesthetic. Local contact may cause contact dermatitis. Causes central nervous system depression in small doses and convulsions in larger doses. A skin irritant. Questionable carcinogen with experimental tumorigenic data. Mutation data reported. Combustible liquid. To fight fire, use water (may be used as a blanket), alcohol, foam, dry chemical. A strong reducing agent. Reacts violently with peroxyformic acid and other oxidizers. See also ALDEHYDES.잠재적 노출
In manufacture of perfumes, dyes, and cinnamic acid; as solvent; in flavors.신진 대사
Benzaldehyde was among 300 volatile constituents detected in the urine of ten adults . It is commonly converted to hippuric acid in vivo. In the rabbit and dog, hippuric acid appears to be the only metabolite there being practically no formation of benzoyl glucuronide. The conversion of benzaldehyde to benzoic acid in the rabbit follows first-order reaction kinetics저장
Benzaldehyde should be kept stored in a tightly closed container and protected against physical damage. Storage of the chemical substance outside or in a detached area is preferred, whereas inside storage should be in a standard flammable liquids storage room or cabinet. Benzaldehyde should be kept separated from oxidizing materials. Also, storage and use areas should be no smoking areas. Containers of this material may be hazardous when empty since they retain product residues (vapors, liquid); observe all warnings and precautions listed for the product운송 방법
UN1990 Benzaldehyde, Hazard class: 9; Labels: 9—Miscellaneous hazardous material.Purification Methods
To diminish its rate of oxidation, benzaldehyde usually contains additives such as hydroquinone or catechol. It can be purified via its bisulfite addition compound but usually distillation (under nitrogen at reduced pressure) is sufficient. Prior to distillation it is washed with NaOH or 10% Na2CO3 (until no more CO2 is evolved), then with saturated Na2SO3 and H2O, followed by drying with CaSO4, MgSO4 or CaCl2. [Beilstein 7 IV 505.]비 호환성
The substance reacts with air, forming explosive peroxides. Reacts violently with performic acid, oxidants, aluminum, iron, bases, and phenol, causing fire and explosion hazard. May self-ignite if absorbed in combustible material with large surface area, or otherwise dispersed over large areas. Reacts with rust, amines, alkalies, strong bases, reducing agents such as hydrideds and active metals.폐기물 처리
Incineration; add combustible solvent and spray into incinerator with afterburner.주의 사항
Workers should be careful when using benzaldehyde because there is a risk of spontaneous combustion. It may ignite spontaneously if it is absorbed onto rags, cleaning cloths, clothing, sawdust, diatomaceous earth (kieselguhr), activated charcoal, or other materials with large surface areas in workplaces. Workers should avoid handling the chemical substance and should not cut, puncture, or weld on or near the container. Exposure of benzaldehyde to air, light, heat, hot surfaces such as hot pipes, sparks, open flames, and other ignition sources should be avoided. Workers should wear proper personal protective clothing and equipment벤즈알데하이드 준비 용품 및 원자재
원자재
일산화탄소(가스)
염화벤질
벤질 알코올
산화아연
톨루엔
염소(기체)
팔라듐
몰리브덴 무수물
인산 아연
오존
아미그달린
trans-Cinnamaldehyde
염화 알루미늄
시나몬 기름
탄산나트륨(경회)
준비 용품
DL-만델산
FLAVANONE
2-amino-5-chloro-diphenyl methanol
Propafenone Hydrochloride
TRANS-2-PHENYL-1-CYCLOPROPANECARBOXYLIC ACID
whitener WG for wool
Benzenemethanol, ar-methyl-, acetate
BENZYLHYDRAZINE DIHYDROCHLORIDE
헥실신나믹알데히드
Diaveridine
광택이나는 BLUE G-250
2,4,5-트리페닐이미다졸
L-Phenylglycine
3,4-Dichlorobenzylamine
Magentagreencrystals
Reactive Blue BRF
4-페닐부트-3-엔-2-온
페니토인
1-AMINO-4-METHYLPIPERAZINE DIHYDROCHLORIDE MONOHYDRATE
2-(Acetylamino)-3-phenyl-2-propenoic acid
Methyl indole-2-carboxylate
산 청색 9 (디암모늄 염)
2-[2-(4-Fluorophenyl)-2-oxo-1-phenylethyl]-4-methyl-3-oxo-N-phenylpentanamide
N, N- 디벤질에틸렌 디암무늄 디 (아세트산)
Bis(dibenzylideneacetone)palladium
ASTRAZON BRILLIANT RED 4G
Reactive Blue 104
NIFE디솔
Epalrestat
4-Hydroxybenzylideneacetone
2-PHENYL-1.3-DIOXOLANE-4-METHANOL
3,5-디페닐피라졸
2-((E)-2-Hydroxy-3-phenylacryloyl)benzoic acid ,97%
(R)-(+)-N-Benzyl-1-phenylethylamine
N,N'-BISBENZYLIDENEBENZIDINE
니트로 BT
L-Arginine hydrochloride
(E)-3-Benzylidene-3H-isochromene-1,4-dione ,97%
염화 2,3,5-트리페닐 테트라졸륨
트리스 (디 벤질 리덴 아세톤) 디 팔라듐
벤즈알데하이드 공급 업체
글로벌( 1054)공급 업체
| 공급자 | 전화 | 이메일 | 국가 | 제품 수 | 이점 |
|---|---|---|---|---|---|
| Hubei Qianjia Chemical Co., Ltd. | +8618972721089 |
sales@meijiachem.com.cn | China | 2 | 58 |
| Hefei TNJ Chemical Industry Co.,Ltd. | +86-0551-65418679 +86-18949832763 |
info@tnjchem.com | China | 2989 | 55 |
| Wuhan Biet Co., Ltd. | +8617320528784 |
min@biet.com.cn | China | 41 | 58 |
| Jiangsu Boquan Biotechnology Co., Ltd. | +86-18168774353 |
boquanshengwu003@boquansw.com | China | 248 | 58 |
| Nanjing Deda New Material Technology Ltd. | +8613223281135 |
niki@njdeda.com | China | 76 | 58 |
| Hebei Guanlang Biotechnology Co., Ltd. | +86-19930503282 |
alice@crovellbio.com | China | 8823 | 58 |
| Hunan aslsen technology co.,ltd | +8619313031929 |
aslsd@aslsen.com | China | 128 | 58 |
| Hebei Mojin Biotechnology Co., Ltd | +8613288715578 |
sales@hbmojin.com | China | 12456 | 58 |
| Anhui Yiao New Material Technology Co., Ltd | +86-199-55145978 +8619955145978 |
sales8@anhuiyiao.com | China | 253 | 58 |
| Anhui Ruihan Technology Co., Ltd | +8617756083858 |
daisy@anhuiruihan.com | China | 994 | 58 |